Science - CLASSIFICATION OF MATTER
1/28
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
29 Terms
Can you separate this item by physical means? (E.g temp, filter, density, spinning) if yes, it is a:
Mixture
How many phases in a mixture?
2 or more
If a mixture is uniform, is it homogeneous or heterogeneous
Homogenous
If a mixture is NOT uniform, is it heterogeneous or homogeneous?
Heterogeneous
What are the 2 types of homogeneous?
Solution, suspension
What is one type of heterogeneous?
Mechanical mixture
What’s a solute
a substance that can be dissolved into a solution by a solvent.
What’s a solution
a mixture of substances that are evenly mixed
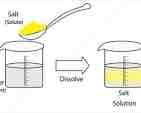
What’s this
Solution
What’s a solvent
a substance that dissolves a solute, resulting in a solution.
Does solute have more or less substance?
has less % of a substance than a solvent in a solution.
Solute example
Salt, sugar
Does solvent have more or less substance?
Has more % of a substance than solute in a solution.
Example of solvent
Alcohol, water
What is a suspension?
Only separates by spinning
Examples of suspensions:
Laundry detergent, milk
What is a mechanical mixture?
A mechanical mixture cannot be mixed to form another substance.
Examples of mechanical mixtures
Sand + water, oil + water
What does m=
Slope
What is mass over?
Volume
Do you divide by mass or volume
Mass / volume

Solve image
10 - 5=5, 6-4=2, 5/2 = 2.5 g/m3
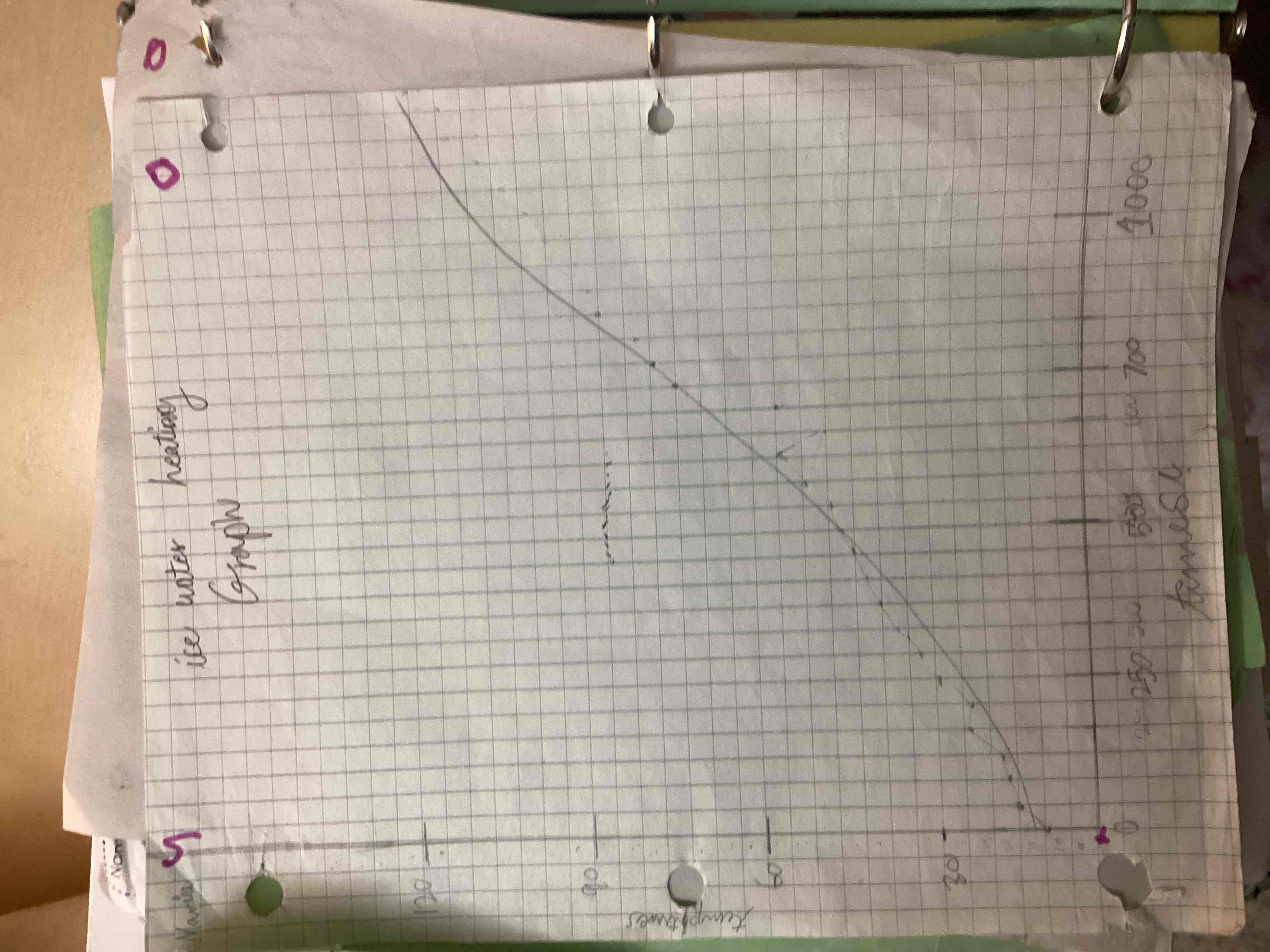
What’s the temps true when the time reaches 700?
60
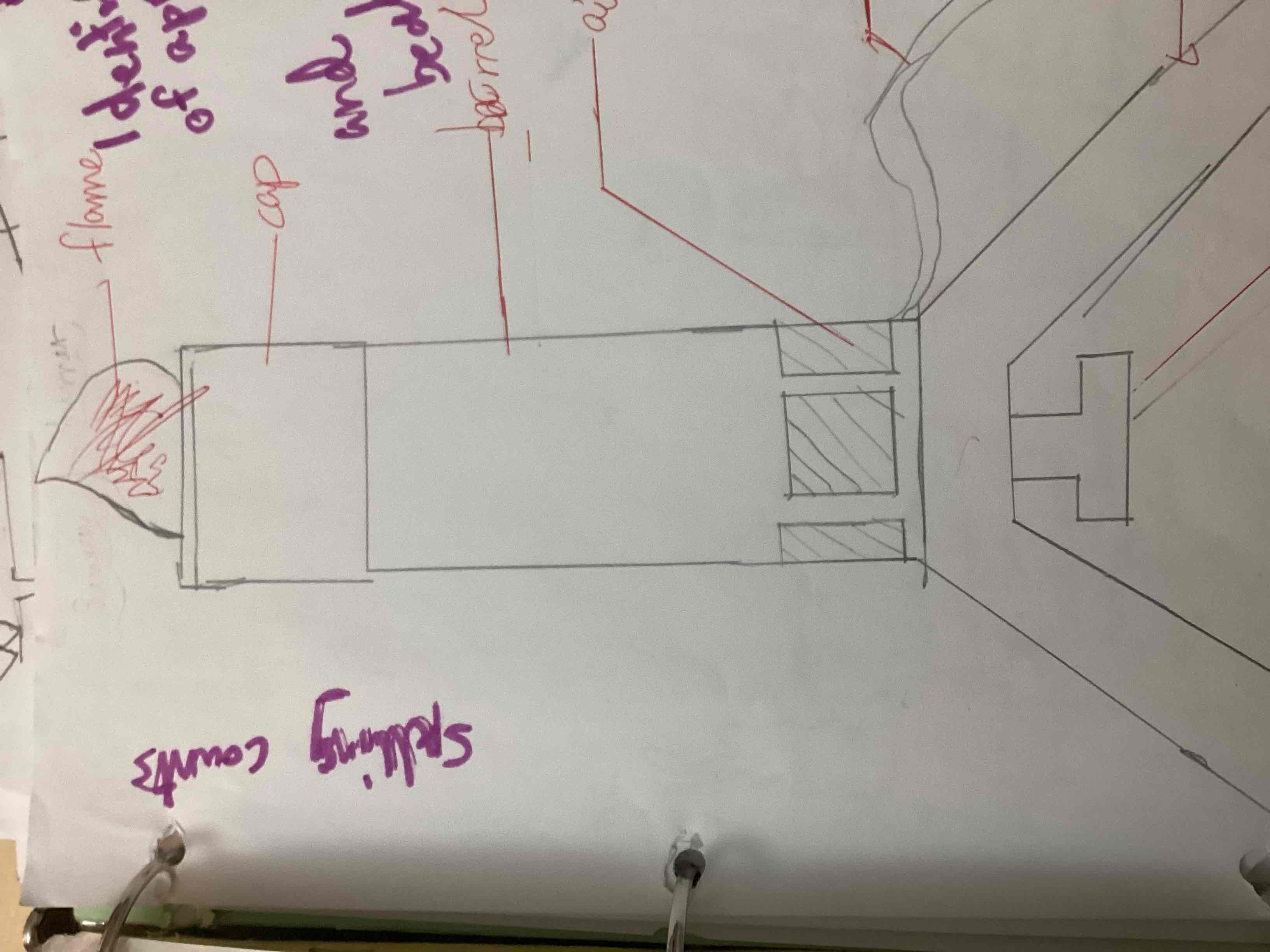
What’s this
Bunsen burner
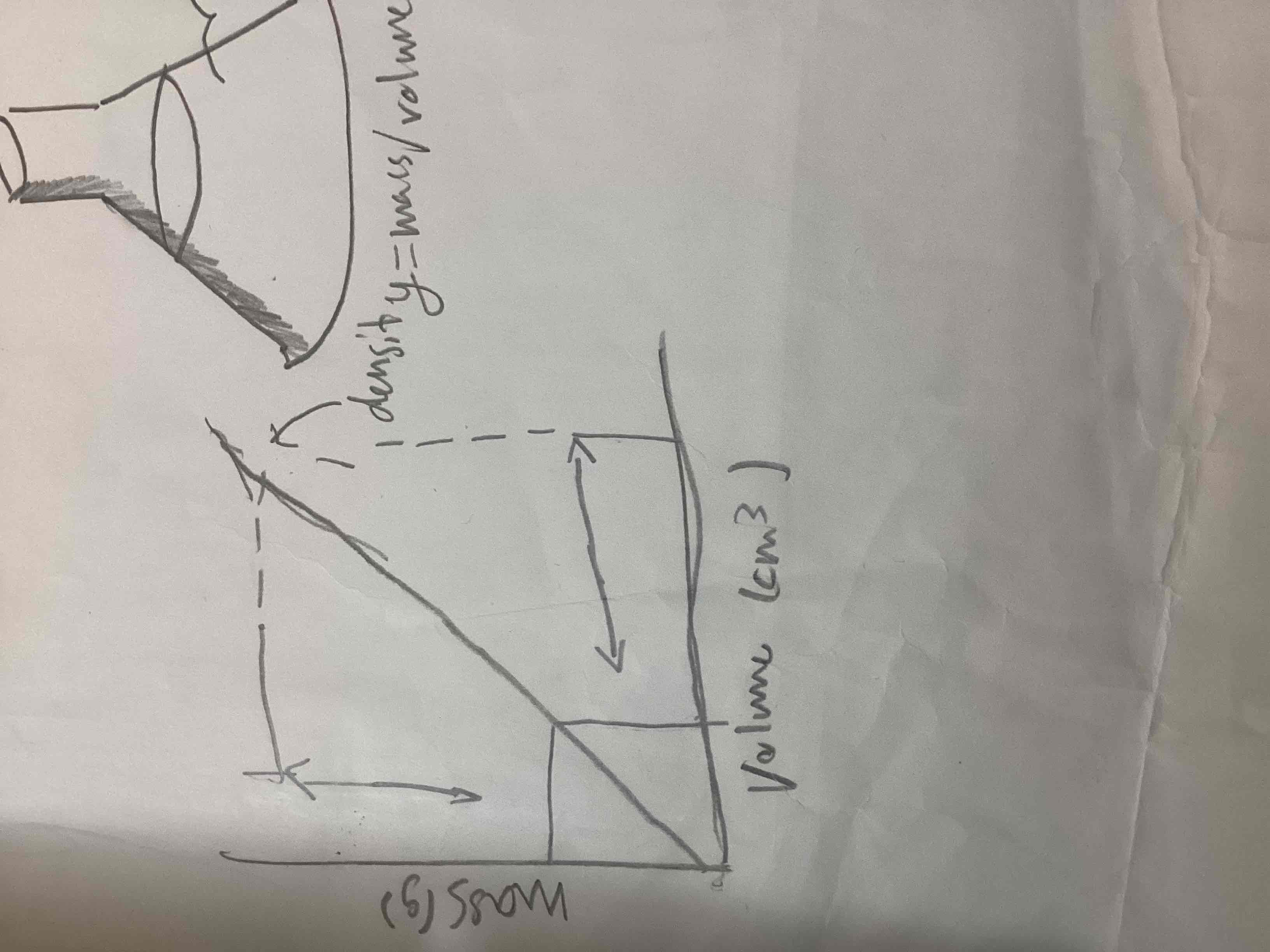
What does density equal
Mass/volume
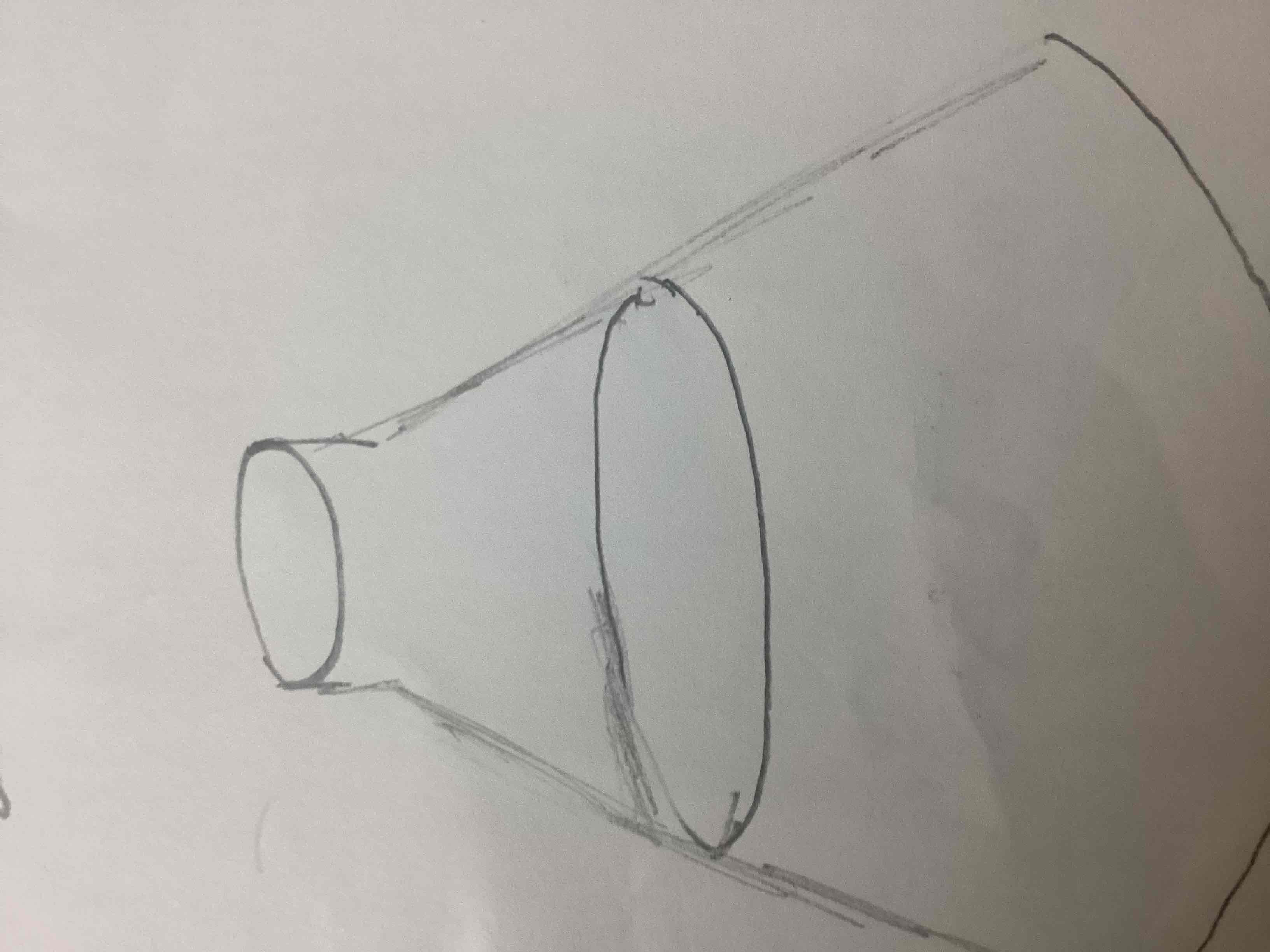
What’s this
Erlenmeyer flask
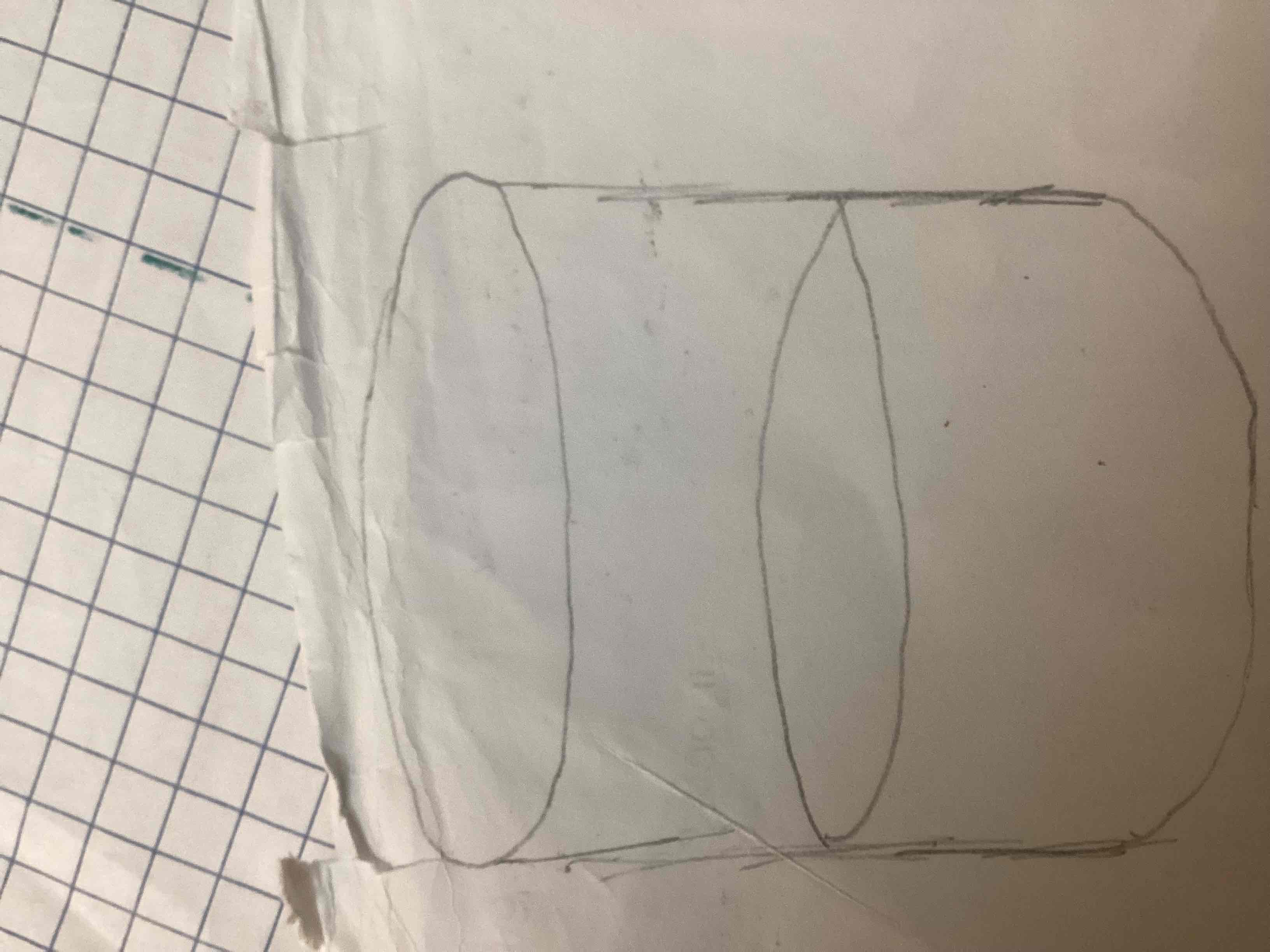
Whaa this
Beaker
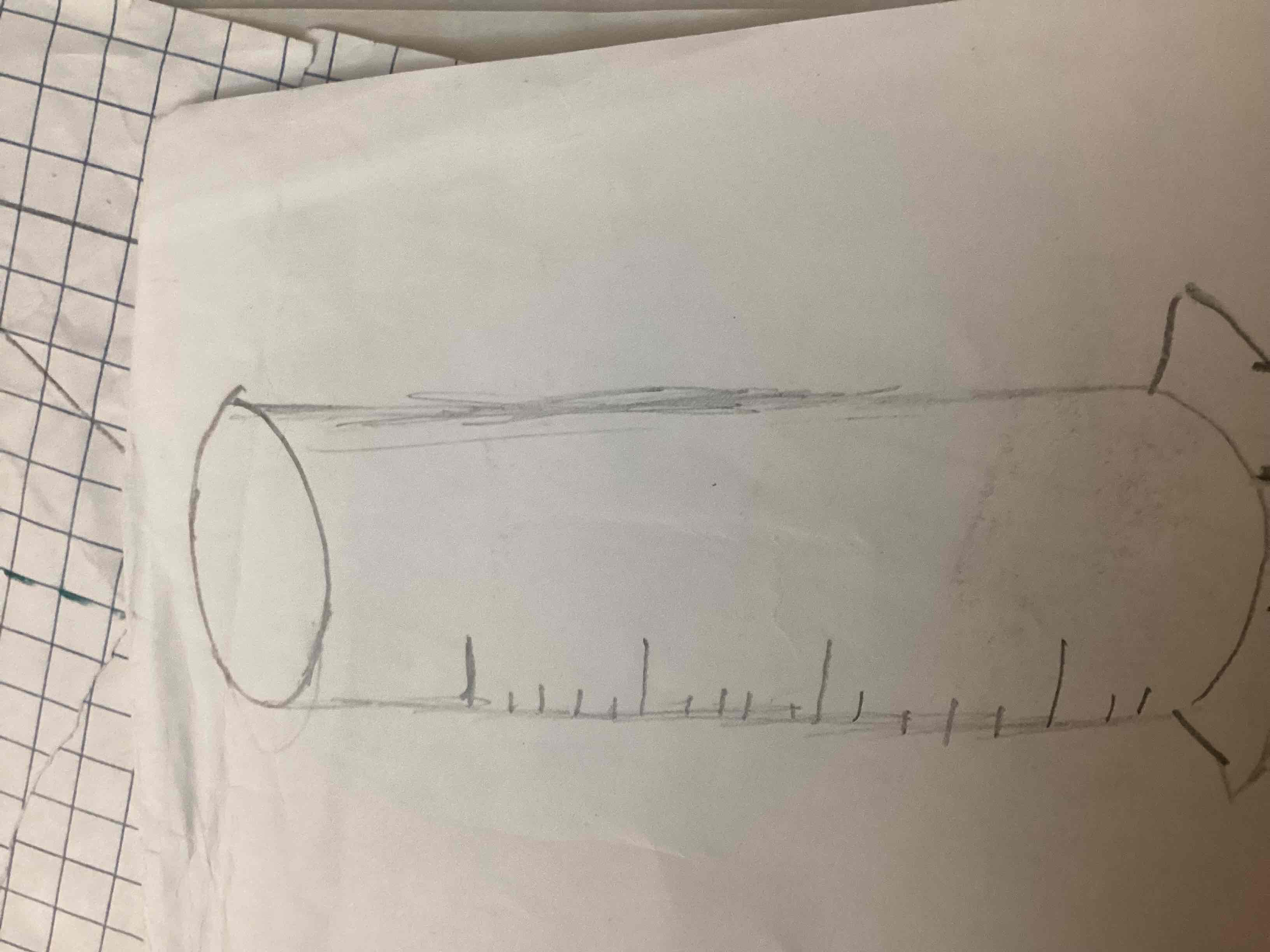
What’s this
Graduated cylinder
What8s change
Subtraction