3-3/4/5 (Amino Acids and Peptides)
1/71
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
72 Terms
Glycine.
Its side chain is hydrogen, so the alpha carbon has two identical groups.
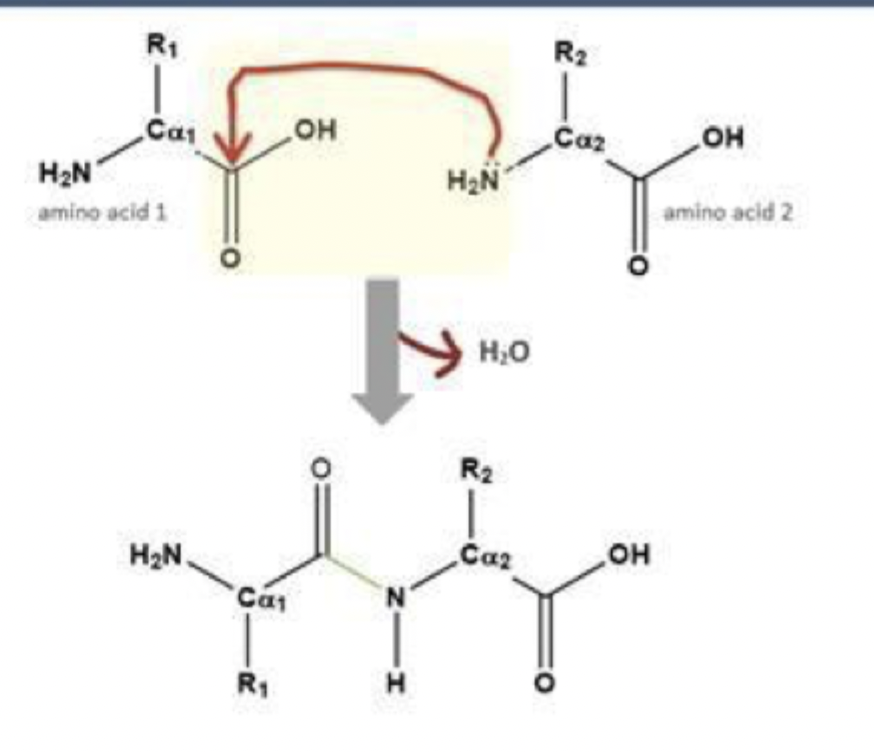
Through a condensation (dehydration) reaction.
removes H2O per reaction
forms between the carboxyl group of one amino acid and the amino group of another.
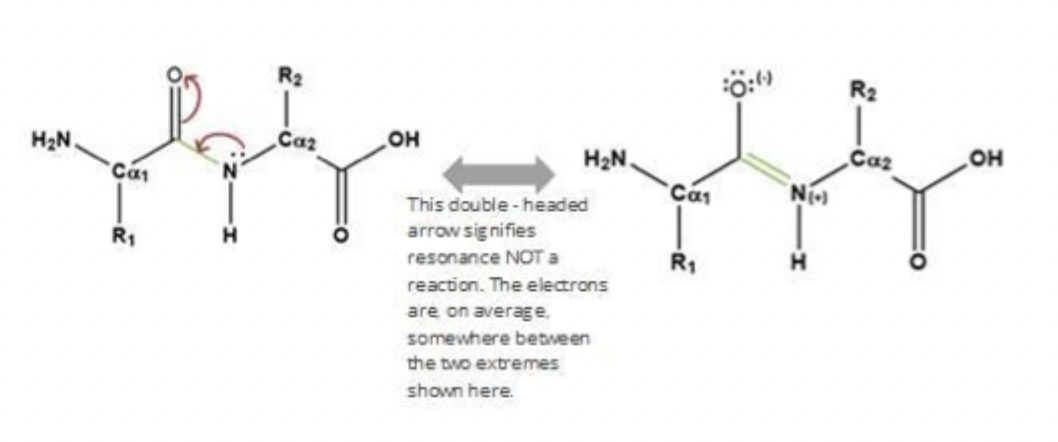
It has partial double-bond character.
so its bc os the Resonance delocalization of electrons.
it’s Rigid and planar with restricted rotation.
Different chemical groups at each end.
N-terminus: Start of polypeptide with a free amino group
C-terminus: End of polypeptide with a free carboxyl group.
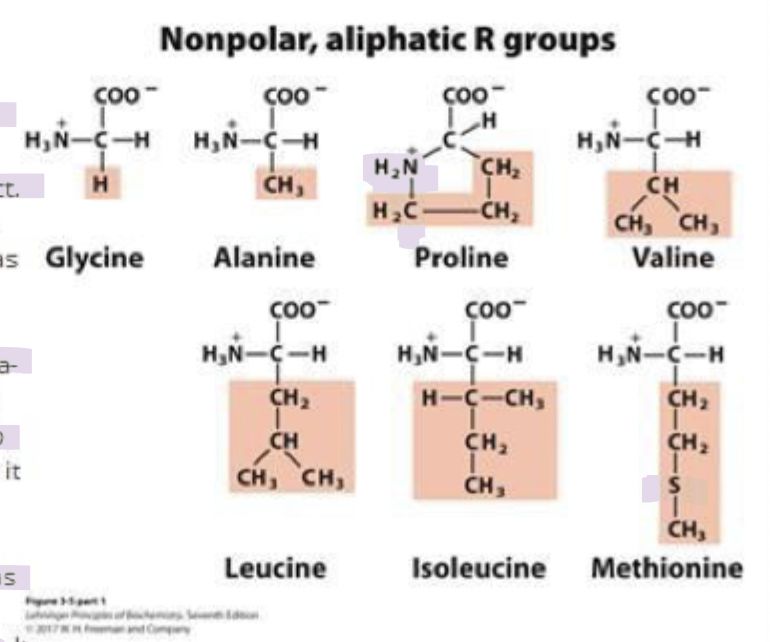
what are the non-polar aliphatic amino acids (7)
NP and hydrophobic
glycine (gly) G
alanine (ala) A
proline (pro) P
valine (val) V
leucine (leu) L
isoleucine (ile) I
methionine (met) M
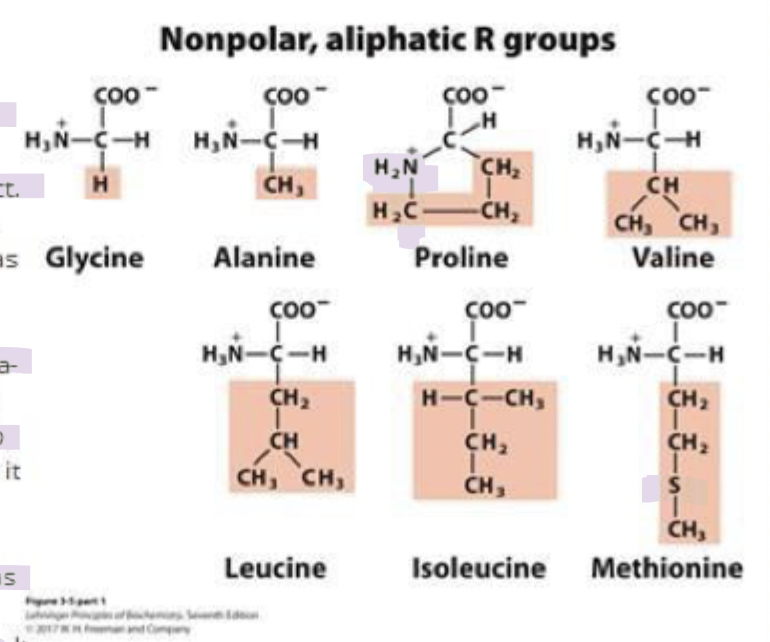
ala, val, leu, ile
cluster together within proteins
stabilizes them through hydrophobic effect
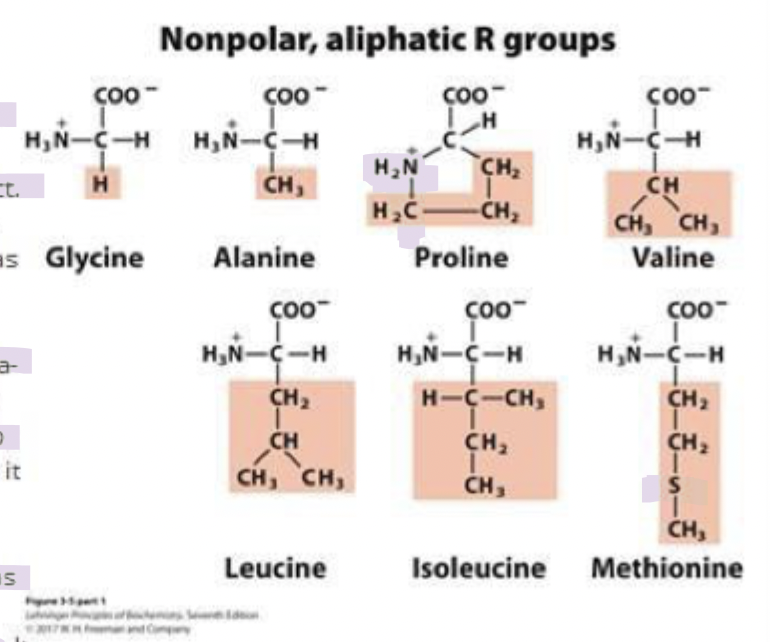
gly and ala
ambivalent (mixed)
they can reside outside or inside
since they have smaller side chains, their hydrophobic effects aren’t so extreme
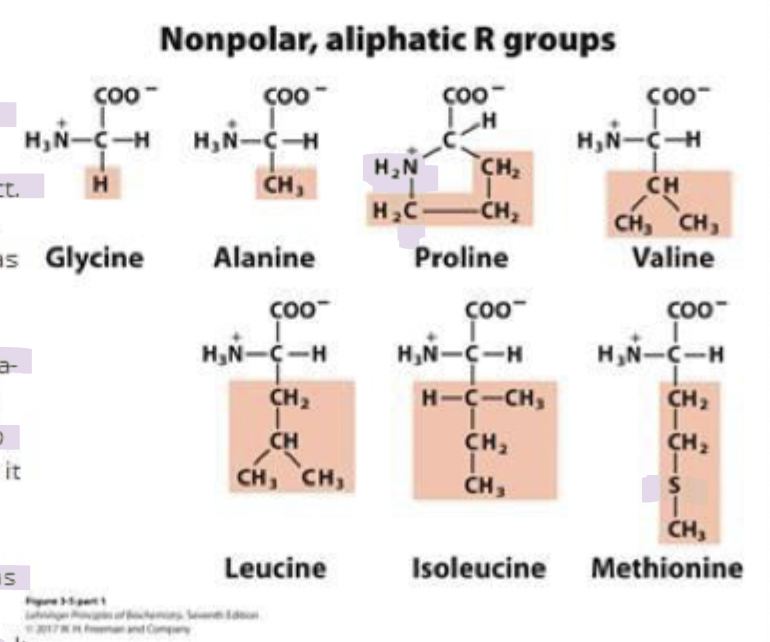
why is proline unique?
has a secondary amino group
the side chain of proline forms a covalent bond with the amino group, making it cyclic
it’s usually on the surface of proteins bc the rigid ring structure is suited for U turns in protein structures
it’s still in the aliphatic classification
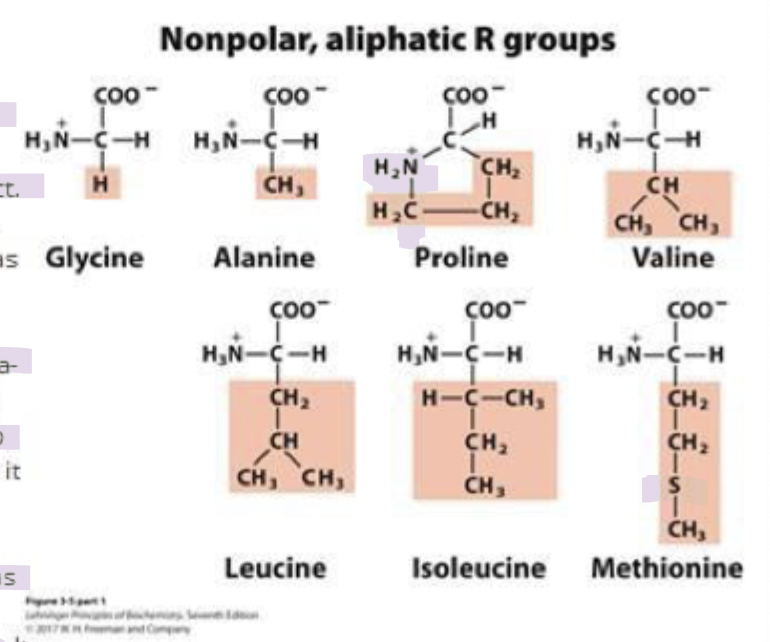
methionine
one of the 2 sulfur containing aa in R group
has a slightly nonpolar thioether side chain
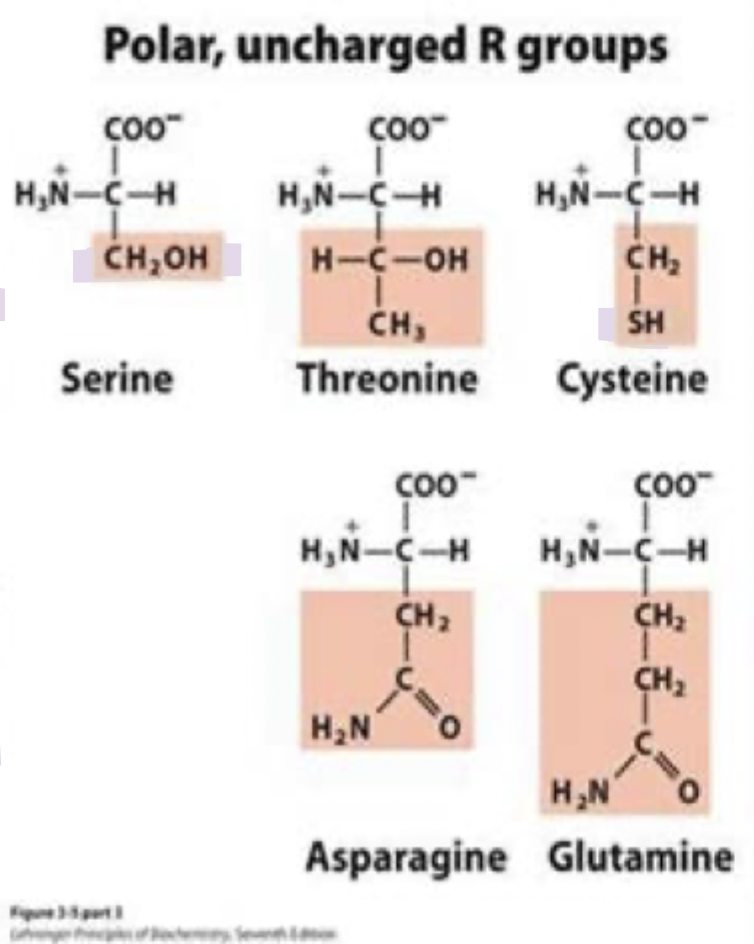
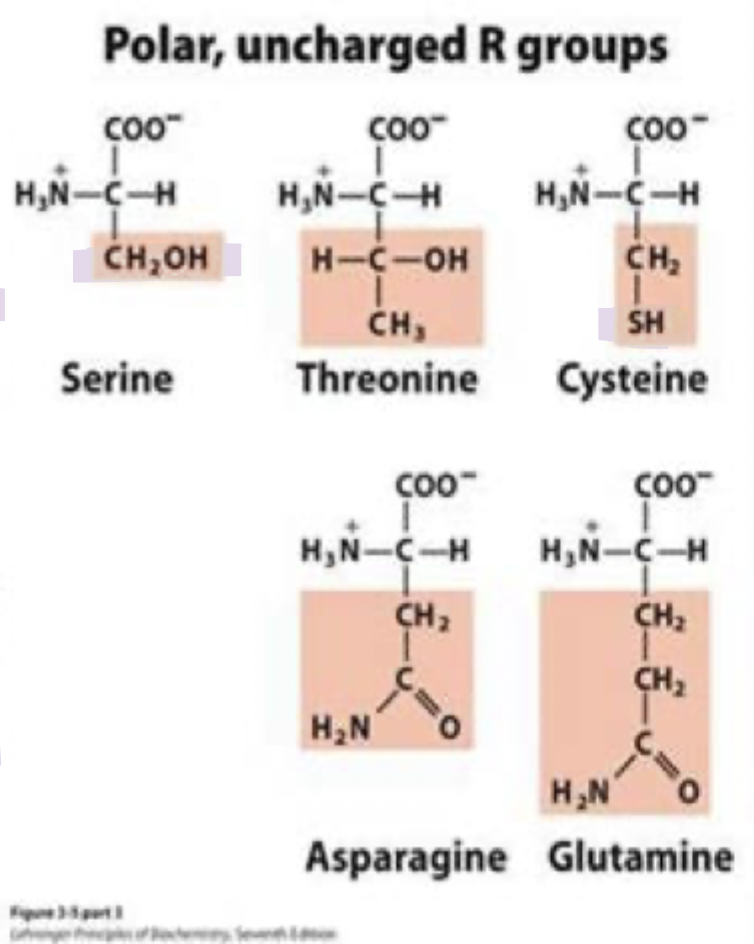
what are the polar uncharged amino acids? (5)
polar side chains that form H bonds with water/other polar molecules
Serine (Ser) S
Threonine (Thr) T
Cysteine (cys) C
Asparagine (asn) N
Glutamine (gln) Q
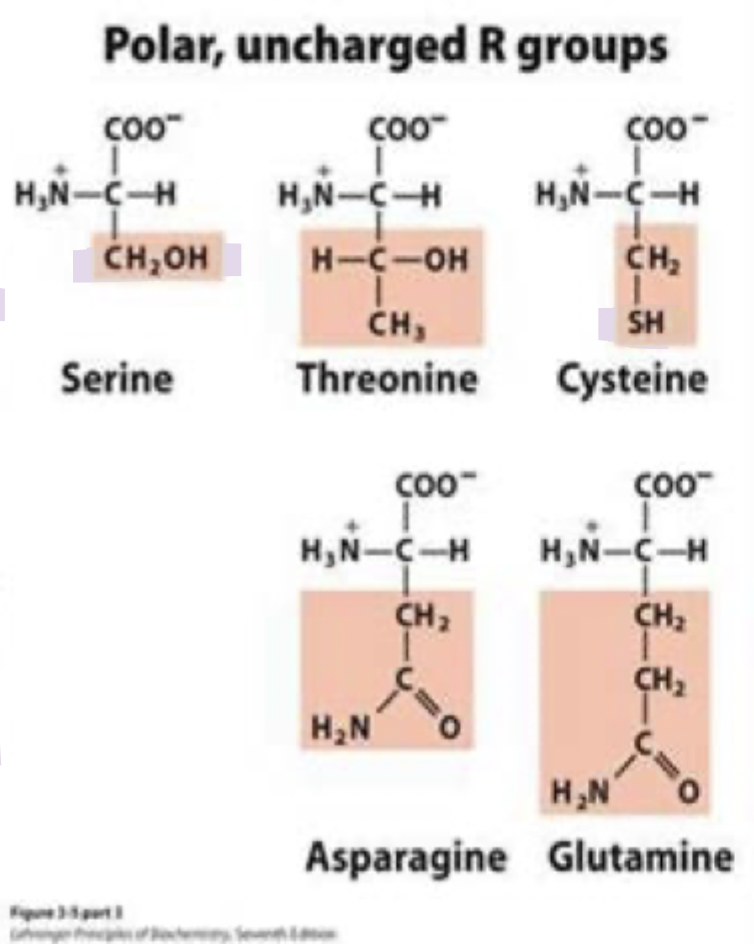
ser and thr
often found on protein surfaces
they make great contact with (aq) end
the -OH is a good nucleophile
serine typically found in enzyme active sites
threonine is secondary alcohol -OH so it’s not as good
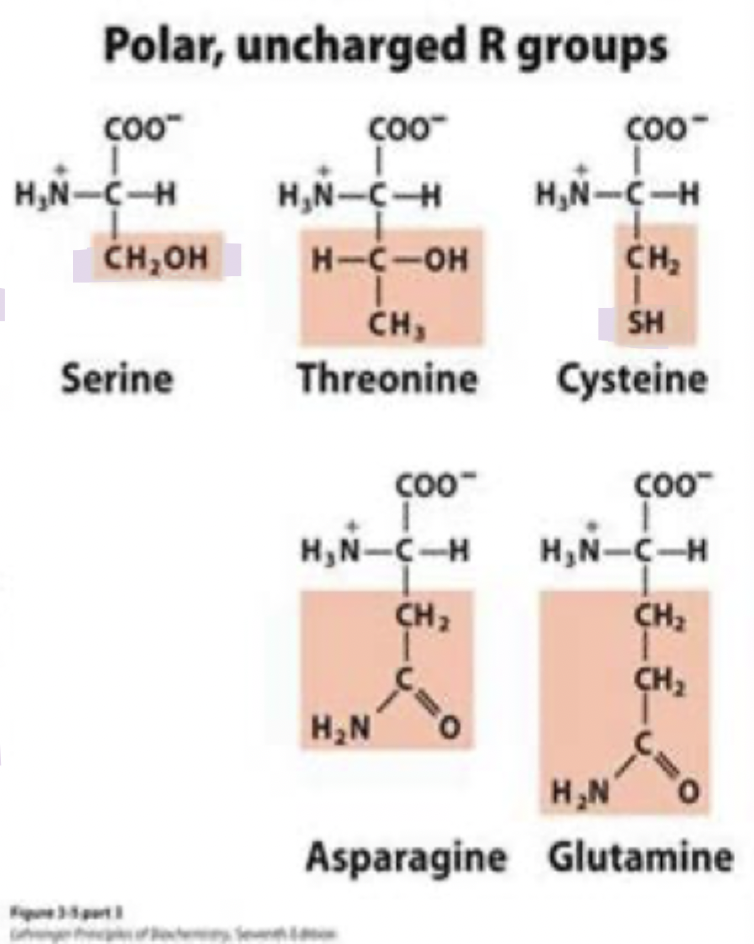
cysteine
can form H-bonds with water or other polar molecules
often found on protein surfaces
-SH group is a great nucleophile
typically found in active sites of enzymes
asn and gln
asparagine and glutamine
have non-ionizable un-charged polar side chains
they’re amide counterparts of asp (aspartic acid) and glu (glutamate)
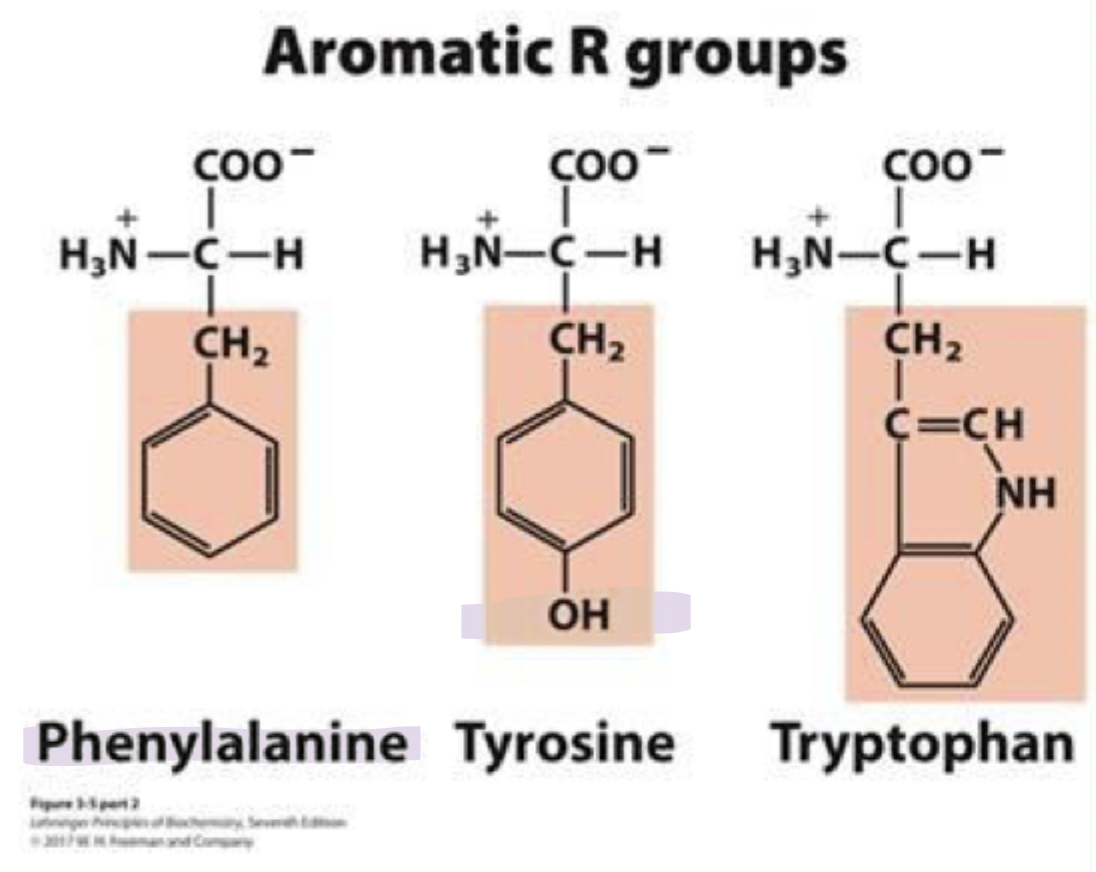
what are the aromatic r group amino acids (3)
they’re hydrophobic and absorb UV light at 280nm
this can be used to measure protein concentrations
phenylalanine (phe) F
tyrosine (tyr) Y
tryptophan (trp) W
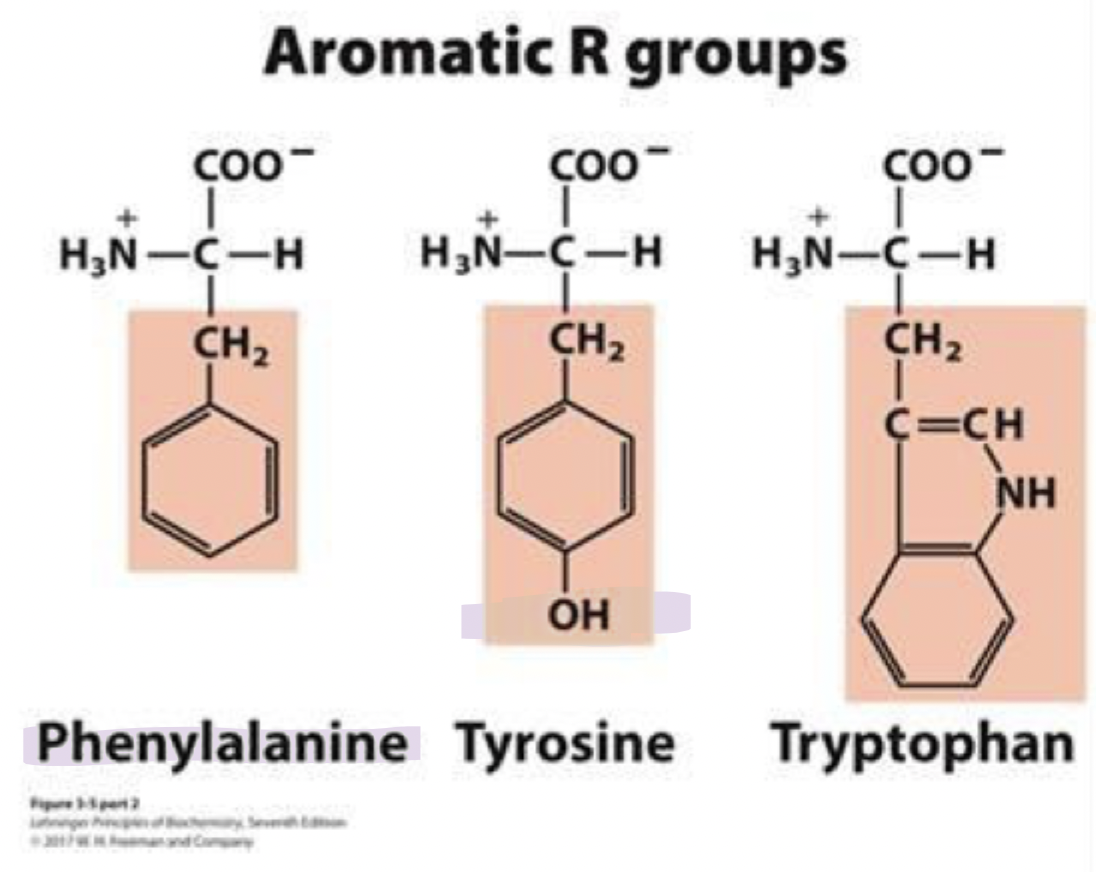
phenylalanine
phe, F
most hydrophobic among the aromatic amino acids
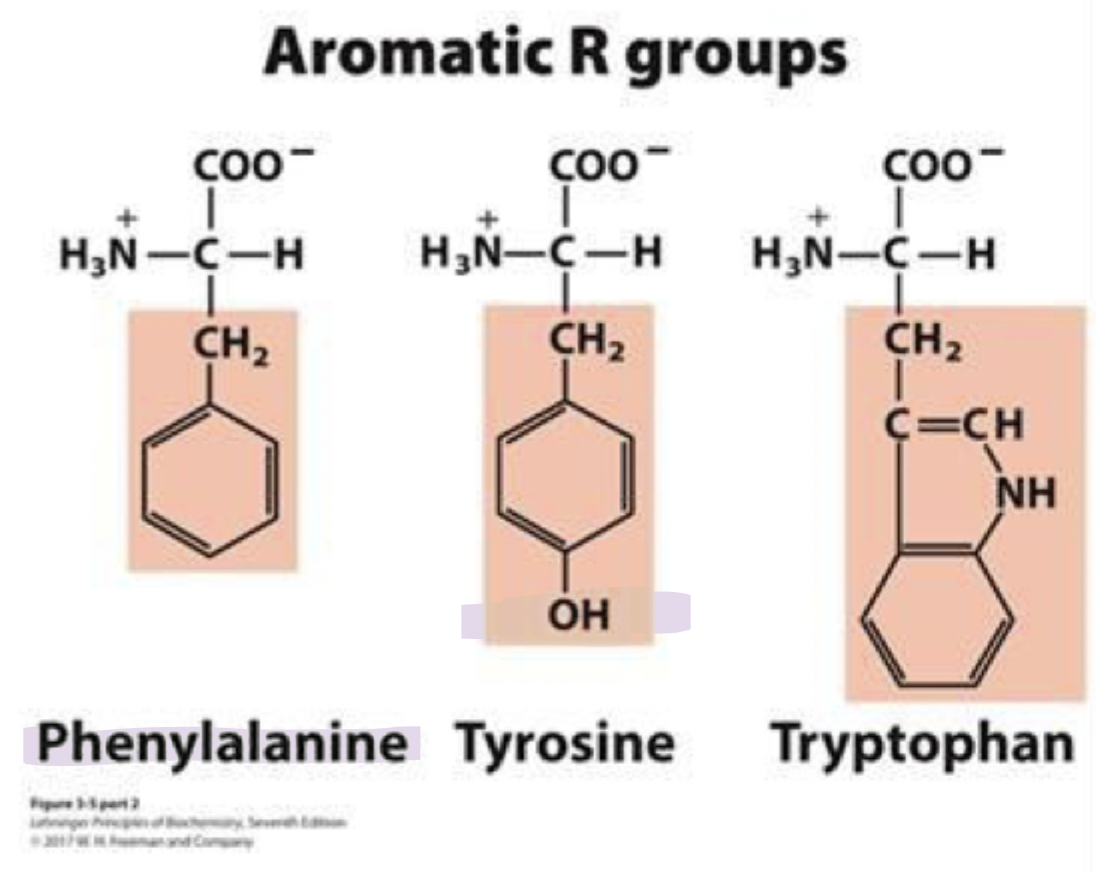
Tyrosine
tyr, Y
has an ionizable group that can form H-bonds
so it’s important in some enzymes
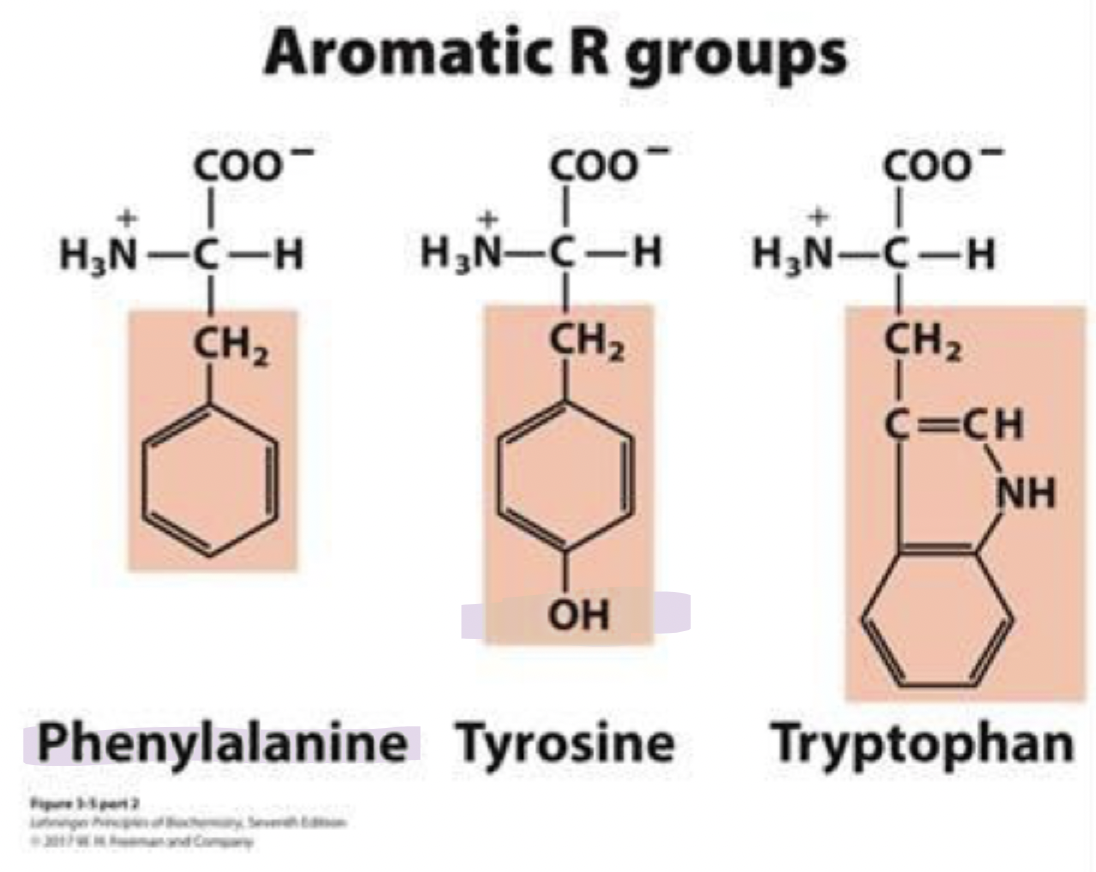
tryptophan
trp, W
bulky af
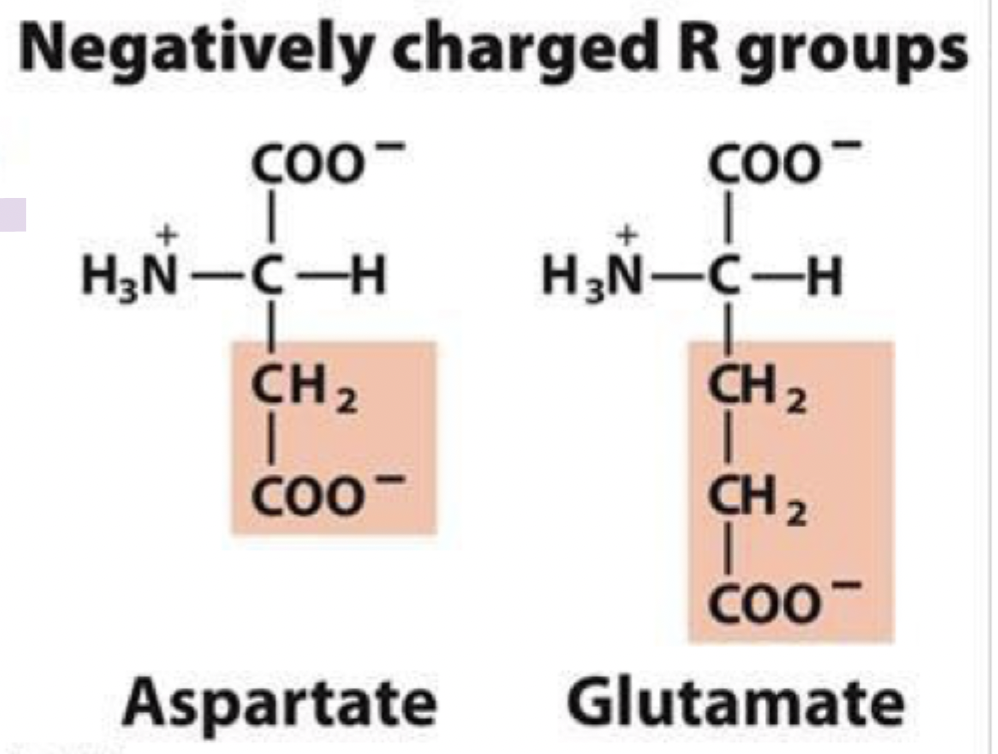
negatively charged R grouped amino acids (2)
they’re (-) at pH 7 and hydrophilic
they’re usually found on protein surfaces
aspartate (aspartic acid) (asp) D
glutamate (glutamic acid) (glu) E
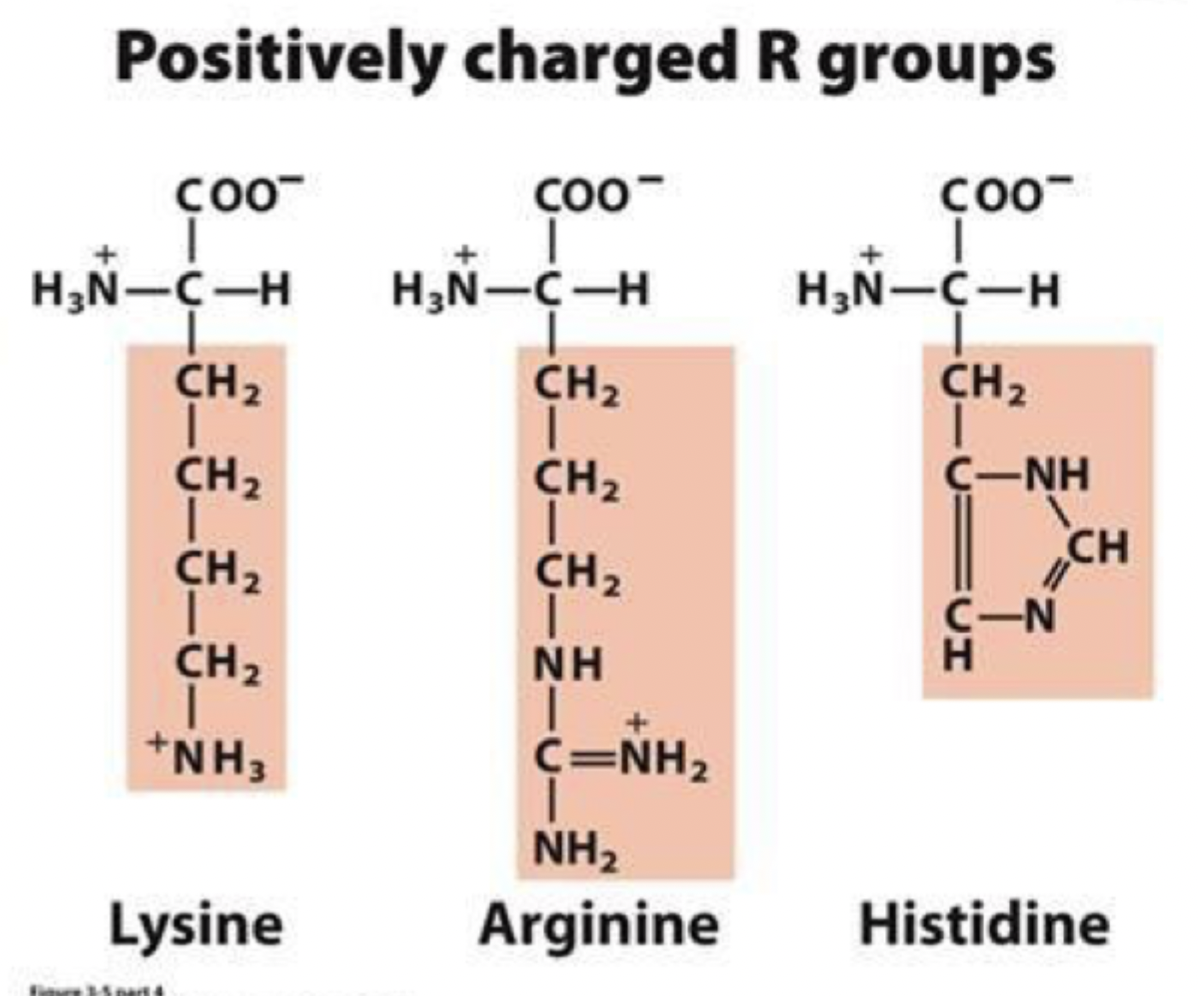
positively charged R grouped amino acids (3)
they’re basic at pH 7
they’re very polar and found on the protein exterior
histidine is the least basic
Lysine (lys) K
arginine (arg) R
histidine (his) H
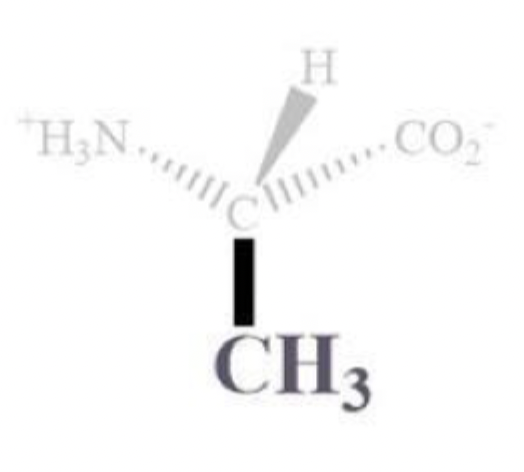
alanine info and side chain
ala, A
CH3 side chain
NP and aliphatic
ambivalent (mixed)
can reside outside or inside bc of a smaller side chains
valine info and side chain
val, v
CH bound to CH3 and CH3 side chain
NP and aliphatic
clusters inside protein bc it’s hydrophobic
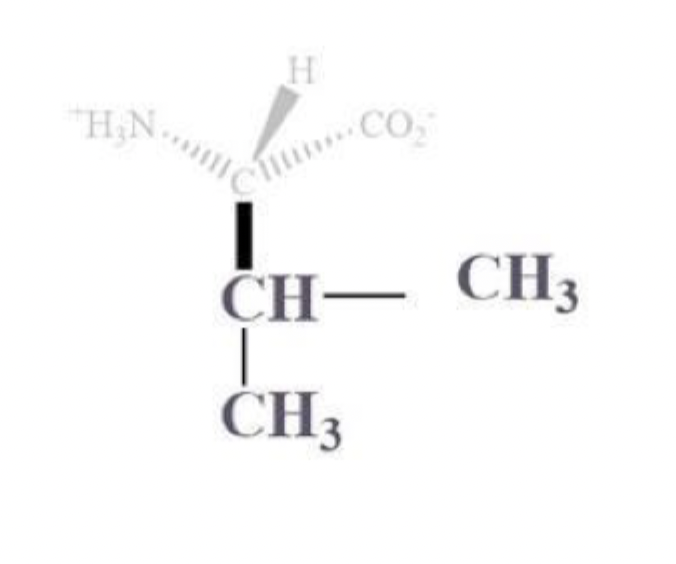
leucine info and side chain
leu, L
CH2 - CH where the CH is bound to CH3 and CH3 sidechain
NP and aliphatic
clusters inside protein bc it’s hydrophobic
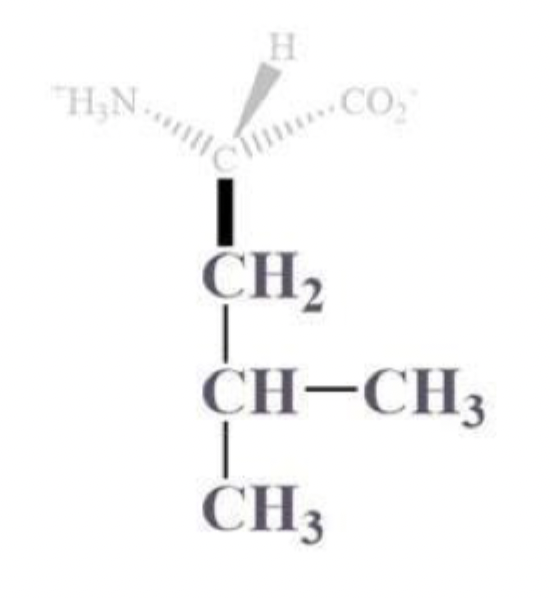
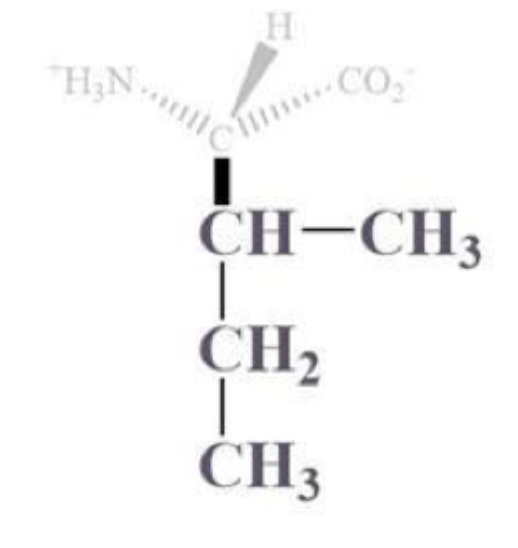
isoleucine info and side chain
ile, I
CH bound to a CH3 and CH2-CH3 side chain
NP and aliphatic
clusters inside protein bc it’s hydrophobic
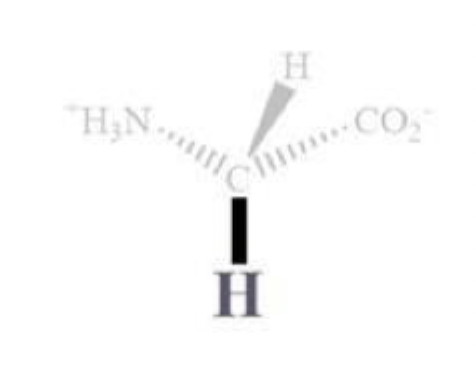
glycine info and side chain
gly, G
H side chain
NP and aliphatic
ambivalent (mixed)
can reside outside or inside bc of a smaller side chains
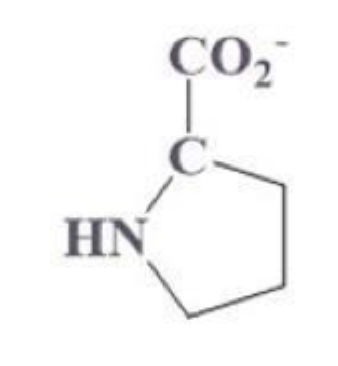
proline info and side chain
pro, P
4 C ring including the secondary carbon (pentagon ring)
has NH within the ring
NP and aliphatic
good for U turns
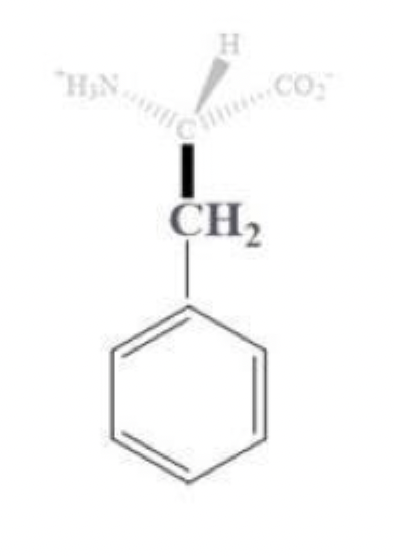
phenylalanine info and side chain
phe, F
has a CH2 - benzene side chain
aromatic amino acid
most hydrophobic among the aromatics
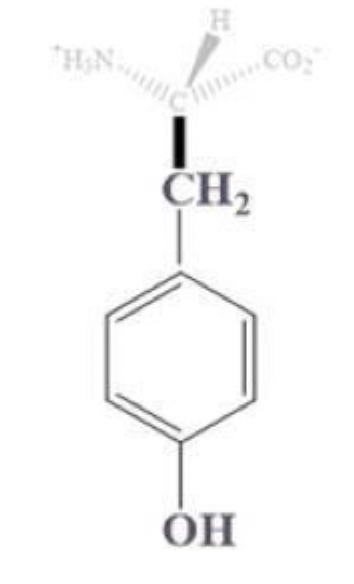
tyrosine info and side chain
tyr, Y
has a CH2 - benzene - OH side chain
aromatic (hydrophobic)
can form H-bonds
so it’s important in some enzymes
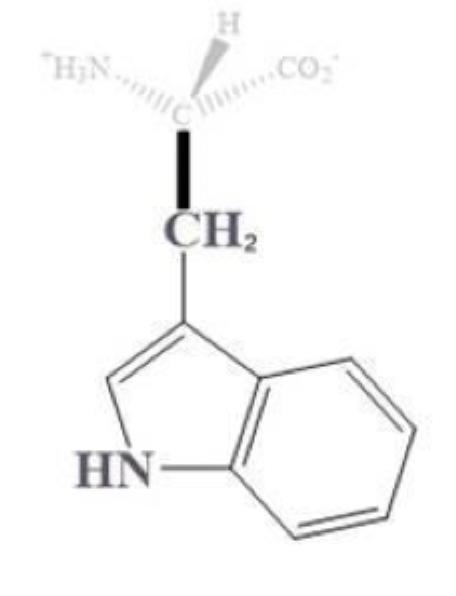
tryptophan info and side chain
trp, W
has 2 rings side chain
aromatic (hydrophobic)
bulky af
serine info and side chain
ser, S
CH2 - OH side chain
polar and uncharged
often found on protein surfaces
the -OH is a good nucleophile
serine typically found in enzyme active sites
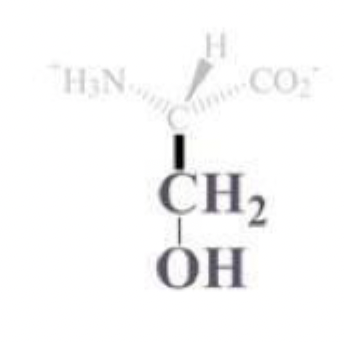
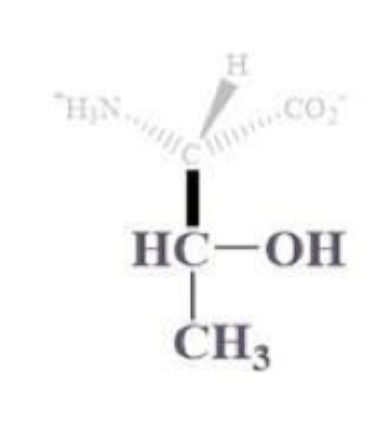
threonine info and side chain
thr, T
CH bound with OH and CH3 side chain
polar and uncharged
often found on protein surfaces
the -OH is a good nucleophile, but not the best bc secondary alcohol
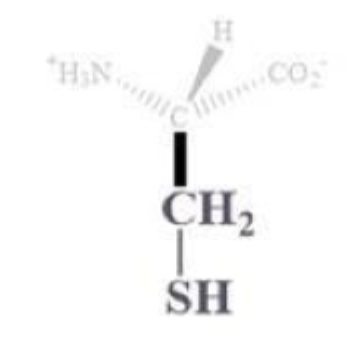
cysteine info and side chain
cys, C
CH2 - SH side chain
polar and uncharged
usually found on protein surface
-SH is a great nucleophile so it’s in enzyme active sites
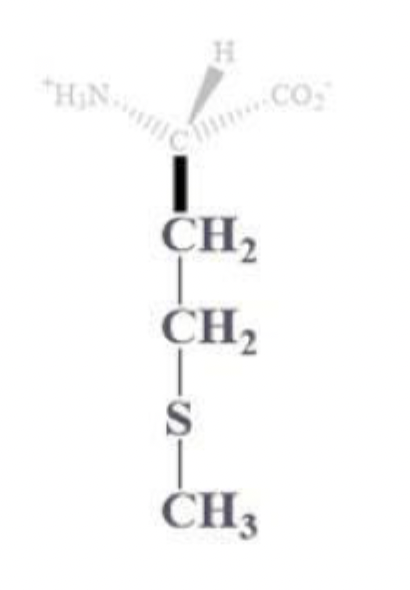
methionine info and side chain
met, M
CH2 - CH2 - S - CH3 side chain
NP and aliphatic
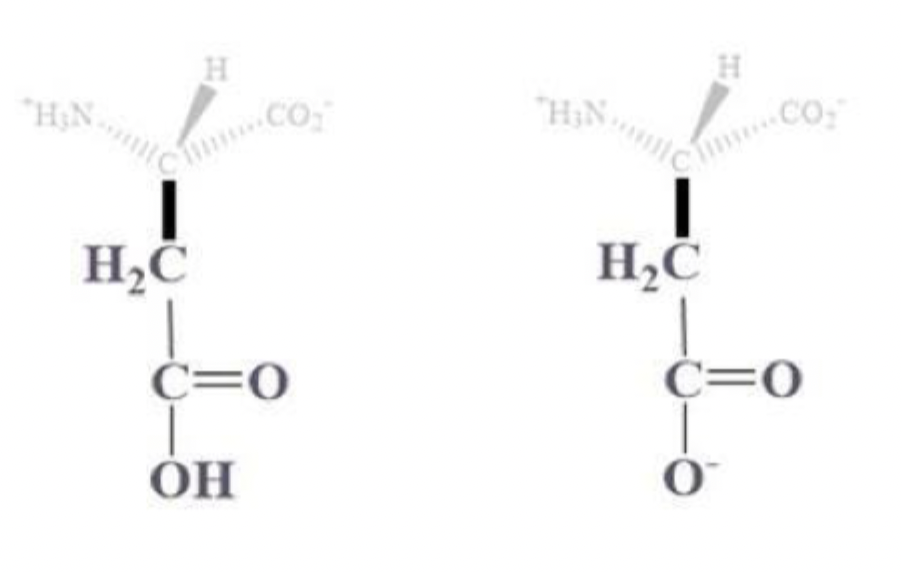
aspartic acid info and side chain
asp, D
CH2 - C which is bound to (=O) and OH (or O-) side chain
(-) charged amino acids at pH 7
hydrophilic and found on protein surface
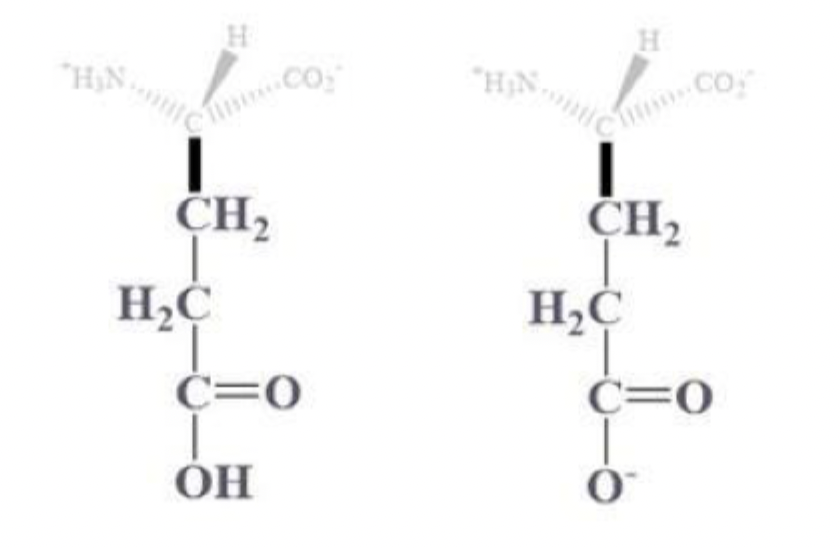
glutamic acid info and side chain
glu, E
CH2 - CH2 - C which is bound to (=O) and OH (or O-) side chain
(-) charged amino acids at pH 7
hydrophilic and found on protein surface
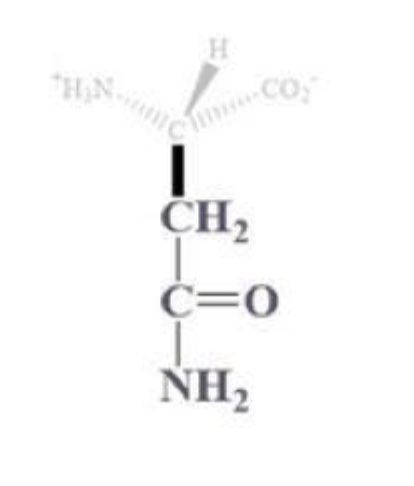
asparagine info and side chain
asn, N
CH2 - C that is bound to (=O) and (-NH2)
amine derivative of aspartic acid
polar uncharged amino acid
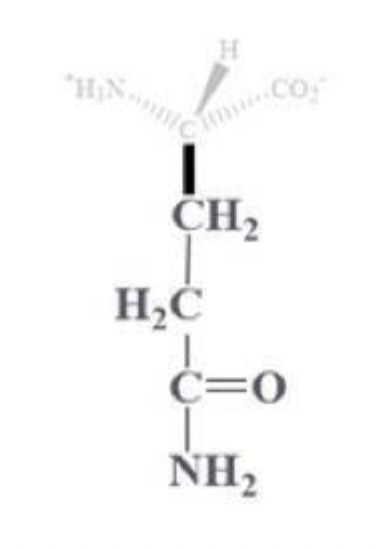
glutamine info and side chain
gln, Q
CH2 - CH2 - C that is bound to (=O) and (-NH2)
amine derivative of glutamic acid
polar uncharged amino acid
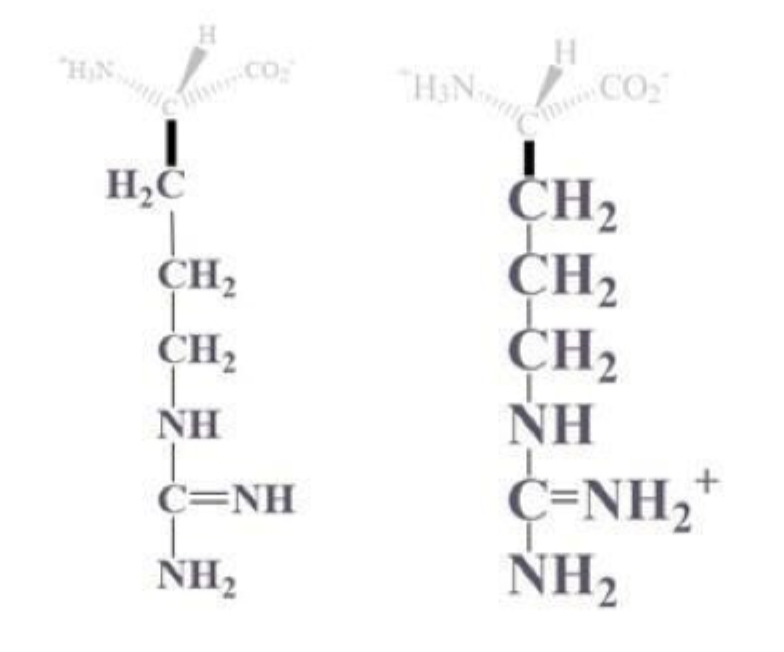
lysine info and side chain
lys, K
CH2 - CH2 - CH2 - CH2 - NH2 or NH3+ side chain
(+) charged at pH 7, basic and polar
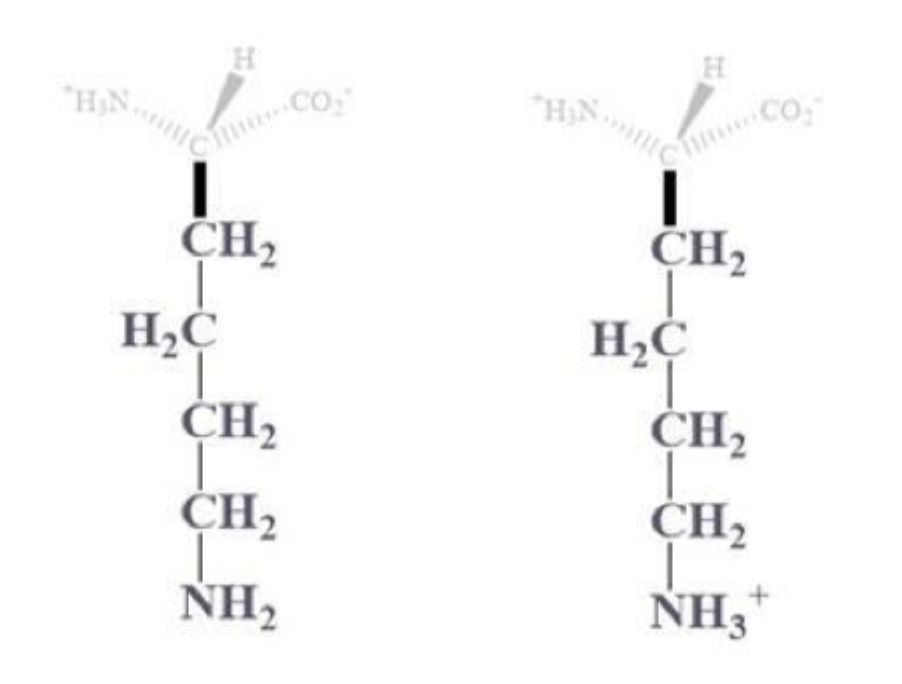
arginine info and side chain
arg, R
CH2 - CH2 - CH2 - NH - C bound to NH2 and (=NH or =NH2+)
(+) charged at pH 7, basic and polar
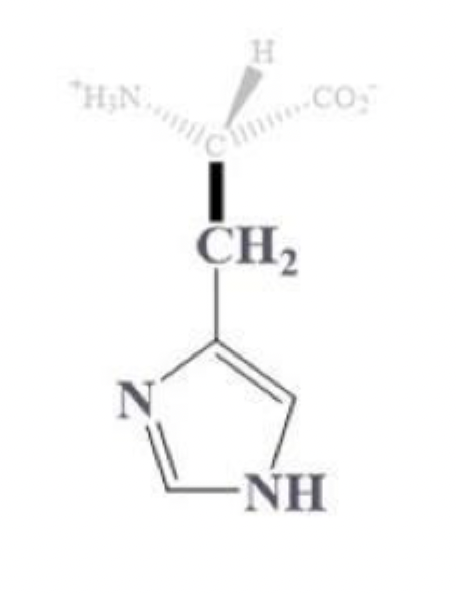
histidine info and side chain
his, H
CH2 - 5 point ring
3C and 2 N
(+) charged at pH 7, basic and polar
least basic among the (+) charged amino acids
SDS Polyacrylamide gel electrophoresis endgoal
seperate proteins based on size
SDS Polyacrylamide gel electrophoresis mechanism
SDS added to a sample of proteins and heated
denatures them
surrounds them with uniform charge
subunits are also seperated
when put through Polyacrylamide gel from the top
it goes towards the bottom (+) bc the polypeptides are (-)
the smaller ones go faster and further
since the charge:mass ratio is the same, the difference in travel is only due to MW
low MW proteins are more mobile