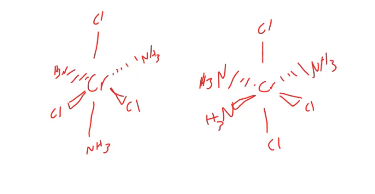isomers
1/15
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
16 Terms
coordination-sphere isomers
differ due to an interchange of ligands among coordination spheres

what kind of isomerization is this, this typically occurs in?
coordination isomerization, water
structural isomers:
linkage isomers
differ in the atom of a ligand bonded to the metal in the complex
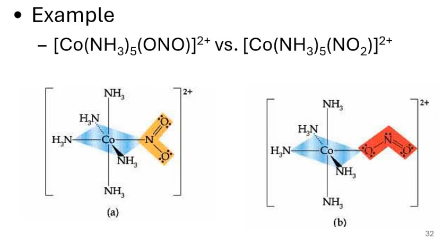
what kind of isomers are these?
linkage isomers, ambidante ligands
what are ambidante ligands?
those that can bond to the metal center at various different atom positions in the molecule. they have different ways to ling to the metl center
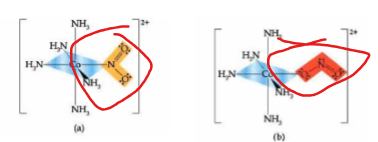

ionization isomers swap _____ for _____
counterions for ligands
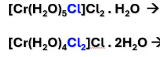
what type of isomers are these
ionization isomers
Geometric isomers and optical isomers are _____
stereoisomers




stereoisomers have different ?
colors, melting points, polarities, spectroscopies, solubilities, reactivities
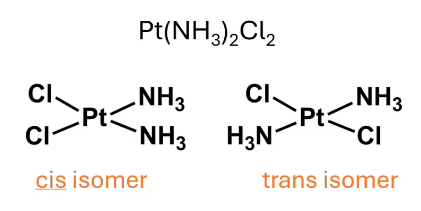
these are ___ isomers, what is the IUPAC name
geometric isomers. cis and trans diamminedichloroplatinum(II)
enantiomers are also called ?
optical isomers
sketch an example of enantiomer isomers for a tetrahedral complex
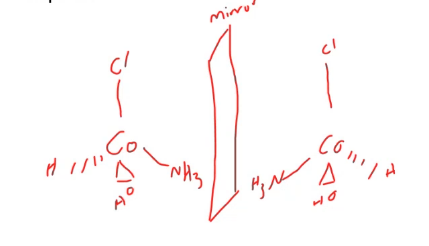
enantiomers are not specific to ___
geometry
sketch the 2 geometric isomers for Cr(NH3)3Cl3