energetics- endothermic and exothermic reactions.
1/65
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
66 Terms
why is it important to know the amount of energy required for chemical reactions to take place?
• can measure the energy values of fuels ( amount of energy released when 1 gram of fuel is burned)
• can calculate the energy requirements for industrial processes
• work out the theoretical amount or energy to break bonds, and the amount of energy released when bonds are made
• it helps to predict whether or not a reaction will take place.
what forms of energy may be involved?
(The energy involved may be in different forms )-
light.
electrical
(most usually) heat
what is thermochemistry?
the study of heat changes during chemical reactions.
What is Enthalpy changes?
Enthalpy change is the name given to the amount of heat evolved or absorbed in a reaction carried out at constant pressure. It is given the symbol ΔH, read as "delta H".(term "enthalpy change" only applies to reactions done at constant pressure)
Standard enthalpy changes
Standard enthalpy changes refer to reactions done under standard conditions, and with everything present in their standard states
Standard states
The standard states of elements are the forms that they adopt at a temperature of 25°C and pressure of 1 atmosphere (1 atm).- (standard temperature and pressure).
Standard conditions
298 K (25°C)
a pressure of 1 bar (100 kPa).
where solutions are involved, a concentration of 1 mol dm-3
Standard states
For a standard enthalpy change everything has to be present in its standard state. That is the physical and chemical state that you would expect to find it in under standard conditions
Standard states of allotropes
For elements which have allotropes (two different forms of the element in the same physical state), the standard state is the most energetically stable of the allotropes.
symbol for standard enthalpy changes
ΔH⦵r
-To denote that the enthalpy change has been calculated at standard pressure and temperature, symbol ⦵ is used
General definition of Standard enthalpy change of reaction, ΔH°r /(Standard change of enthalpy).
The standard enthalpy change of a reaction is the enthalpy change which occurs when equation quantities of materials react under standard conditions, and with everything in its standard state.
/
(The standard enthalpy change of a reaction is the enthalpy change when the amount of reactants shown in balanced equation at standard pressure and temperature react to give products at standard pressure and temperature.)
What does kJ mol-1 refer to in enthalpy change equations?
(kilojoules per mole) doesn't refer to any particular substance in the equation.
Instead it refers to the quantities of all the substances given in the equation. In this case, 572 kJ of heat is evolved when
2 moles of hydrogen gas react with 1 mole of oxygen
gas to form 2 moles of liquid water.kJ mol-1
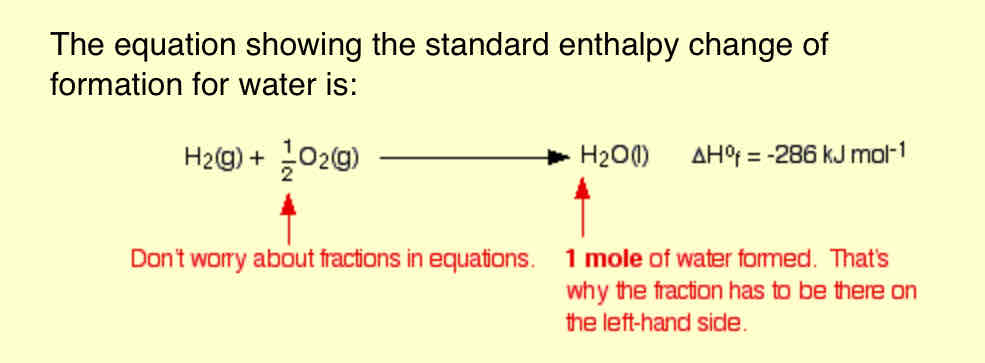
Define Standard enthalpy change of formation, ΔH°f
The standard enthalpy change of formation of a compound is the enthalpy change which occurs when one mole of the compound is formed from its elements under standard conditions, and with everything in its standard state.
standard enthalpy change of formation example:
standard enthalpy change of formation of an element
★The ΔH⦵f of an element in its
standard state is zero.
(For example, if you "make" one mole of hydrogen gas starting from one mole of hydrogen gas you aren't changing it in any way, so you wouldn't expect any enthalpy change.)
( equally true of any other element. ΔH⦵f of any element = 0 —→ cuz of the way ΔH is defined)
Standard enthalpy change of combustion, ç
The standard enthalpy change of combustion of a compound is the enthalpy change which occurs when one mole of the compound is burned completely in oxygen(excess) under standard conditions, and with everything in its standard state
Activation energy
The minimum energy that colliding reactant particles must possess in order to start a reaction.
Standard enthalpy change of neutralisation
The standard enthalpy change of neutralisation is the enthalpy change when 1 mol of water is formed when acid and alkali react under standard conditions.
★ ALWAYS MEASURED —> PER MOL OF WATER FORMED
ALWAYS- NEGATIVE (energy is given out- when acids and alkali react).
enthalpy change- EQUATION
ΔH=mcΔT
(-where m is the mass of water in grams,
-c is the specific heat capacity of water (4.18 J/g⁰C)
-ΔT is the change in temperature in degree Celsius (⁰C)
calorimetry assumptions
In calorimetry, acids, alkalis and salts are assumed to consist of large amounts of water .
• In case of solutions, it is assumed that 1 cm3 of
solution has 1 g.
• It is also assumed that the specific heat capacity of solutions = same as water.
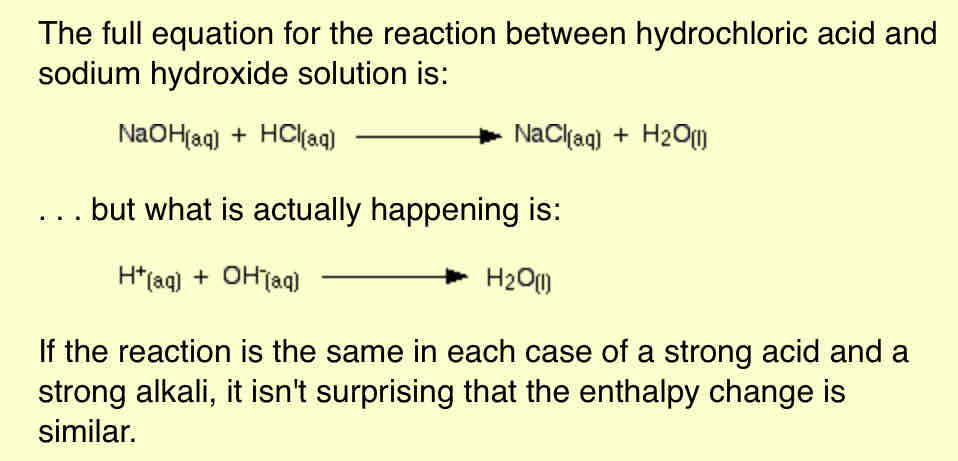
Why do strong acids reacting with strong alkalis give closely similar values?
We assume that strong acids and strong alkalis are fully ionised in solution, and that the ions behave independently of each other.
For example, dilute hydrochloric acid contains hydrogen ions and chloride ions in solution. Sodium hydroxide solution consists of sodium ions and hydroxide ions in solution.
-The equation for any strong acid being neutralised by a strong alkali is essentially just a reaction between hydrogen ions and hydroxide ions to make water. The other ions present (sodium and chloride, for example) are just spectator ions, taking no part in the reaction.
Biofuels reactions
Plants producing glucose (photosynthesis):
6CO2+ 6H2O —> C6H12O6+6 O2
Fermentation:
Glucose to ethanol:
C6H12O6–> 2CO2+2 C2H5OH
Ethanol combustion:
2C2H5OH+ 6O2 —> 4CO2+ 6H2O
Biofuels
Argued to be carbon neutral—> cuz total number of moles of CO2 produced
=
Total moles of CO2 absorbed (6 moles)
OXIDATION OF ALCOHOLS
Alcohols—> can be oxidized to :
Aldehydes
Ketones
Carboxylic acids
Reagent: ACIDIFIED POTASSIUM DICHROMATE ( K2CR2O7)
(Mild oxidizing agent -(reduces itself))
Reagent of Oxidation of alcohol
Acidified potassium dichromate
Notes
In some cases—> enthalpy change can be called different names.
Example→ enthalpy change of formation of CO2= enthalpy change of combustion of carbon
-only ONE MOLE of substance—> must be formed—> even if fractions have to be used in the equation.
+you MUST always START WITH THE ELEMENTS—> the ONLYcompound in equation—> one being formed.
EVERYTHING must be PRESENT —> in their STANDARD STATE- (THE STATE YOU WOULD EXPECT IT TO BE IN AT 298K and 1 atm
Standard state (definitions)
The state an element is expected to be at in standard conditions—>
Temp: 298K
Pressure: 1 atm
(Also implies to the fact—> hydrogen is H2 molecules, Nitrogen is N2 molecules (diatomic). —>(this is hope they exist in std. conditions)
Notes:
Over all enthalpy change—> determined by the gap between the reactants and the products- on the enthalpy diagram, and not affected by the route used to get from the reactants to the products.
(Whichever route you use the overall enthalpy change is the same )
Enthalpy change of formation of an element
ALWAYS—> ZERO
(In std. state)
Hess’s law
The enthalpy change accompanying a chemicals change is independent of the route by which the chemical change occurs
-my interpretation -Meaning—> (the total enthalpy change from the reactants to the products will remain the same- regardless of the route the chemical change takes to reach from the reactants to the product(reactant and product needs to be the same)).
-Hess's Law is saying that if you convert reactants A into products B, the overall enthalpy change will be exactly the same whether you do it in one step or two steps or however many steps.
(Hess's Law is saying that if you convert reactants A into products B, the overall enthalpy change will be exactly the same whether you do it in one step or two steps or however many steps.)
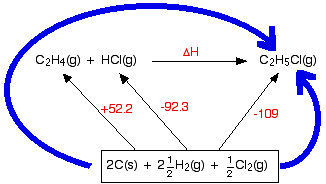
Key notes- when drawing Hess’s law diagram
You will notice that I haven't bothered to include the oxygen that the various things are burning in. The amount of oxygen isn't critical because you just use an excess anyway, and including it really confuses the diagram.
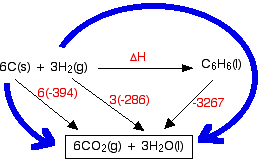
Designing Hess’s cycle
Write down the enthalpy change you want to find as a simple horizontal equation, and write ΔH over the top of the arrow. (In diagrams of this sort, we often miss off the standard symbol just to avoid clutter.)
Then fit the other information you have onto the same diagram to make a Hess's Law cycle, writing the known enthalpy changes over the arrows for each of the other changes.
Finally, find two routes around the diagram, always going with the flow of the various arrows. You must never have one of your route arrows going in the opposite direction to one of the equation arrows underneath it.
(Notice that you may have to multiply the figures you are using. For example, standard enthalpy changes of combustion start with 1 mole of the substance you are burning. In this case, the equations need you to burn 6 moles of carbon, and 3 moles of hydrogen molecules. Forgetting to do this is probably the most common mistake you are likely to make.
How were the two routes chosen? Remember that you have to go with the flow of the arrows. Choose your starting point as the corner that only has arrows leaving from it. Choose your end point as the corner which only has arrows arriving.)
Which direction does the rows point in ΔH°c And ΔH°f
ΔH°C ——> arrows point DOWN
ΔH°f ——> arrows point UP
Hess’s cycle for —→ ΔH°c of benzene
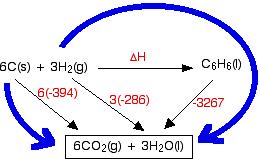
Hess’s cycle for —→ ΔH°f of reaction between ethene and hydrogen chloride gases to make chloroethane gas
Calomitery (why are polystyrene cups used)?
-An expanded polystyrene cup is used.
-It’s a good insulator -helping it prevent heat losses
-also cuz it hardly absorbs any heat itself
Calorimetry calculations
Heat evolved/ absorbed = mcΔT
M= mass
c= specific heat capacity
ΔT= change in temperature
what determines amount of energy given out/ taken in ?
-QUANTITY OF REACTANTS
(amount of energy given out or taken in- during chemical reaction depends on- reactant quantities).
what is the importance of studying thermochemistry ?
-enables us to compare the efficiency of different fuels.
(crude oil (non- rewnewable) alternatives/ replacements, are being researched on)
(inc.
Hydrogen produced from water electrolysis
ethanol from plant material
specific heat capacity
amount of energy needed to raise the temperature of1 gof a substance by 1 K(or 1 ˚C)
quantities
The amount of heatgiven out or taken in during a chemical reaction depends on the quantity of reactants.
This energy is usually measured in kilojoules per mole, kJ mo1 -1
useful enthalpy changes
when fuels- burnt => ↑ large heat output => VERY ↑EXOTHERMIC REACTIONS.
EXAMPLES;
(For example, coal is mostly carbon—> (Carbon gives out 393.5 kJ when one mole, 12 g) )
C(s) + 0 2 (g) -. C02 (g)
natural gas, methane, gives out 890kJ when one mole is burnt completely ro carbon dioxide and water.
Physiotherapists often treat sports injuries —> with cold packs. these produce 'coldness' by an endothermic reaction such as:
[NH4N03(S) + (aq)-. NH4N03(aq)”
This absorbs 26 kJ mo1 -1 —>of heat energy.
importance of thermochemistry study
-One important practical application of the study of thermochemistry is that it enables us to compare the efficiency of different fuels.
(most common fuels are from crude oils- non rewnewable
=> therefore researching on alternatives ny comparing the energy
→ For use as fuels:
,the energy given out per gram of fuel burned, or the
★energy density of a fuel, is more important.
what is a important factor when considering a fuels efficiency?
ENERGY DENSITY OF A FUEL
/ or/
ENERGY GIVEN OUT PER GRAM OF FUEL BURNT
(hydrogen is very high in energy per gram but is a gas—> therefore hard to store)
enthalpy change
measuring heat change at a constant temperature
reasoning for std. contitions
The amount of heat given out or taken in by (∆H) a reaction varies with solution conditions:
temperature
pressure
concentration
temperature relation to reaction key points
though reaction itself causes a —>∆temp.(due to ∆ Heat)
L >
Heat changes—> measured in constant temperature (temp. 298K of the reactants)
★ reaction isnt thought of reaching end => until product cools back down to 298k)
★(the heat given out to surroundings→ while reaction mixture cools down
=>
the ∆H - (of reaction)
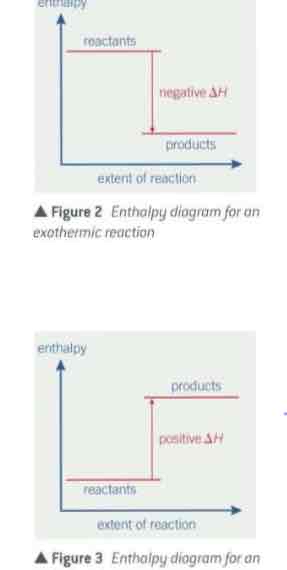
∆ H reasoning for exothermic reactions
exo. reactions—> products have ↓ less energy than reactants- cuz heat energy is lost to the surroundings- when they heat up the surroundings.
∆H => negative (-)
endothermic reactions reasoning
★endo. reactions- taking place in aqeous solutions → heat from water is absorbed ( water temp lowers/ cools)
+(reactionisn’t thought of being over until prod. have ↑warmed up to temp. of reactants( temp they started)
-(reaction (the entire system incl. the water ) then takes in heat from the. surroundings → is why reaction which is absorbing heat( endo.) gets cooler at first )
Notes
Don't be confused by the different
terms. Heat is a form of energy
so a heat change can also be
described as an energy change.
An enthalpy change is still an
energy change but it is measured
under stated conditions of
temperature and pressure.
One way of making sure that
both reactants are at the same
temperature is simply to leave
them in the same room for
sometime.
pressures affect on heat energy (∆H)
★Pressureaffects the amount of heat energy given out by reactions thatinvolve gases
+reactions producing gas→ require some energy to push away atmosphere-
= so ↑ GREATER THE ATMOSPHERIC PRESSURE = ↑ THE MORE ENERGY USED UP IN PUSHING AWAY ATMOSPHERE WHEN GAS IS RELEASED= ↓ LESS ENERGY LEFT TO PRODUCE AS HEAT ENERGY BY THE REACTION
+(therefore important to have std. pressure- measuring energy changes)
the physical state of reactants and products
the physical state (s, l or g)=> of reactants and products affect the ∆H of reaction
example- ( liquid requires heat to change into gas)
( gas gives out heat when change into liquid)
Enthalpy level diagrams
enthalpy level diagrams = energy level diagrams
represent enthalpy changes
show relative enthalpy (H) level of reactants and products
horizontal axis= extent of reaction( reaction progress) vertical axis= enthalpy
heat and temperature
★temperature ≠ (not dependent) on number of particles present
+temperature ≈ (is related to ) AVERAGE kinetic energy
L
As particles move ↑ faster
=
(their) average kinetic energy (KE) of particles in a system ↑ inc.
Heat
HEAT -is a measure of → total K.E. of all particles in a given amount of substance
(it DOES depend on→ HOW MUCH SUBSTANCE IS PRESENT)
(INCLUDES ENERGY OF EVERY PARTICLE IN THE SUBSTANCE)
- ( E.G: a luke warm water bath (larger particle number) will have more energy than a red hot nail)
(heat and temperature are not exactly the same as something can have less heat than the other object and still have a higher temperature→ causing flow from the higher temp. region to lower) ← (my interpretation)
L
EXPLAINATION CONNECTING TO THE PREVIUS EXAMPLE:
(Heat always flows from high to low temperature, so heat will flow from the nail into the bath water. even though the water has much more heal than the nail.)
measuring heat
unlike temperature +heat ≠ measured directly
L
therefore ∆ H of a substance is used→ (heat is transferred to a particular mass of a substance)- (often water)
REQUIRES 3 THINGS:
-MASS of substance (being heated or cooled)
-temperature (∆H) change
- specific heat capacity ( c )→ of substance.
specific heat capacity ( c )
★the amount of energy (J) needed to raise the temperature of 1 g of the substance by1 K
( the specific heat capacity of
water is 4. l 8Jg-1 K-1• This means that it takes 4.18 joules to raise the
temperature of I gram of water by I kelvin. This is often rounded up
to 4.2 J g-1 K-1 )
EQUATION:
∆ H= m x c x ∆T
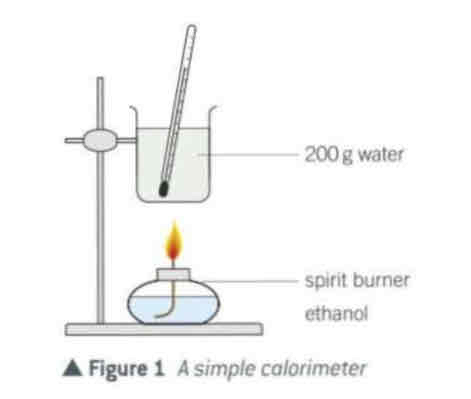
calorimeter (q = m c ∆ T)
can use the apparatus → to find the approximate ∆H when a fuel burns.
You burn the fuel to heat a known mass of water and then measure the temperature rise of the water.
You assume that→ all the heat from
the fuel goes in to the water.
HESS’S LAW
Hess's law states that the enthalpy change for a chemical reaction
is the same, whatever route is taken from reactants to products
mean bond enthalpy
The average value of the
bond dissociation enthalpy for a given type of bond
taken from a range of different compounds.
(aka bond energy term)
(as they are avg. calc. using them = approx. answer
bond energy
MEASURE of the amount of ENERGY NEEDED to BREAK a BOND
DIATOMIC MOLECULES
-Ones that contain only two atoms(e.g; Cl2 )- same or (e.g: HCl)- different
( question picked up points) why do the energy values for the same reaction differ?
Heat loss to the surroundings
incompeletecombustion
heat capacity of the apparatus
STANDARD ENTHALPY CHANGE OF VAPOURISATION (ΔH°v )
-the heat needed to convert one mole of substance from liquid → gas
standard enthalpy change of atomisation ΔH°a
the heat needed to produce 1 mole of gaseous atoms from the element in its standard state.