L18- Preclinical models
1/48
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
49 Terms
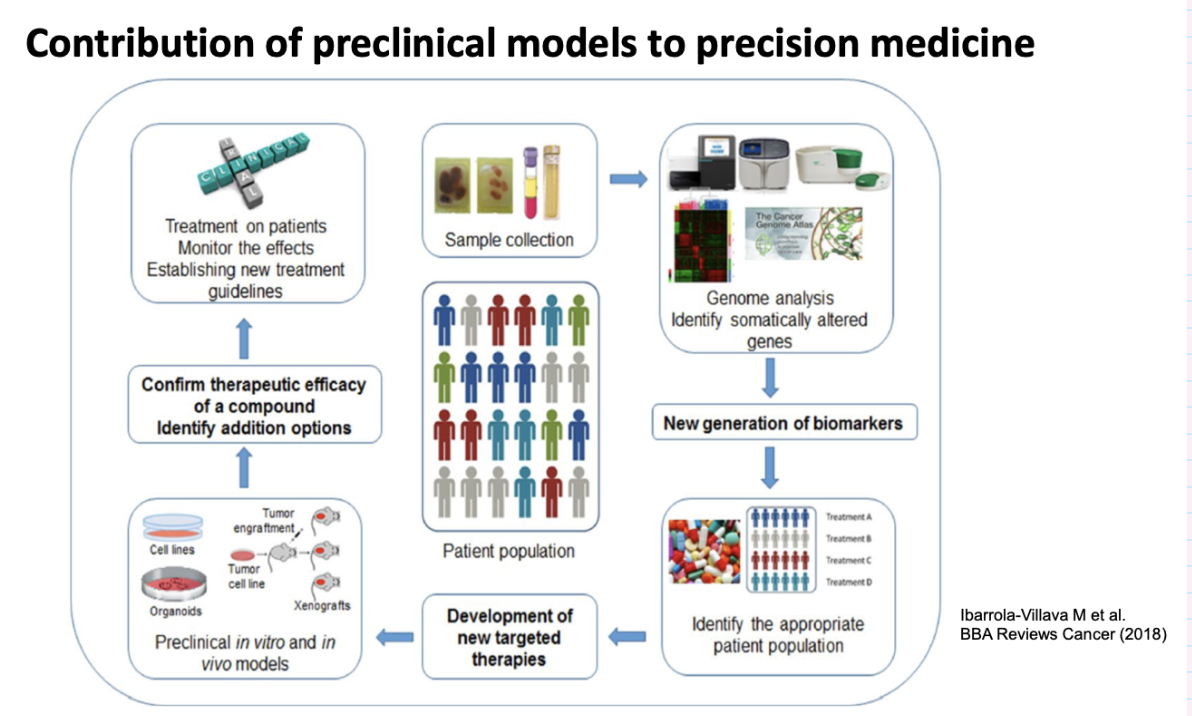
overview of contribution of preclinical models to precision medicine
what material can be studied for in vitro analysis of cancer cells
Cell lines
Spontaneous
Immortalised (oncogene)
Tumour-derived material
Primary cultures
Tissue slices
Transformed cells
Co-cultures
Stromal and epithelial cell populations
3D-cultures
Single or mixed populations embedded in matrix to mimic stromal microenvironment (more representative of tumour microenvironment) – spheroids, organoids
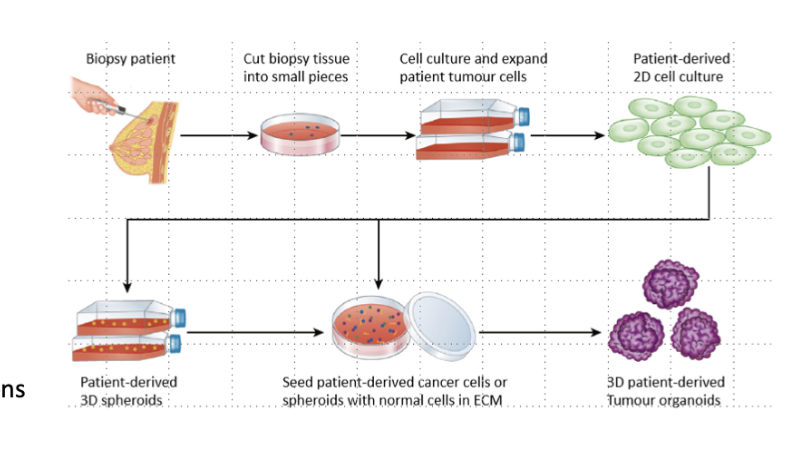
what are primary cultures
Tumour-derived material
Take samples from biopsy and try to grow in lab
- tend to die out after few passages
what is a tissue slice
tumour derived material
biopsy from patient and cut into 200 micron think slices, put in culture and grown- die out after about a week
what are transformed cells
tumor derived material
takes a lot of work and time, need to be validated and characterised which delays models
what is co culture
stomal and epithelial populations mixed to see what effect stroma has
where are 3D cultures derived from
Derived from stem cells- forms 3D structure containing cancer cells and other cell types from TME, hard to produce, complex
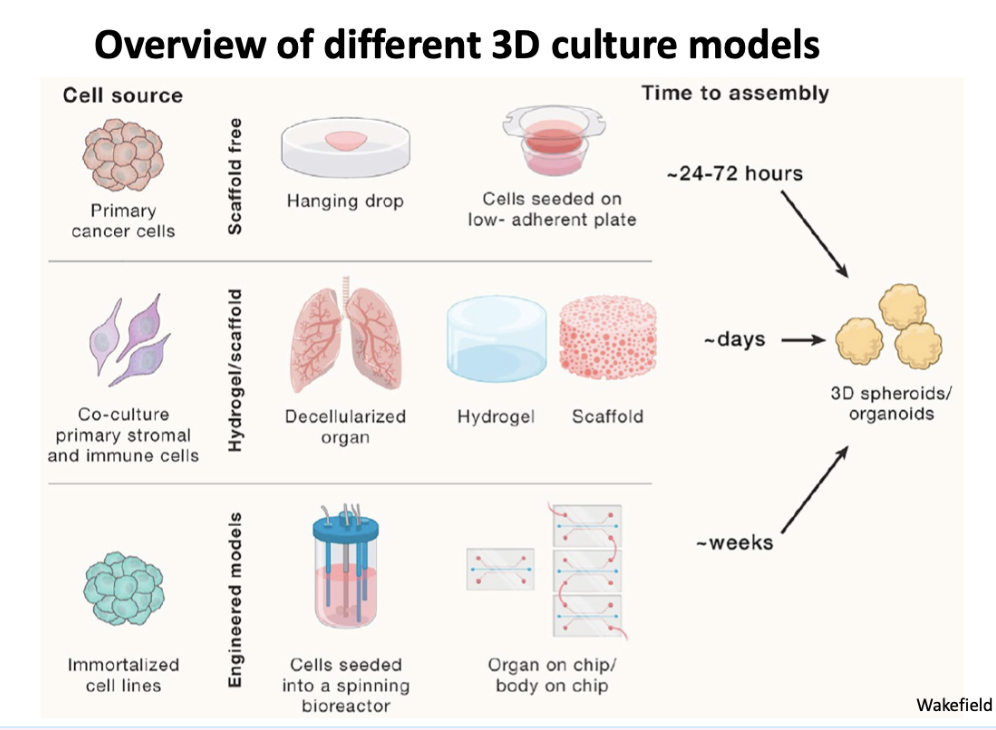
overview of different \3\d models
Primary grown as a spheroid , scaffold free
Mix with other cell types
Is a scaffold
Decellularized organ
Grown in liquid culture, can change pH etc
Cancer cells can be hard to grow as haven't got types of growth media right
how did we go from in vitro platforms to ex vivo models
what are 2 types of proliferation assay
direct cell count
indirect cell count (growth)
what is direct cell count- proliferation assay
Haemocytometer (graticule on glass slide)
Coulter counter (cell numbers passing through light source)
Flow cytometer –including cell cycle analysis (cell morphometric properties detected as pass through light source)
what is indirect cell count (growth)- proliferation assay
MTT (mitochondrial turnover)
Sulforhodamine B (SRB) (cell protein content)
Tritiated thymidine (DNA replication)
Scanning microscope (IncuCyte) (cell confluency)
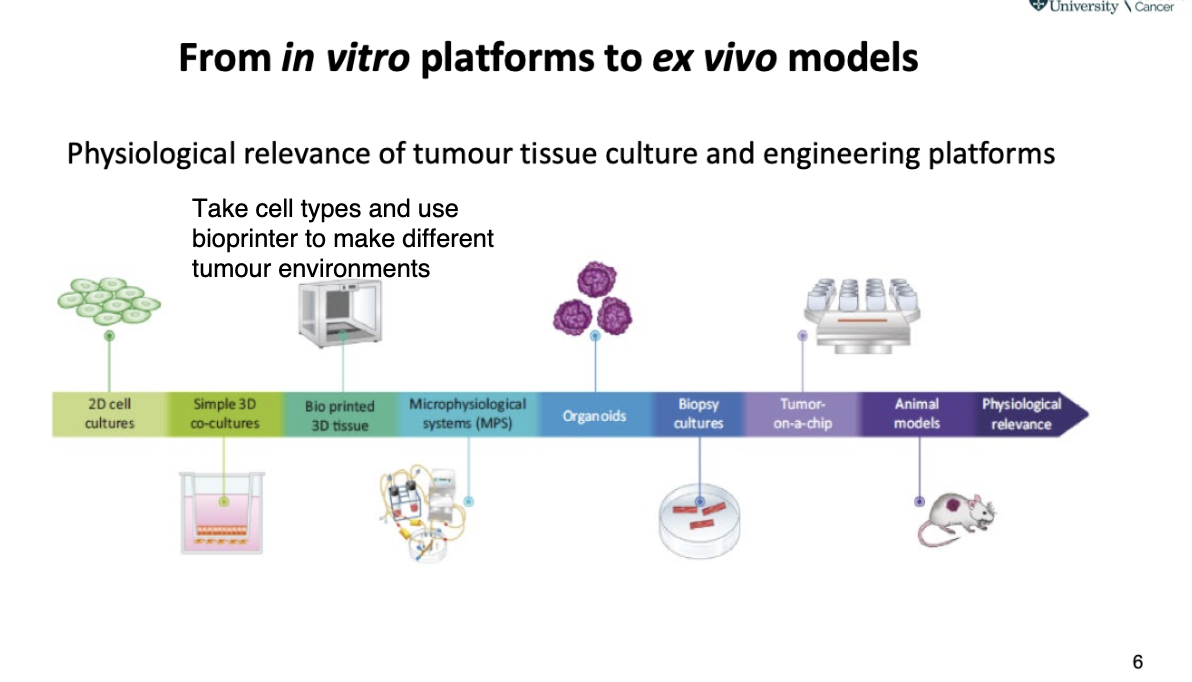
what is an adhesion assay
example- measure integrin-mediated cell-cell interactions
coat plates with ECM components
add radiolabelled or fluorescently labelles cells
allow to adhere, wash wells
measure radioactivity/fluorescence
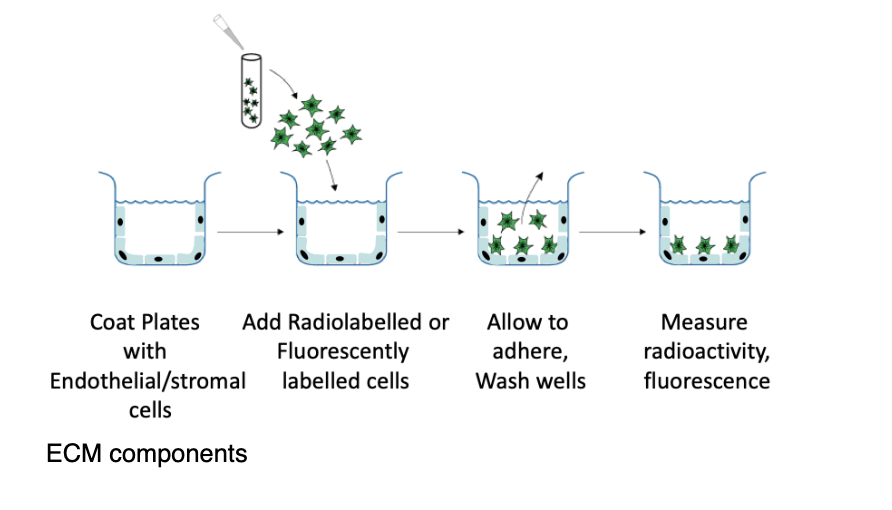
what is cell motility
Essential for multiple cellular processes - requires spatial and temporal regulation of contractile and protrusive forces induced in cell body and adhesion strength
Actin cytoskeletal network (dynamic and versatile intracellular scaffold) – critical for driving projection of cell membrane and orchestrating directed movement
what 3 actions allow for cell motility
Protrusion of cell membrane
– Formation of leading edge
Initiation of new cell-substratum attachment s ites
– Adhesion molecules stabilise structure at leading edge
– Structural and signalling molecule complex form on cytoplasmic side – focal contacts
Contractile forces
– Derived from activity of actin-myosin motors
– Forward movement of cell
Disassembly of adhesions at rear of cell
– Cell retraction
what is the scratch wound migration assay
a motility assay
Cancer cells growing in culture on well
Allow to cover whole dish
Scratch a wound into cell layer- gap between cells
Use microscopes, measure how cells will migrate to fill in the gap
Over time, gap fills as cells migrate
Put conditions/drugs into environment to see how can modulate cells to be more or less motile

what is quantitative fluorescent speckle microsopy
a more sophisticated motility assay
Monitor how actin forms macromolecular structure
Can put drugs in to modulate and see effects
Fluorescent labelled protein - Rhodamine-G-actin (monomer) incorporated into macromolecular structures (F-actin)
•Visualised by real-time imaging technique
•Fluorescent speckles act as markers of actin dynamics
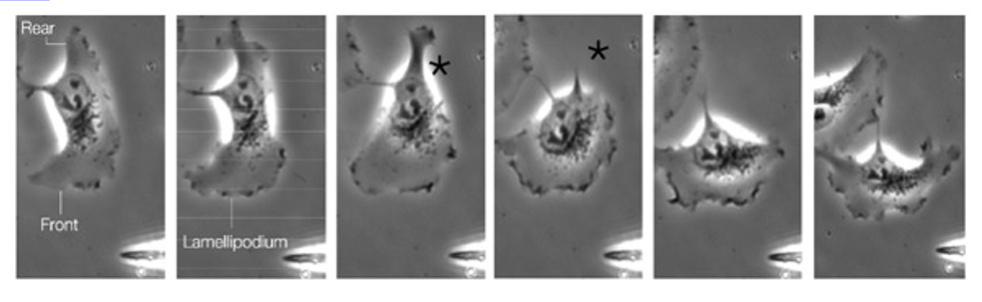
describe the invASION ASSAY- BOYDEN CHAMBER
Boyden chamber- well of tissue culture disc, insert a chamber that fits into well, bottom part has small holes (smaller than a cell), cells squeeze themselves through to enter well below
Chemoattractant in bottom of chamber attracts cancer cells
Measure motility to some extent
Can put collagen matrix at bottom of chamber- a barrier for cancer cells to cross, in order for them to cross they need to digest using proteases
Take bottom part if chamber and stain it- darker areas in image are cancer cells, smaller than pores, but can squeeze through, can change shape and size to be motile and invasive
what is an issue with using cancer cell lines
Issue with cancer cell lines- come from patients with advanced cancer with lots of mutations
Useful for looking at late stage of cancer
Not many models for early stages
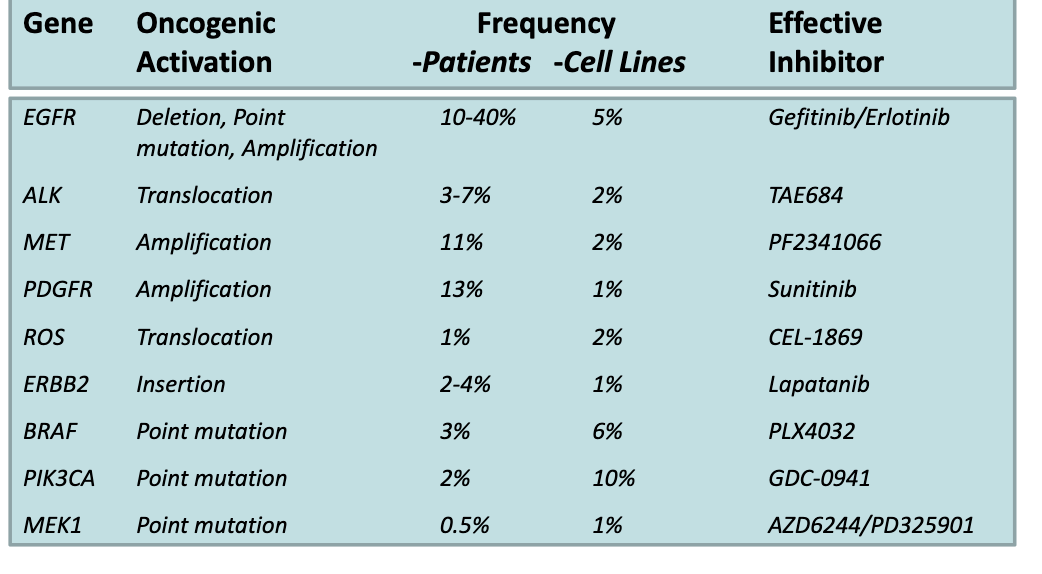
describe the applications of cell lines to stratify cancer types based on activating mutations
NSCLC 139 cell line panel as models for therapeutic intervention – mutational profile defined by DNA sequencing
• Characteristic cell line mutations reflect mutations observed in NSCLC patient sub-groups
• Investigate how patients with specific mutation will respond to different therapeutic agents
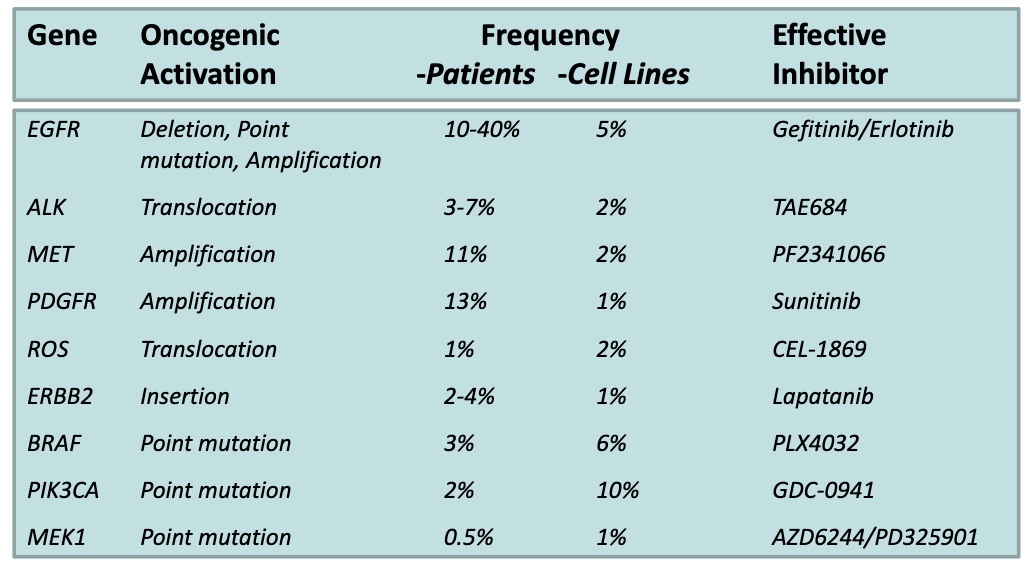
How are cell lines used in non-small cell lung cancer research?
Cell lines have been DNA sequenced, so all mutations are known
Large bank of ~139 cell lines representing different patient-like phenotypes
Used to study drug effects by:
Applying a drug to a specific cell line
Observing the resulting cellular response
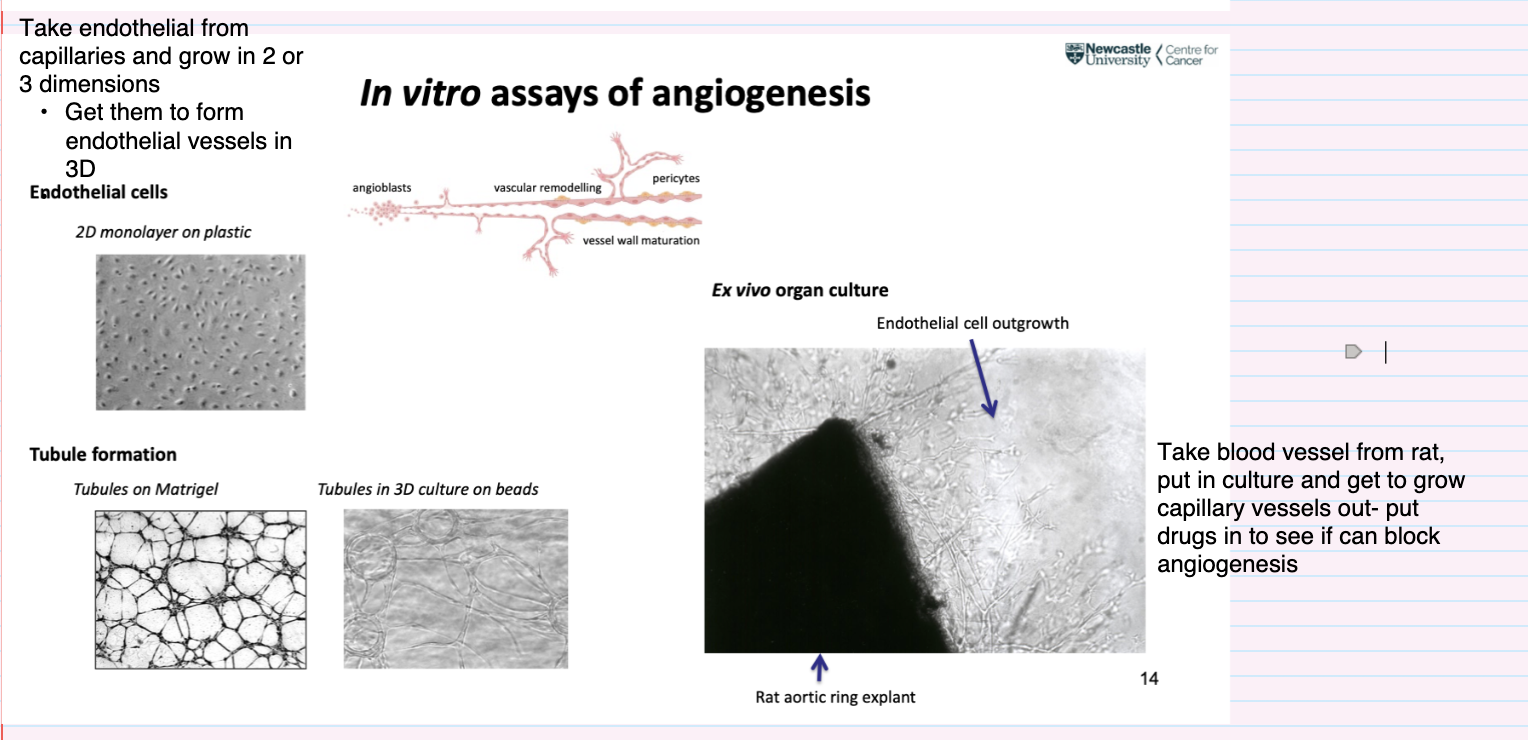
How are in vitro assays used to study angiogenesis?
Use endothelial cells from capillaries
Grow in:
2D (monolayer on plastic)
3D → form endothelial vessel-like structures
Tubule formation assays:
On Matrigel → capillary-like networks
In 3D culture on beads → vessel structures
Models angiogenesis stages:
Angioblasts → vascular remodelling → vessel wall maturation (with pericyte
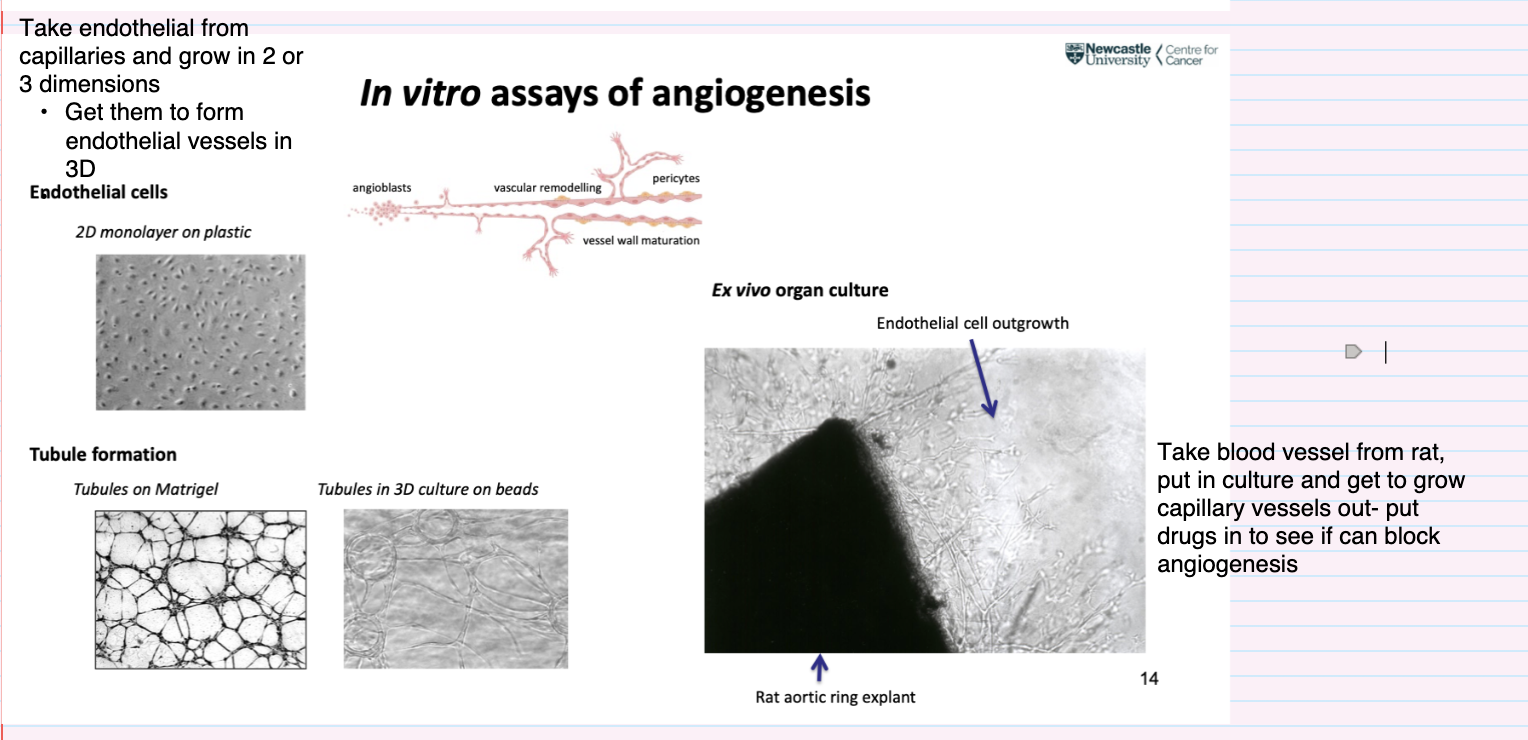
How is the ex vivo angiogenesis assay performed and what is it used for?
Rat aortic ring explant assay:
Take blood vessel (rat aorta) and culture it
Observe endothelial cell outgrowth and capillary sprouting
Used to:
Study angiogenesis in a more tissue-like context
Test drugs by adding them to see if they block angiogenesis
How is the chick chorioallantoic membrane (CAM) assay used to study angiogenesis in vivo?
Uses 7-day-old chick embryos (lack a mature immune system)
Remove eggshell to access the CAM (highly angiogenic membrane)
Place test substances, tumour cells, or drug pellets onto the membrane
Observe capillary/angiogenic response after several days
Readout: visible changes in blood vessel growth
Useful for drug screening to assess pro- or anti-angiogenic effects
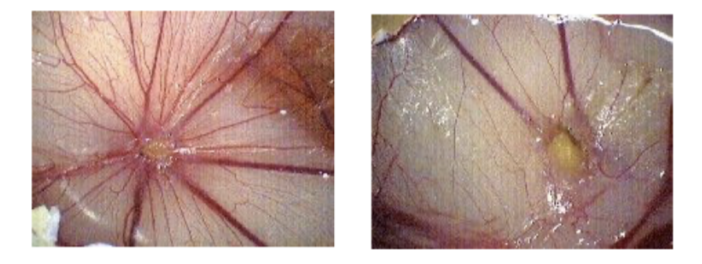
How are chamber assays used to study angiogenesis in vivo?
Transparent chamber in rabbit ear, dorsal skinfold (mice), cranial window (mice)
Allows continuous measurement of angiogenesis, blood flow, pH, tumour growth and wound healing
Invasive and technically difficult
Lose skin in back of mouse, attach chamber that holds skin and can visualise under microscope- look at blood flow in response to being treated with drug- look at vessels and what's happening inside them
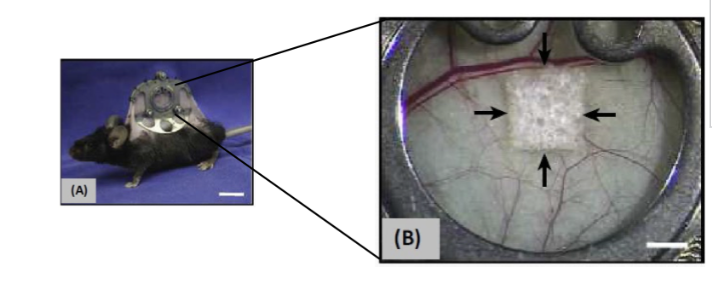
How are tumour models used in in vivo assays of angiogenesis?
Tumours grown in mice subcutaneously or orthotopically
Implant tumour cells in skin of mouse (skin at back is very loose)
Can see tumour grow under skin and follow its size
Effect of test substance on tumour size and animal survival established at regular intervals
Tumour can be taken then looked at under microscope
Histological analysis determines effects of drug on necrosis, thrombosis and microvessel density
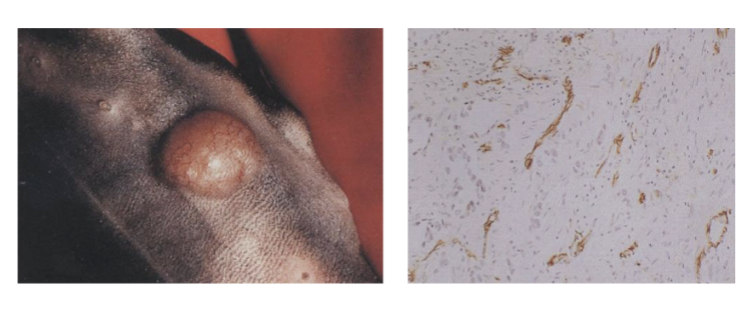
what different things can be done using murine models in cancer studies
Injection of cell lines/ human tumour tissue
– Subcutaneous, tail vein, cardiac, orthotopic,
metastatic site (bone)
• Over-expression of transgene
– Powerful promoters eg. CMV, SV40
• Depletion of gene
– Knockout
– short inhibitory RNAs
• Site-specific expression
– Gene expression driven by tissue specific promoter
• Regulated expression
– Temporal gene induction
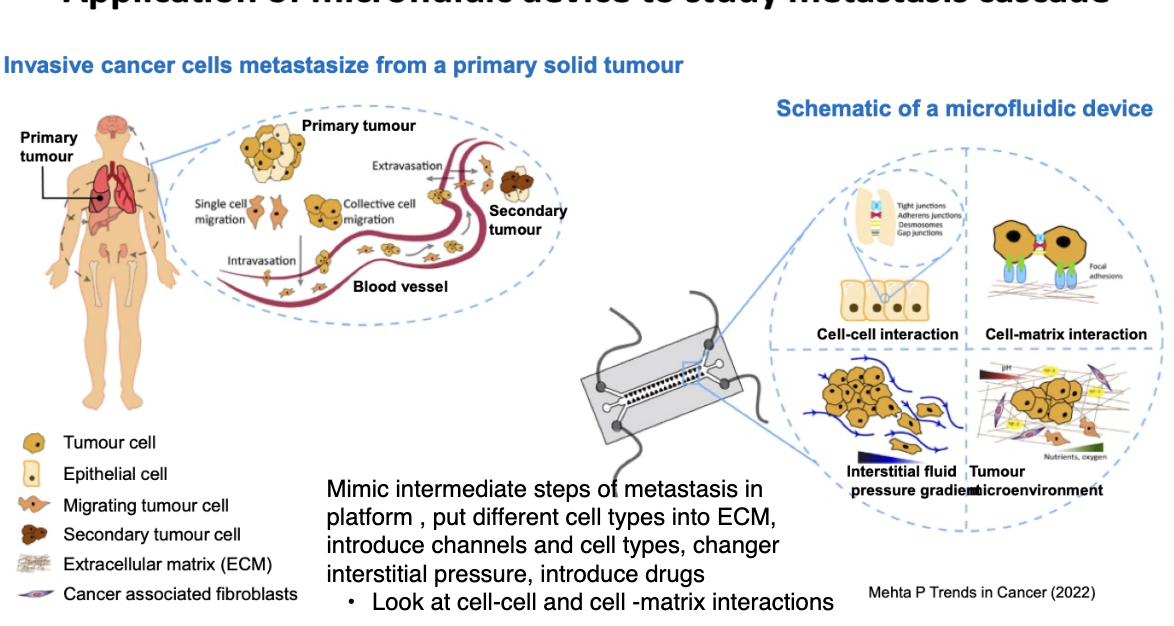
describe the application of microfluidic device to study the metastasis cascade
Mimic intermediate steps of metastasis in platform , put different cell types into ECM, introduce channels and cell types, changer interstitial pressure, introduce drugs
Look at cell-cell and cell -matrix interactions
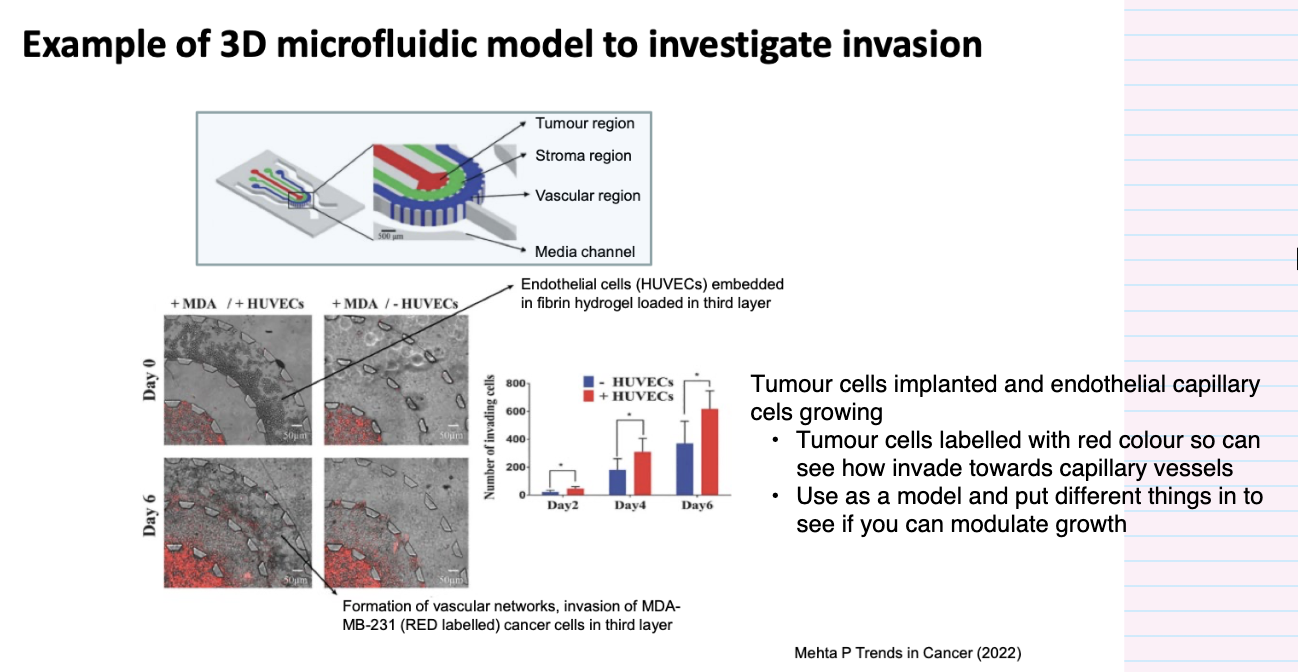
give an example of using a 3D microfluidic model to investigate invasion
Tumour cells implanted and endothelial capillary cels growing
Tumour cells labelled with red colour so can see how invade towards capillary vessels
Use as a model and put different things in to see if you can modulate growth
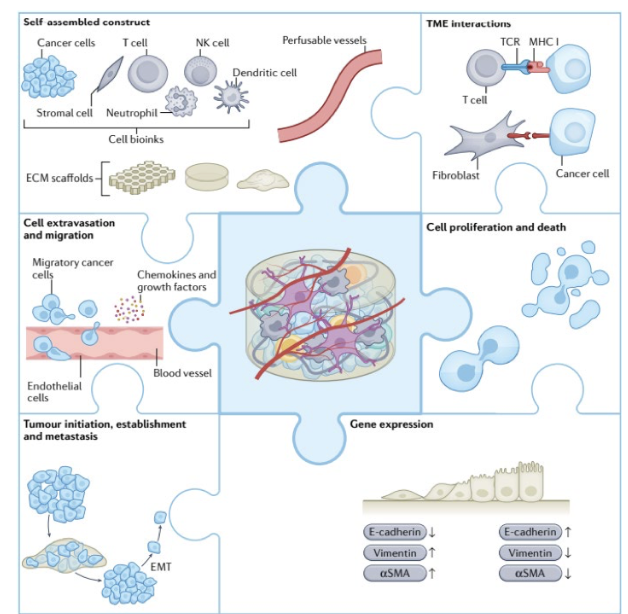
how can 3D bioprinted models be used for applications in cancer biology
3D printer- reconstruct TME introducing cell types and cancer cells, cells that form capillaries, use it to study interaction with immune cells etc
Much more complex
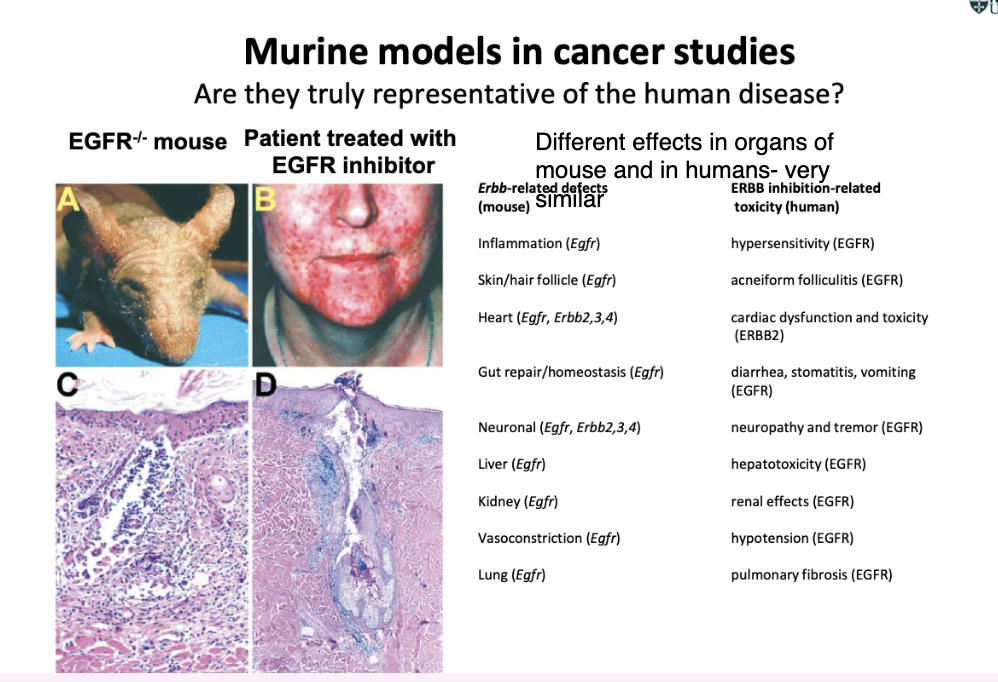
what are the limitations of murine models in cancer studioes
Might not be representative of human- e.g. drug works in mouse but not when put into human
HOWEVER
EGFR knocked out in mouse- skin inflammation and hair follicle damage
Human patient treated with small molecule inhibitor (mimics the knockout )- many features seen in the human are the same as in the mouse- very similar pathology as in mouse
What is the biological role of c-Myc and how does it affect cell behaviour?
c-Myc expression correlates with high proliferative activity
c-Myc is a transcription factor rapidly induced by mitogenic stimuli
Drives G1–S progression
Inhibits terminal differentiation and sensitizes cells to apoptosis
In vitro manipulation:
Overexpression → increases proliferation and promotes apoptosis
Knockdown → reduces proliferation
Around 50% of cancers have affected Myc (usually upregulated)
Helps drive cancer cells
How is c-Myc deregulated in cancers and studied in murine models?
Elevated/deregulated c-Myc in ~50% of cancers
Breast, colon, cervical, small-cell lung carcinoma, osteosarcomas, glioblastomas, melanomas, myeloid leukaemias
Associated with aggressive, poorly differentiated tumours
Mouse models:
Knockout Myc → lethal (embryos don’t survive) → shows importance of Myc
Transgene overexpression of Myc → induces many cancer types
What happens when c-Myc is disrupted or constitutively expressed in in vivo murine models?
Targeted gene disruption of both c-Myc alleles → embryonic lethal (day 9.5–10.5)
Transgenic mice with c-Myc constitutively expressed develop variety of phenotypes:
Lymphomas, skin ulcerations, multiple tumours
What are the limitations of constitutive c-Myc expression models and how can they be overcome?
c-Myc constitutively expressed in single cell type via tissue-specific promoter
Problem: expressed during embryonic development (long latency)
Issue: tumour in humans doesn’t arise in embryo but later stage
Overcome:
Express Myc using specific promoter
Only expressed when activating agent is added
Turn on in one cell type and specific organ
What are conditional transgenic c-Myc models and why are they useful?
Conditional transgenic:
c-Myc can be switched on in specific adult tissues
Activating agent stabilises and activates MYC signalling
Models are useful to:
Study tumour development in adult context
See how different organ backgrounds react
How is conditional transgenic c-Myc expression used in different tissue environments?
Conditional transgenic (c-Myc) expression:
Subrabasal epidermis (involucrin promoter)
Pancreatic islet β-cells (insulin promoter)
Constitutive Myc expression (inactive), tamoxifen acts as activating ligand
One model: gene only in skin basal cells → skin tumours
Another model: under insulin promoter → pancreatic tumours
Need to ensure model reflects what you're trying to study
What are the tumour characteristics of c-Myc activation in different tissues?
In subrabasal epidermis:
c-Myc activation induces proliferation and impaired differentiation
No apoptosis
In pancreatic islet β-cells:
Rapid proliferation (1 day)
Apoptosis of β-cells and onset of diabetes in 9 days
Difference:
Skin tumours → no apoptosis
Pancreatic tumours → high apoptosis levels
why is imaging in cancer important
Accurate positional information of tumour/ metastases are required for modern clinical treatment
what are the important things to learn from imaging in cancer
Tumour location
– Many biopsies fail to recover cancer cells
• Tumour size
– Conventional imaging techniques detect tumours > few mm (>109 cells)
• Tumour confined/spread locally to lymph nodes
– Stage 1 cancers frequently >90% 5 year survival
– Early stage tumours often curative
• Involvement of critical anatomical structures
– Vital organ/ blood supply may alter treatment strategy
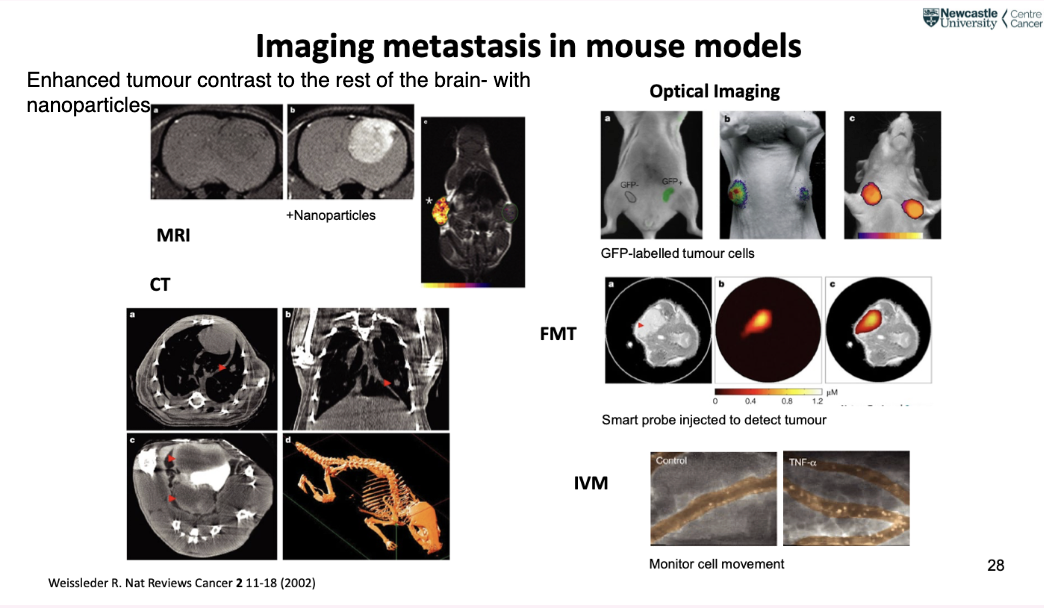
describe the features of metastases imaging techniques in mouse models

what do results of different imaging techniques look like
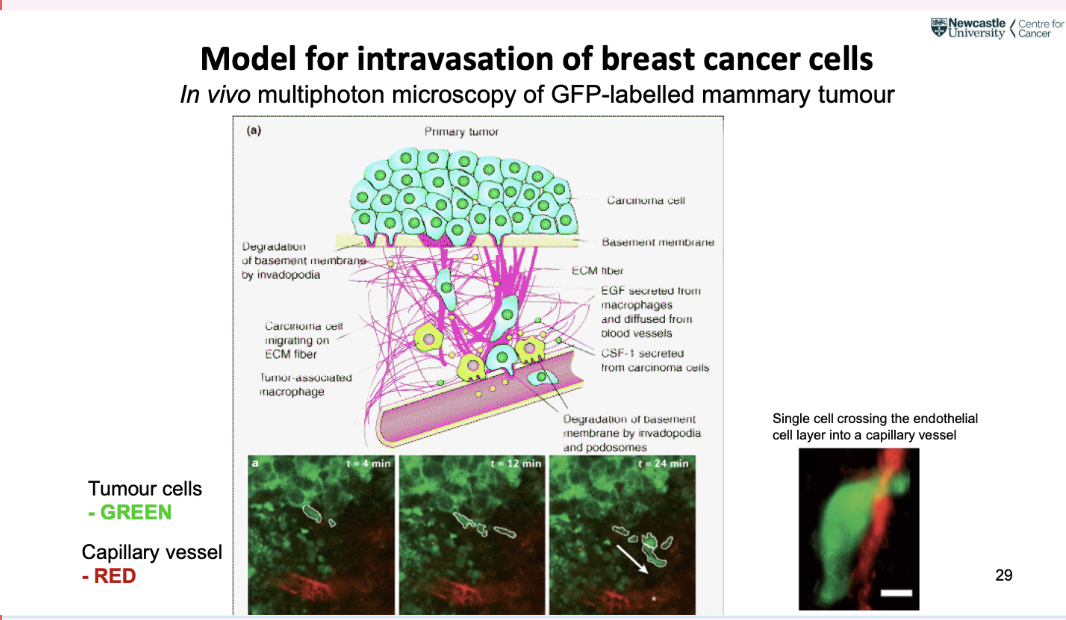
what is a common model for intravasation of breast cancer cells
in vivo multiphoton microscopy of GFP labelled mammary tumour
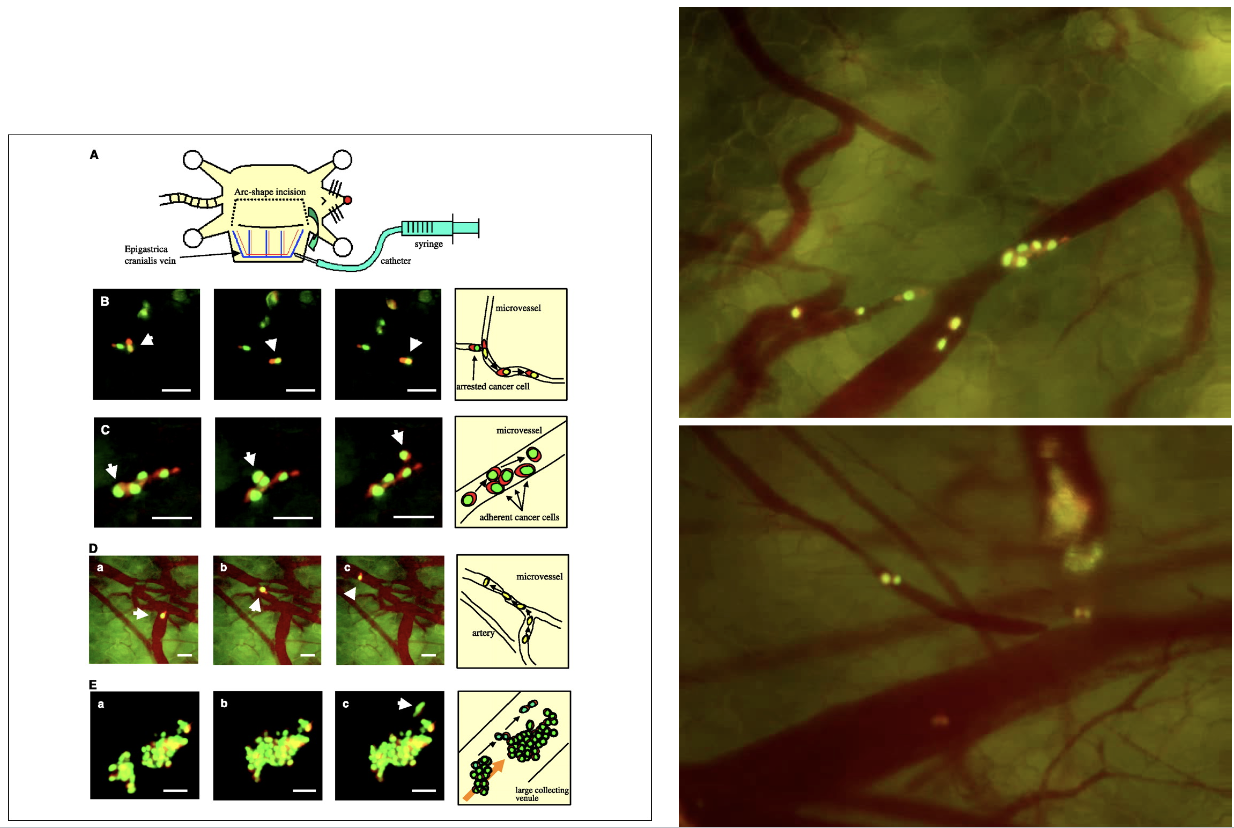
how can intravascular trafficking be imaged
Cancer cells moving through bloodstream
HT1080 cancer cells – Nuclear green, Cytoplasmic red
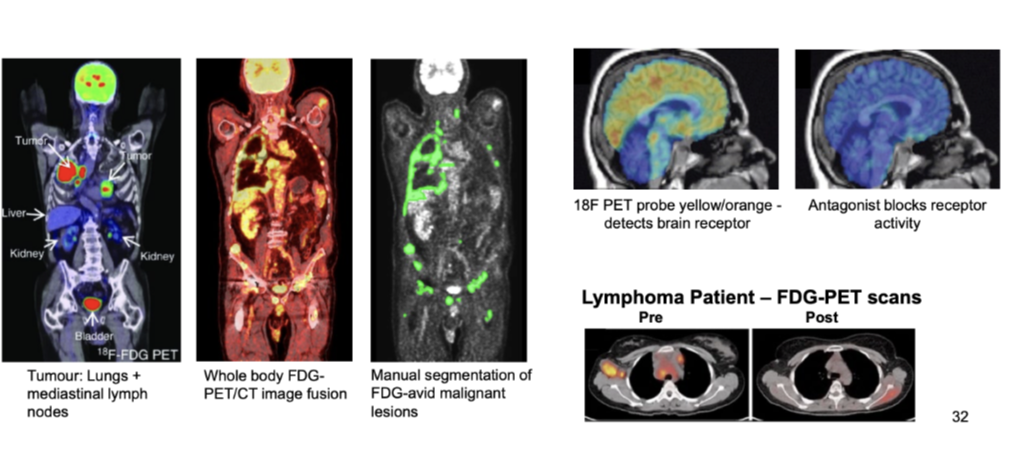
what is positron emission tomography (PET)
• Widely used in clinical staging and restaging for many malignancies
• [18F]fluorodeoxyglucose (FDG) – glucose analogue used in >90% of cancer scans
• FDG selectively taken up by cells with high rate of glucose metabolism (tumours)
• FDG-PET used for staging of many cancers (eg breast, colorectal, oesophageal, head
and neck, melanoma, lymphoma and non-small cell lung cancers)
How is FDG-PET imaging used to predict tumour response to therapy and survival?
Sequential FDG-PET imaging in advanced cancer is often a more accurate predictor of treatment response than clinical or histopathological criteria
Detects metabolically active tumour sites
Combining PET and CT:
Visualises active tumour regions
Shows where cancer has metastasised
Applications:
Whole body FDG-PET/CT image fusion
Identification and manual segmentation of FDG-avid malignant lesions
Example: tumours in lungs and mediastinal lymph nodes
Can assess treatment response (e.g. lymphoma pre vs post scans)
Other tracers:
18F PET probe detects brain receptor activity (yellow/orange)
Antagonist blocks receptor activity
What are the limitations of current imaging probes and the advantages of ‘smart’ probes in tumour imaging?
Current imaging probes:
Non-specific (small molecules, leak into extracellular space → high background)
Targeted (monoclonal antibodies – unbound antibodies give high background)
Smart probes:
Optically silent in native (quenched) state when injected
Highly fluorescent following enzyme-mediated release of fluorochrome at target site
Signal amplification – several 100 fold
Highly specific, enzyme cleavage specific peptide sequences
Single enzyme can cleave multiple fluorochromes
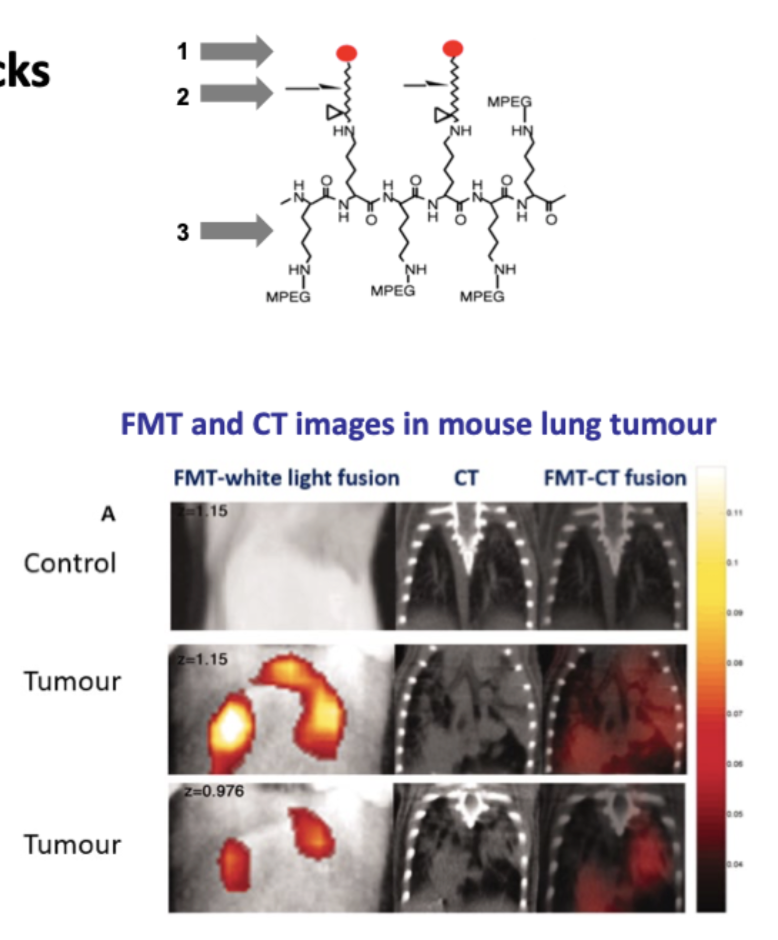
What are the components of smart probes in tumour imaging?
Smart probes comprise 3 biocompatible building blocks:
Near infrared fluorochrome
Enzyme-specific peptide substrate link
Delivery vehicle – dextran backbone
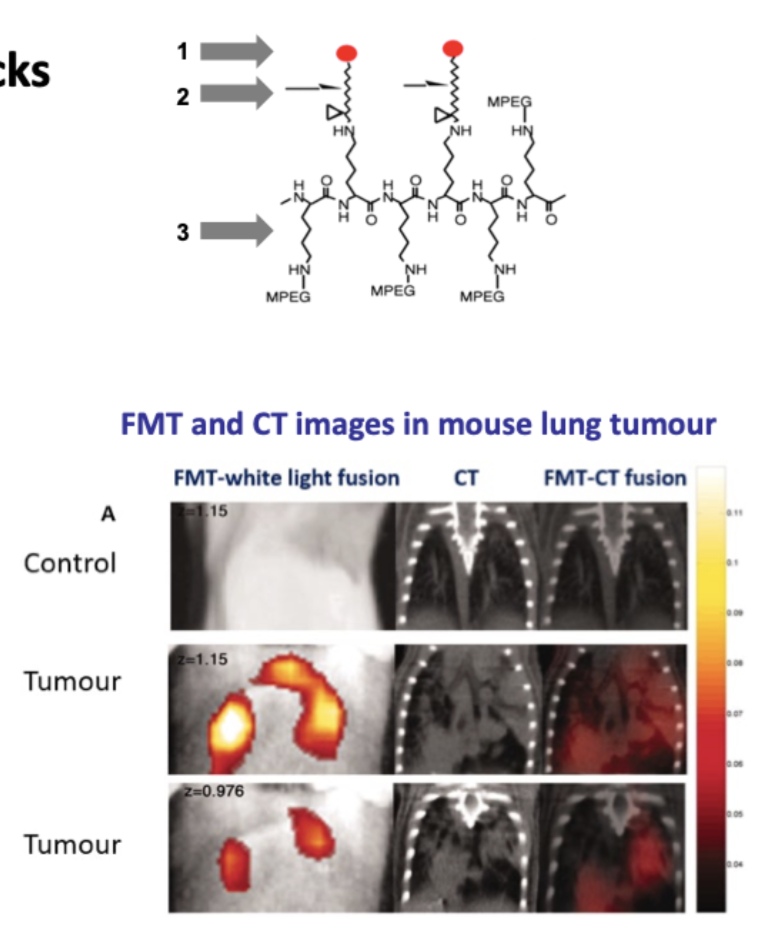
Why are cancer-associated proteases used as targets for smart probes?
Important in invasion, metastases, angiogenesis
Frequently overexpressed at tumour site
Cathepsins, MMPs, Caspases, PSA, Thrombin
Smart probe activated → emits strong signal detected by imaging