reaction rates and equilibrium
1/33
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
34 Terms
how do rates of reaction differ?
some happen very fast, while other reactions take ages
what does the rate of a reaction do?
measures how fast a reactant is being used up/ how fast a product is being formed
what is the rate of a reaction?
the change in concentration of a reactant or product in a given time
rate =?
change in concentration/ time
units of rate?
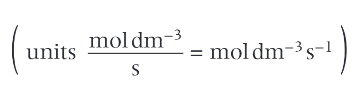
when and why is rate of a reaction fastest?
at the start of the reaction, as each reactant is at its highest concentration
when and why does the rate of a reaction slow down?
as the reaction proceeds, as the reactants are being used up and their concentrations decrease
when is the rate of a reaction zero?
once one of the reactants have been completely used up so the concentrations stop changing
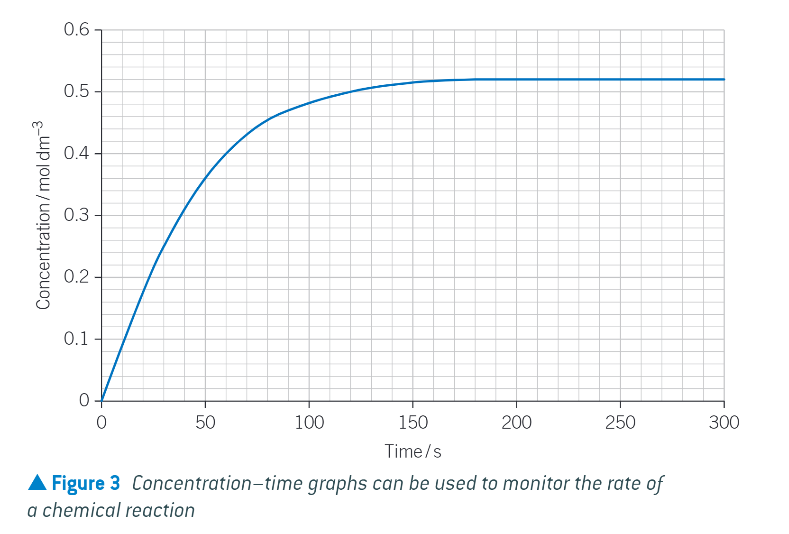
concentration time graph example:
shows the formation of a product over the course of a chemical reaction
what are the factors that can change the rate of a chemical reaction?
concentration (or pressure when reactants are gases)
temperature
use of a catalyst
surface area of solid reactants
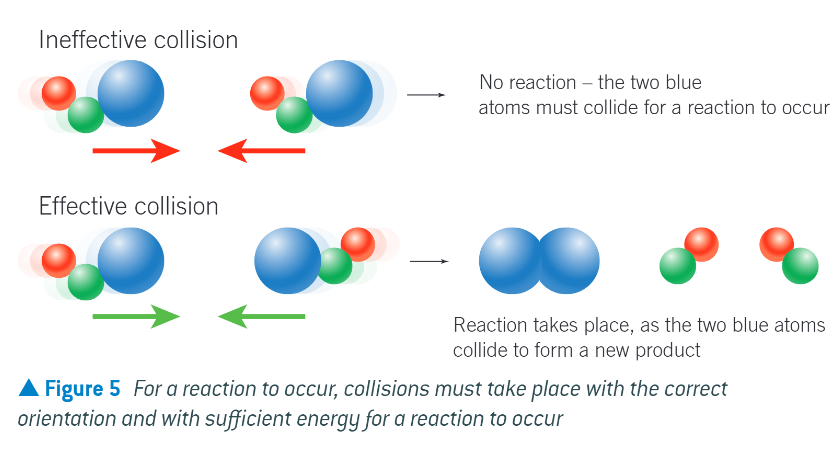
what is collision theory?
states that 2 reacting particles must collide for a reaction to occur. usually only a small number of collisions start a chemical reaction
what happens in most collisions?
the molecules collide but then bounce off each other and remain chemically unchanged
what are the 2 conditions that need to be met for a collision to be effective?
the particles collide with the correct orientation
the particles have sufficient energy to overcome the activation energy barrier of the reaction
how does increasing the concentration affect the rate of reaction?
an increase in conc increases the number of particles in the same volume
the particles are then closer together and collide more frequently
there will therefore be more effective collisions in a given period of time and an increased rate
how does increasing the pressure of a gas affect the rate of reaction?
the conc of the gas molecules increases as the same number of gas molecules occupy a smaller volume
the gas molecules are closer together and collide more frequently, leading to more effective collisions in the same time
how can the progress of a chemical reaction be followed?
monitoring the removal (decrease in conc) of a reactant
following the formation (increase in conc) of a product
what will the method chosen depend on?
the properties and physical states of the reactants and products in the reaction
what are other measurable properties that can be used to measure rate?
gas volume, mass of reactants/ products, colour
how can you determine the rate of a reaction if the reaction produces a gas?
monitor the volume of gas produced at regular time intervals using gas collection
monitor the loss of mass of reactants using a balance
what are volume of gas produced and mass loss both proportional to?
the change in concentration of a reactant/ a product. this means that the change in volume with time/ mass loss with time both give a measure of the rate of a reaction
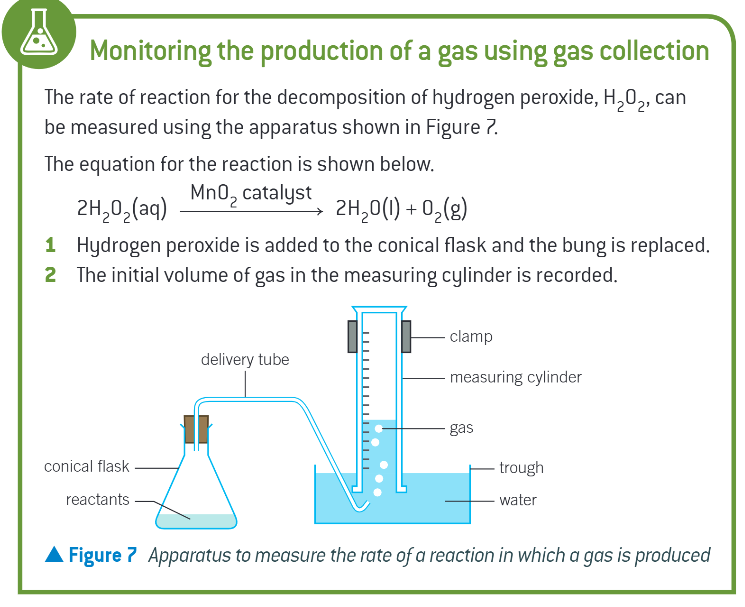
monitoring the production of gas
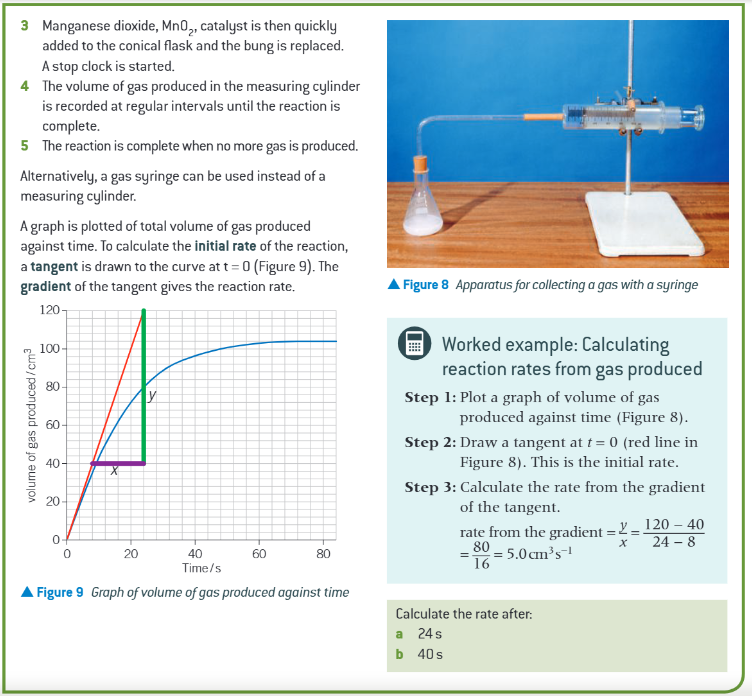
eg. how can the rate of reaction between calcium carbonate and hydrochloric acid also be determined?
by monitoring the loss in mass of the reactants over time
equation for the reaction:
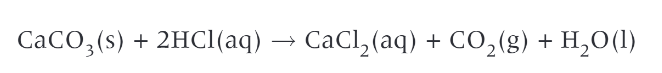
how does this work?
add carbonate and acid to conical flask on a balance
mass of flask and content recorded initially and at regular time intervals
the reaction is complete when no more gas is produced so no more mass is then lost
you can then plot a graph of mass lost against time
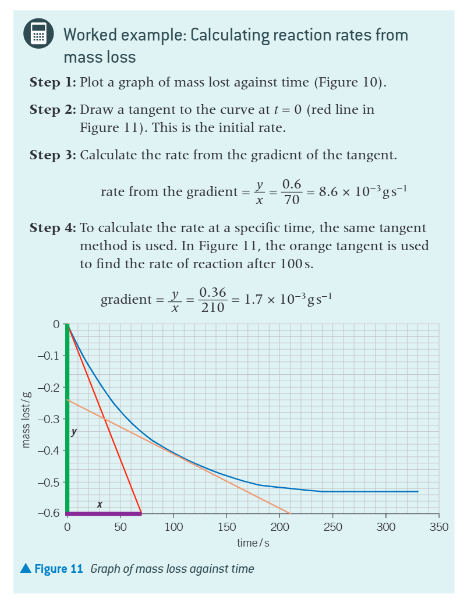
worked example:
what is a catalyst?
a substance that changes the rate of a chemical reaction without undergoing an permanent change itself
features of catalysts:
the catalyst isnt used up in the reaction
the catalyst may react with a reactant to form an intermediate or may provide a surface on which the reaction can take place
at the end of the reaction the catalyst is regenerated