Chap 4B - chemical bonding
1/27
Earn XP
Description and Tags
Polarity of molecules + VSEPR theory
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
28 Terms
Define intermediate bonding
Def. : refer to bonds that do not fit neatly into the traditional categories of purely covalent or purely ionic bonding. Instead, these bonds exhibit characteristics of both types
Most chemical bonds are neither purely covalent nor purely ionic but have characteristics of both ionic and covalent bonding
Compare non polar and polar covalent bonds
Non-polar covalent bond : bonding electrons equally shared as same atoms have the same electronegativity (Eg. H-H , CI-CI , O=O)
Electron distribution is symmetrical -> bond is nonpolar
Polar covalent bond : bonding electrons are not shared equally as different atoms have different electronegativities (Eg. HCI)
Describe the covalent bonding in HCI
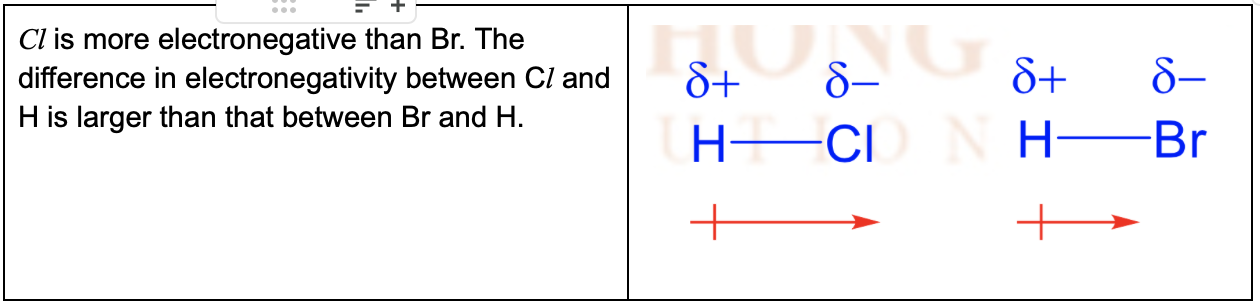
The Cl atom is more electronegative than the H atom
The bonding electrons will be pulled closer to Cl than to H, resulting in a partial charge separation or dipole
Cl acquires a partial negative charge (–) while H acquires a partial positive charge (+)
ELectron distribution is asymmetrical and bond is polar
The 'cross-and-arrow' symbol can also be used to show the dipole moment (towards more electronegative atom)
What is dipole moment
Degree of polarity measured by dipole moment (vector quantity)
Factor :
Increasing electronegativity difference between the two atoms -> greater dipole moment -> covalent bond is more polar
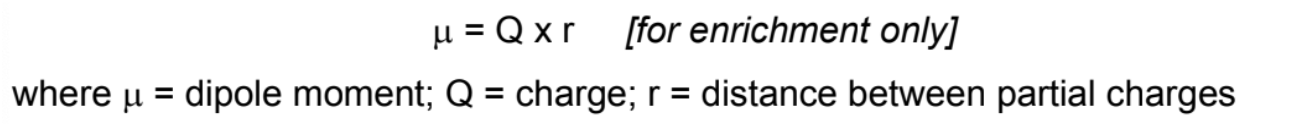
Describe Ionic bonds with covalent character
In ideal ionic bonding, there is complete electron transfer -> The nucleus of the cation and that of the anion only attracts its own electrons -> the electron cloud of each ion is spherical and do not overlap
However, in many cases, highly charged cations exhibit strong polarising power, enabling them to distort the electron cloud of the anion -> this distortion pulls the electron cloud of anion into the space between the nuclei of the cation and anion, resulting in a partial overlap of orbitals and ionic bond gains covalent character
Large anions have large electron clouds which are more easily polarised by the cation
What is degree of covalent character dependent on
Polarising power of the cation (charge density of cation)
Cations that are small and highly charge have high charge densities and high polarising power
These cations have a high tendency to distort the anion’s electron cloud -> greater covalent character in the ionic bonding
Polarisability of anion (radius of anion)
Anions that are relatively large have high polarisability
Their valence electrons are further from and less strongly attracted by the nucleus so the electron cloud is easily distorted by a cation -> greater covalent character in the ionic bonding
Explain why NaI has greater covalent character than KI
The polarising power of Na+ is greater than K+ since it has a greater charge density
Both cations have the same charge of +1 but sodium ion has a smaller radius than potassium ion
Na+ ion will polarise the charge cloud of iodide ion to a greater extent, leading to greater covalent character in the ionic bond
Explain why FeBr3 has greater covalent character than FeCI3
The electron cloud of the bromide ion is more polarisable than the CI-ion since Br- ion has a larger ionic radius
The electron cloud of Br- ion is polarised to a larger extent than CI- ion by the cation, leading to greater covalent character in the ionic bond.
Which of the bonds is more polar , the bond in HCL or HBr? Explain with the help of diagrams.
Account for difference in value of experimental and theoretical lattice energy of silver iodide and silver chloride
Iodide ion has a larger radius than fluoride ion resulting in a greater degree of covalent character in the ionic bond of AgI than AgF.
A larger charge cloud is more easily polarised by the cation (or Ag+) leading to greater additional covalent bonding resulting in a stronger bond and a greater deviation from the pure ionic model.
Define a polar molecule
Polar molecule (Def.) : is a molecule that has an uneven distribution of electron density, resulting in regions with partial positive (δ+) and partial negative (δ−) charges. This occurs when the molecule contains polar bonds (bonds with unequal sharing of electrons) and has a geometry that does not cancel out the bond dipoles
State the criteria for polar molecules
Presence of polar bonds
Molecular Geometry – Asymmetrical Molecular Shape
Even if a molecule has polar bonds, it can be nonpolar overall if the bond dipoles cancel out due to the molecule's symmetry
If the overall or net dipole is zero, the molecule is nonpolar
Explain whether PBr3 and CH3CH3 are polar
Eg. PBr3 (Answering)
PBr3 has 3 polar P-Br bonds which are asymmetrically arranged about P -> dipole moments not cancelled out -> polar molecule
Eg. CH3CH3
CH bonds are very weakly polar due to insignificant difference in electronegativities of C and H
Furthermore, six CH bonds are symmetrically arranged in the molecule resulting in cancellation of any dipole moments -> CH3CH3 non-polar
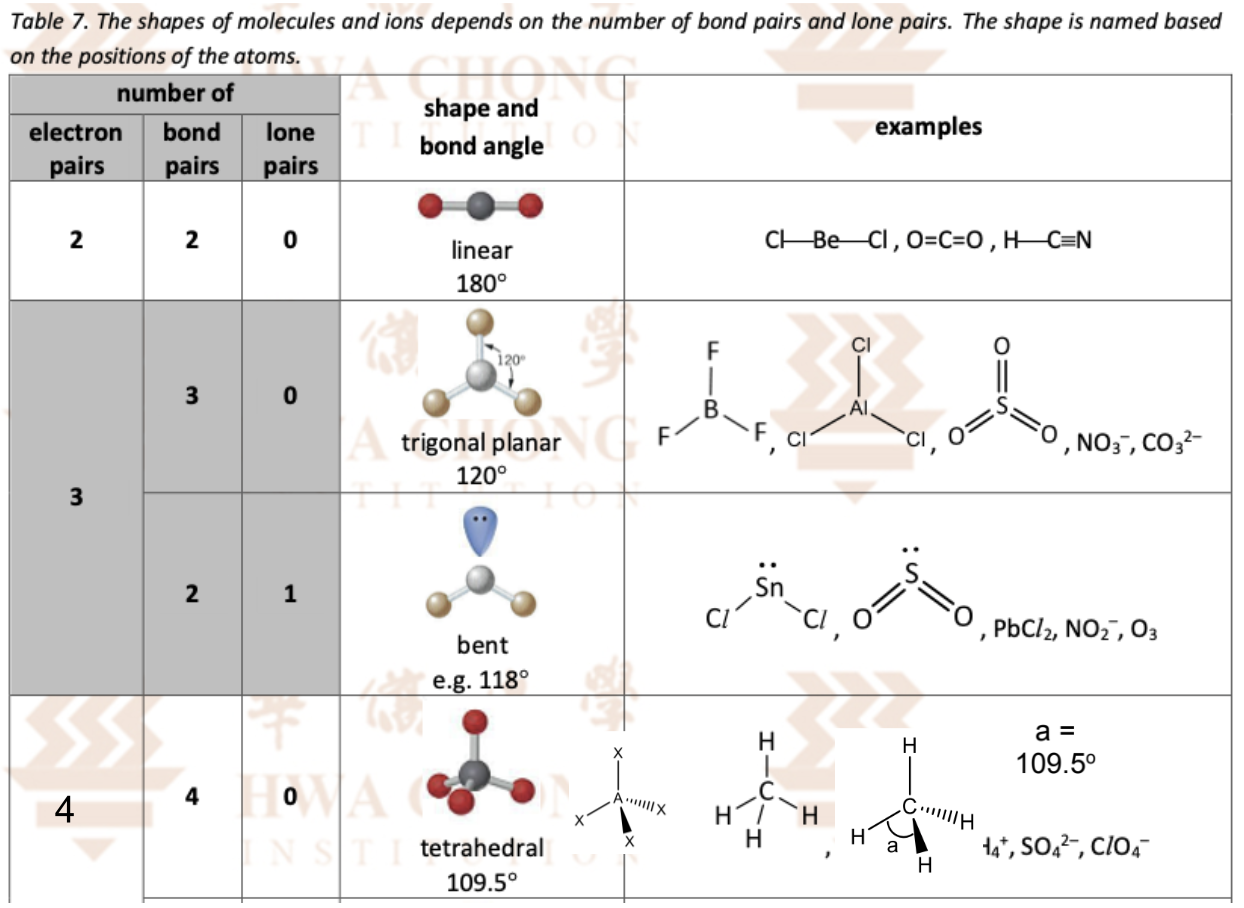
Define VSEPR theory
Def. : states that electron pairs around the central atom will arrange themselves as far apart as possible to minimise repulsion, with lone pairs causing greater repulsion than bond pairs
Bond pair : Electrons that are involved in covalent bonds between atoms
Lone pair : Non-bonding pairs of electrons that belong solely to one atom
State the 2 principles of the VSEPR theory
The electron pairs around the central atom of a molecule (or ion) arrange themselves as far apart as possible so as to minimize their mutual repulsion
The repulsion between lone pair and lone pair > lone pair and bond pair > bond pair and bond pair
A lone pair is attracted by only one nucleus while a bond pair is attracted by two nuclei
The electron cloud of a lone pair is less 'elongated' than a bond pair
Lone pairs have greater repulsion as they occupy more space being closer to the nucleus -> reduce the bond angle between bond pairs -> each lone pair will reduce bond angle between bond pairs by about 2 degrees
Describe the ways bonds affect molecular shape
Pi bonds | Sigma bonds |
|
|
Describe the steps to deduce molecular geometry
Draw dot and cross OR Lewis structure
Count the number of sigma bond pairs and lone pairs surrounding the central atom
There are two bond pairs and one lone pair surrounding the central sulfur atom in SO2
These three electron pairs are in a trigonal planar arrangement
The two sigma bond pairs are in a V shape or Bent arrangement
SO2 has a bent shape
Pi will not affect shape of molecule as they are formed after the sigma bond
Deduce bond angle
The bond angle of trigonal planar shape is 120 degrees
Since there is one lone pair in SO2, the O-S-O bond angle is reduced to about 119 degrees
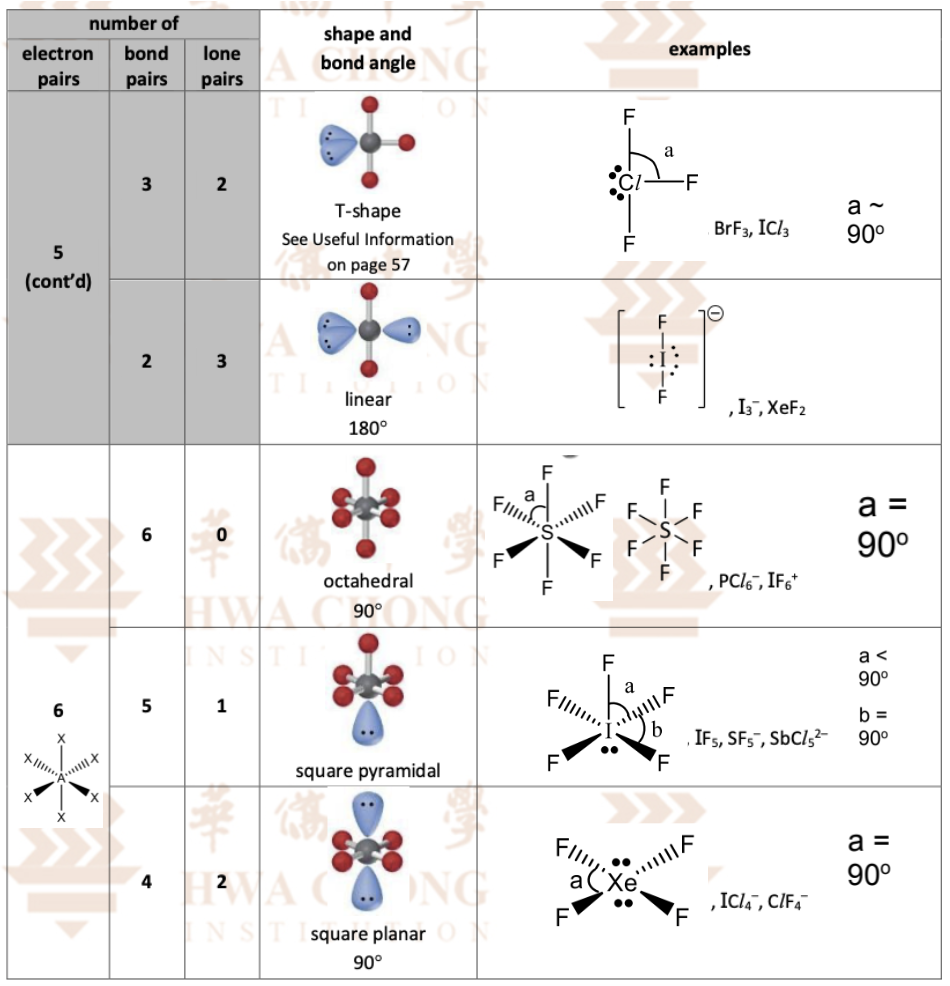
the 2,3,4 electron pairs stuff (VSEPR theory)
the last 2 4 + 5 stuff (VSEPR theory)
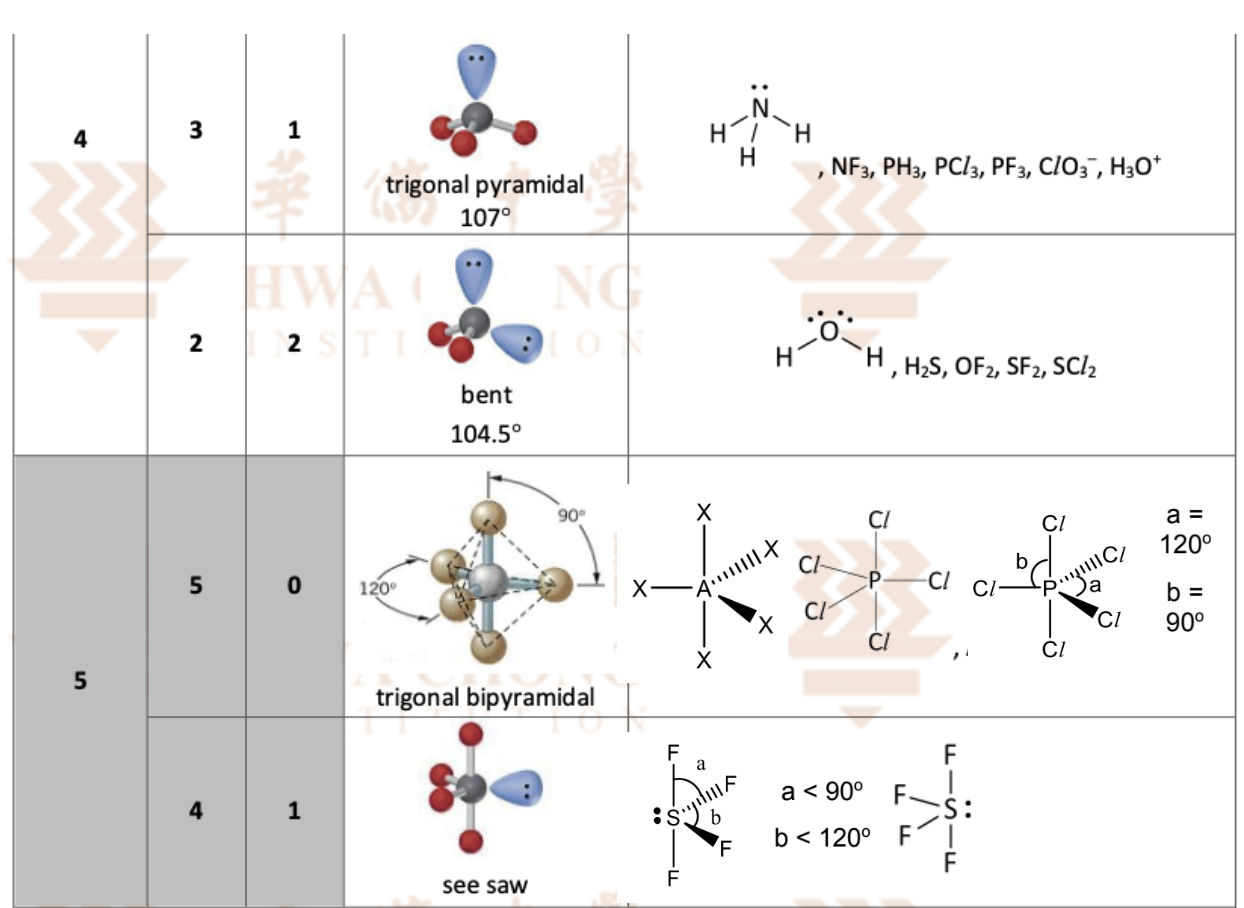
Explain why Lone pairs prefer Equatorial Positions over Axial in a See-saw and Trigonal Bipyramidal Geometry
Axial positions – two vertical positions, 180° apart, and 90° from the three equatorial positions
Equatorial positions – three horizontal positions, 120° from each other and 90° from the axial positions
Lone pairs are more electron-dense and occupy more space than bond pairs
In an axial position, a lone pair would be at 90° to three other equatorial electron pairs, -> creates stronger repulsions.
An equatorial position places the lone pair at 120° from two adjacent pairs, reducing repulsions -> lowers the energy of the molecule -> more stable
the last 2 5 + 6 electron pair stuff (VSEPR theory)
Describe the molecular shape of CO32-
Since there are 3 sigma bonds and no lone pair around the carbon atom, the molecule has a trigonal planar shape with a bond angle of 120 degrees
Describe the molecular shape of PCI3
The central atom P has three bonding pairs (from the three Cl atoms) and one lone pair
The lone pair exerts more repulsion than bonding pairs, pushing the Cl atoms closer together
The ideal tetrahedral angle is 109.5°, but due to lone pair repulsion, the bond angle reduces to ~107°
Describe and compare the molecular shape of SO3 and SO2
Both molecules involve sulfur as the central atom and have three groups of electrons around it, but their shapes and bond angles differ because of the presence (or absence) of lone pairs :
SO₃ |
|
SO₂ |
|
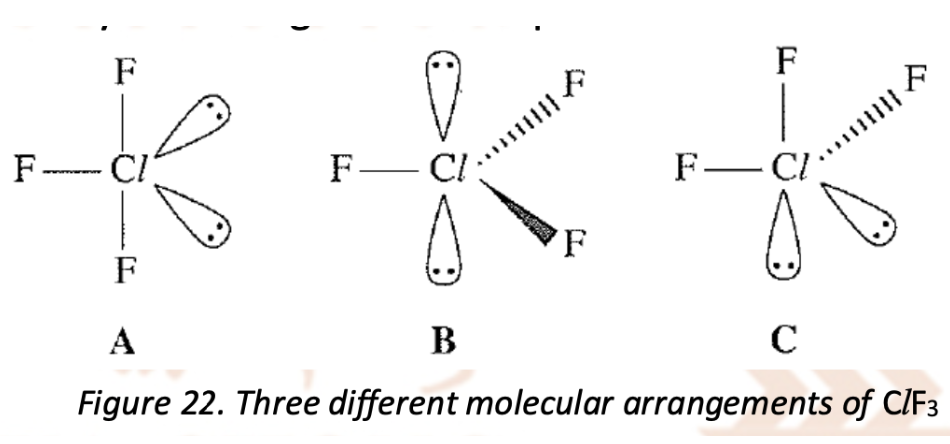
Describe and explain the molecular shape of CIF3 (draw 3 possible arrangements of it)
The central atom is surrounded by five pairs of electrons (three bond pairs and two lone pairs). There are three ways to arrange these five pairs of electrons
C (one lone pair in axial position and the other in equatorial position) is the least stable as the 2 lone pairs are only 90 apart so lone pair-lone pair repulsion is the greatest
B (both in axial position) is less stable than A as B has more significant lone pair-bond pair repulsions
Although the 2 lone pairs in B are 180 apart, the 3 bond pairs are perpendicular to the lone pairs, resulting in significant lone pair-bond pair repulsions
In A (both in equatorial position), there are only 2 bond pairs at 90 to the lone pairs. Hence, ClF3 molecule has a T-shape
Explain how electronegativity affects VSEPR theory
Bond pair position and electrongativity
A central atom which is more electronegative is more likely to draw the electron density of the bond pair towards itself -> The bond pair electrons are nearer to the nucleus of the central atom resulting in more repulsion between the electron pairs
A central atom which is less electronegative is less likely to draw the electron density of the bond pair towards itself -> bond pair electrons are further away from the nucleus of the central atom resulting in less repulsion between the electron pairs.
greater repulsion means larger bond angles
Bond pairs that are closer to the central atom create stronger repulsion with other electron pairs, increasing the bond angle.
If the bond pairs are pulled away toward terminal atoms, the repulsion near the central atom decreases, leading to smaller bond angles
Compare bond angles and shape of H2O and H2S
Oxygen (O) is more electronegative than sulfur (S).
In H₂O, the O-H bond pairs are pulled closer to the central oxygen atom because oxygen attracts electrons strongly.
In H₂S, the S-H bond pairs are further away because sulfur is less electronegative.
Effect on Bond Angles :
In H₂O, the bond pairs are closer to the central oxygen atom, causing stronger repulsion with the lone pairs. This increases the bond angle to 104.5°.
In H₂S, the bond pairs are drawn farther from the nucleus of the central sulfur atom, reducing repulsion, so the bond angle is smaller (approximately 92°)
Explain how size of neighbouring atoms affect VSEPR theory
The larger the size of the neighbouring atoms, the greater the repulsion between the electron clouds of the neighbouring atoms -> larger bond angles
Significant when the neighbouring atoms are much bigger compared to the central atom