adaptive immunity
1/270
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
271 Terms
goal of adaptive immunity
maintain homeostasis by protecting the body from pathogens, foreign molecules, and harmful toxins
does adaptive immunity always do good?
no- sometimes more harm than good
lines of defense
first line: physical barriers
second line: innate immune system
third line: adaptive immune system
first line of defense
physical barriers of the skin and mucus membranes as well as other intrinsic barriers
intrinsic: mechanical, chemical, microbiological barriers
mechanical barriers
Expulsive forces (coughing, sneezing, defecation and urination) help to rid the body of pathogens before they can attach to and breach the mucus membranes)
Ciliary beating helps sweep pathogens from upper and lower airway
Tight junctions in epithelium prevent pathogens from slipping between cells to enter underlying host tissue
chemical barriers
Low pH (stomach, vaginal fluids and sebaceous fatty acids) prevent pathogenic bacterial colonization
Proteolytic enzymes (lysozymes, pepsin in gut) break down pathogenic components
microbiological barriers
Commensal flora compete with pathogens for resources (space and nutrients)
innate immune system
use of surface receptors on phagocytic cells that recognize evolutionary conserved patterns unique to pathogens (ex. LPS- lipopolysaccharide, a sugary lipid found in outer cell wall of gram -ve bacteria)
cells and components in innate immune system
phagocytes
NK cells
inflammation
antimicrobial proteins
fever
phagocytes
Mainly consist of tissue resident and “wandering” macrophages
Ingested pathogens occupy phagosome then fuses with lysosome to produce a phagolysome where digestion of pathogen occurs
NK cells
Do not phagocytose pathogens
Secrete toxic chemicals or induce apoptosis in target cells
antimicrobial proteins
interferons and compliment
interferons
released by infected cells to help prevent infection of neighboring cells by inducing neighboring cells to produce anti viral proteins that block protein synthesis and degrade viral RNA
compliment proteins
help enhance inflammation, promote phagocytosis, and cause cell lysis (membrane attack complex)
fever
Systemic result of infection
Initiated by release of pyrogens which include bacterial toxins and/or components. Antigen-antibody complexes or substances released by phagocytes
pyro= fire
adaptive immunity
eliminates pathogens that may have circumvented or overwhelmed the first two lines of defence and to confer protection from new and emergent strains of pathogens
2 streams of adaptive immunity
cellular and humoral
cellular response
involve targeting killing of infected or abnormal cells
humoral response
involve production fo soluble immunoglobins (antibodies) that confer protection from specific pathogens through variety of effector functions
adaptive immunity characteristics
specificity (self vs non self) and memory
specificity
The ability to mount a response to a particular pathogen or foreign substance while being able to discriminate between self vs non self antigens
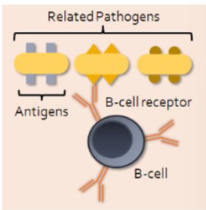
consequences of specificity
B cell may only be able to mount a response to one particular strain of pathogen without recognizing any of a number of closely related pathogens
memory
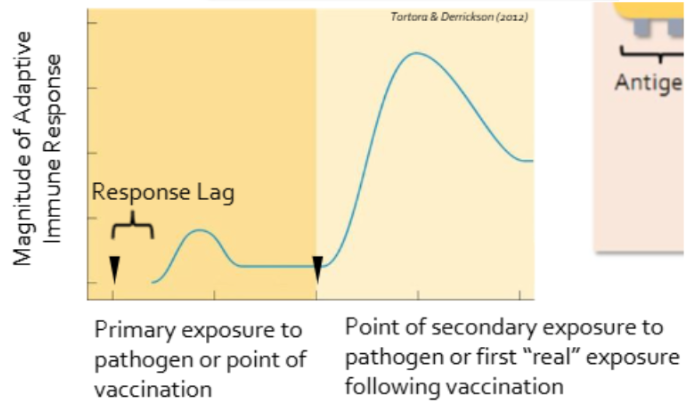
ability to recall exposures to specific pathogens and provides the adaptive immune system with the ability to mount an even stronger response to a pathogen upon repeat exposure
how to vaccines work
memory component of adaptive immunity
how does the magnitude of the adaptive immune response change with exposure
innate vs adaptive specificity
innate: broad specificity
Recognize broadly conserved PAMPs (pathogen associated molecular patterns)
PAMP location: on structures that are essential for pathogen survival and accessible to host pattern recognition receptors (PRRs) such as the Toll-like receptors (TLRs) which are present on phagocytes
TLRs recognize PAMPs that are common to broad classes of organisms (ex. TLR-4 recognizes LPS which is a component of the outer cell wall of all gram -ve bacteria)
adaptive: narrow specificity
Both B-cell and T-cell receptors recognize very specific antigenic determinants (8-10 amino acids) that may be specific for only one particular strain of pathogen
These antigenic determinants can be any protein on the pathogen and may not be essential for pathogen survival
innate vs adaptive repertoire
innate: limited repertoire
PRRs are encoded in the host germ line DNA, limiting absolute number of PAMPs for which PRRs are produced
If pathogens modify their PAMPs then the host PRRs will not mount a response
adaptive: vast repertoire
B and T cell receptors which recognize antigenic determinants are produced through somatic recombination (mixing, matching, and recombing) of host gene segments, resulting in the generation of T cell and B cell receptors that can detect virtually any antigenic determinant
innate vs adaptive response
innate: immediate response
Components are preformed
Response may include increased phagocytosis, complement activation and inflammation
adaptive: slow response
Response is initiated by antigen presenting cells (APCs) which travel from site of infection to nearby lymph node where immunocompetent cells of the adaptive immune system reside
Process involves antigen presentation, differentiation and clonal expansion of T cell and/or B cell resulting in a cellular or humoral response
3-5 days to initiate
innate vs adaptive memory
innate: lacks memory
Responds in same way each time
adaptive: memory
Memory cells ensure a large reservoir of differentiated cells ready to tackle infection of previously encountered pathogens
where is initiation of an adaptive response
secondary lymphoid tissues:
spleen
MALT
lymph nodes
spleen as origin of adaptive response
filters blood of pathogens
MALT as origin of adaptive response
MALT= mucosal associated lymphoid tissue
tissues that eliminates pathogens at mucosal surfaces before spreading to underlying tissues
includes: adenoids, tonsils, Payer’s patches of the gut, respiratory tract, GU tract, appendix
lymph nodes at origin of adaptive response
Immune surveillance of lymphatic fluid returning from the tissues
Over 1000 in body
Become swollen during infection and may be palpated or visually seen
how does lymph fluid enter and leave lymph node
enter via afferent lymph vessels
leave by efferent vessels
why are lymph vessels important
allows for slow flow to make sure resident T and B cells have enough time to interact with pathogens
what kind of cells do lymph nodes contain
macrophages and dendritic cells that have migrated from infected tissue
this is where adaptive and innate immune systems interact
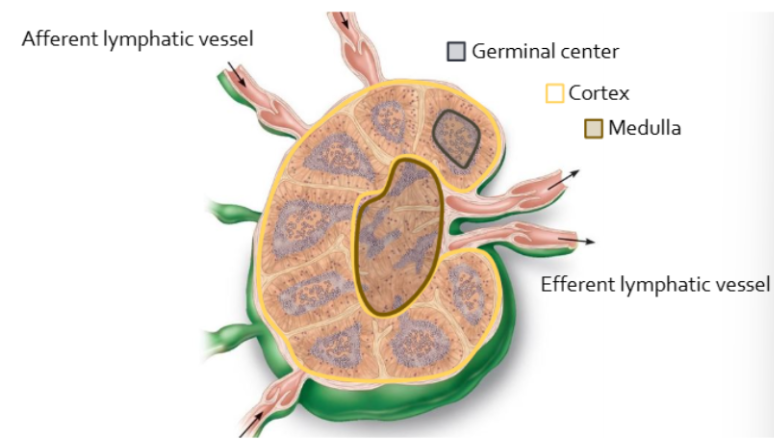
lymph nodes during infection
contain actively proliferating B cells which are largely found within germinal centers of the cortex
B cells will differentiate into antibody secreting plasma cells and migrate to the medulla or enter body tissues where they carry out their immune functions
structure of a lymph node
T vs B cell origin
T cell
red bone marrow
B cell
red bone marrow
T vs B cell site of maturation
T cell
thymus
B cell
red bone marrow
T vs B cell nature of response
T cell
cell-mediated
secreted cytokines
B cell
humoral (antibodies)
T vs B cell target pathogen
T cell
intracellular pathogens and cancers
B cells
extracellular pathogens
look between T (first pic) and B cell (second pic)
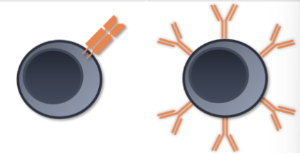
what is the importance of t and b cell receptors
create specificity
both receptors only specific for one antigen or pathogenic determinant because all receptors will be identical
T cell receptors
only recognize antigens that have first been processed by specialized immune cells known as antigen presenting cells (APCs)
APCs process protein antigens by breaking them down into 12-18 amino acid segments that are then packaged and presented to T cells via cell surface receptors termed MHC (major histocompatibility complex)
B cell receptors
recognized unprocessed (native) antigens so no need for APCs
how many pathogenic determinants are there
10^12 since only each recognize one antigen
T cell receptor structure
2 polypeptide chains:
one alpha
one B chain
T cell receptor antigen recognition
bind processed antigen in the context of MHC
B cell receptor structure
modified antibody
4 polypeptide chains:
2 identical heavy chains
2 identical light chains
*heave chains make up the inner v/y
B cell receptor antigen recognition
bind native antigen
somatic recombination
random combination of DNA segments in T and B cell receptor gene regions to create permutations (like shuffling a deck of cards)
different concepts in somatic recombination
VDJ segments
point mutations, deletions, insertions
VDJ segments
random selection fo one V, D, and J segment to be joined (B cell)
V D and J gene segments located on different chromosomes (T cell)
purpose of point mutations, deletions, and insertions
to increase numbe rof possible permutations done before transcription
end result of somatic recombination
array of unique T cell and B cell receptors
how can somatic recombination result in autoimmune disorders
sometimes it can lead to the generation of receptors that recognize self antigens
2 categories for autoimmune disorders
cell mediated
anti-body mediated
cell mediated autoimmune disorders
multiple sclerosis
type 1 DM
multiple sclerosis description
progressive neurodegenerative disorder involving demyelination of CNS axons
MS immunological mechanism
cytotoxic T cells target myelin basic protein expressed by CNS oligodendrocytes
T1DM description
endocrine disorder resulting in hyperglycemia due to insulin insufficiency
T1DM immunologic mechanism
cytotoxic T cells selectively destroy pancreatic beta cells (auto-antibodies against islet cells and insulin also produced)
antibody mediated autoimmune diseases
systemic lupus erythematous
myasthenia gravis
grave’s disease
rheumatoid arthritis
glomerulonephritis
myasthenia gravis description
a neuromuscular disease resulting in progressive weakening or paralysis of skeletal muscle
myasthenia gravis immunologic mechanism
auto-antibodies bind ACh receptors at the neuromuscular junction and induce changes in the postsynaptic membrane, interfering with neuromuscular transmission
grave’s disease description
thyroid disorder resulting in overproduction of thyroid hormone (thyroxine) leading the diffuse goitre and exophthalmos
grave’s disease immunological mechanism
autoantibodies bind to and stimulate thyroid-stimulating hormone receptors (TSH-R) on follicle cells
rheumatoid arthritis description
chronic inflammatory disorder of synovial joint
rheumatoid arthritis immunologic mechanism
auto-antibodies (rheumatoid factor) bind IgG, forming immune complexes in synovial joints resulting in synovitis with destruction of articular cartilage and bone
glomerulonephritis description
impairment of renal function due to inflammation of glomerular basement membrane
glomerulonephritis immunologic mechanism
autoantibodies directed against antigens in basement membrane or trapped antigen-antibody complexes (ex. SLE) induce glomerular inflammation leading to renal dysfunction
systemic lupus erythematous (SLE) description
chronic, complex, multisystem, inflammatory disease
Inflammation from deposition of immune complexes within various organs throughout body which may be affected concurrently or one at a time
SLE immunologic mechanism
Immune complex mediated (type III hypersensitivity reaction)
Composed of IgG antibodies and nuclear antigens such as DNA and histones although IgG antibodies may also complex with erythrocytes, coagulation factors, lymphocytes, and platelets as well
Auto-antibodies commonly produced against nuclear proteins (nucleic acids, histones, ribonucleoproteins)
Nuclear antigens often appear in circulation following cell damage associated with trauma, certain drugs (hydralazine), and infections
clinical manifestations of SLE
Arthritis (90%)
Vasculitis and rash (70-80%)
Renal disease (40-50%)
Anemia (50%)
Cardiovascular disease (30-40%)
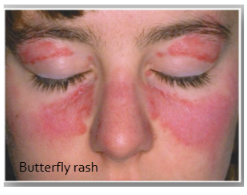
Butterfly rash
Result of exposure to UVB radiation because of release of nuclear antigens from the skin
are male or female more prone to SLE
females
10:1
suggests that estrogen may favour development of SLE while androgens may be protective
what is the goal of SLE treatment
to control the severity of symptoms
pharmacological SLE treatment
NSAIDs
corticosteroids
immunosuppressives
antimalarials
biogenic
IL-2 therapy
NSAIDs for SLE
reduce inflammation adn pain by inhibiting cyclooxygenase enzymes responsible for generating prostaglandins
corticosteroids for SLE
for acute, active disease by reducing immune activity
many undesirable side effects
immunosuppressives
including methotrexate (apo-methotrexate) and azathioprine (imuran) to treat severe symptoms involving internal organs
Interrupt lymphocyte replication by halting cell division resulting in decreased antibody production and a decline in Lupus-related immune complexes
antimalarials for SLE
reduce antigen processing by APCs
Reduce need for corticosteroids
Inhibit early events of antigen processing and cytokine production by macrophages, reducing lymphocyte activation and then autoantibody production
biologics for SLE
BLyS inhibitor (belimumab) reduces B cell survival in active disease
Belimumab reduces B cell numbers so autoantibody titres that, if left unchecked, would otherwise promote immune-complex formation and exacerbate SLE symptoms
Trials indicate that belimumab + standard therapy → decrease severity of SLE symptoms and reduce corticosteroid use
Approved in Canada for use as adjunct therapy in those with active, autoantibody positive SLE
Ex of biologics: vaccines, blood, blood components (antibodies), and recombinant proteins
IL-2 therapy for SLE
promotes T reg cell (regulatory T helper cells) survival
These cells are important in preventing inappropriate immune responses by reducing the action of lymphocytes
This cell population is reduced in SLE
Still being investigated
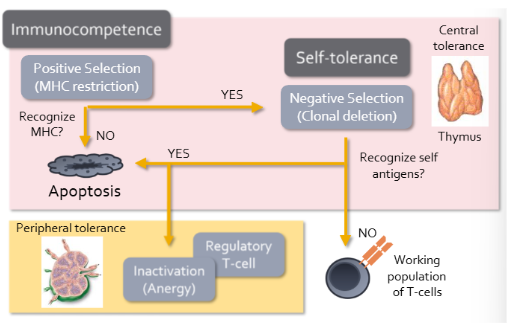
T cell development
Begin with process called positive selection/MHC restriction
Development only continues if T cell that has receptors that can interact with MHC molecules on thymic cells
If it cannot bind to MHC it is useless because cannot communicate to other immune cells
“Dead weight” that the immune system call ill afford
Will receive signal to undergo apoptosis
Those that continue will undergo negative selection/clonal deletion
Specialized thymic epithelial cells present self antigens to developing T cells in the context of MHC
Most T cells that bind strongly to self antigens undergo apoptosis in thymus (prevents autoimmune disease)
T cells that do not bind to self antigens migrate to secondary lymphoid tissues where contribute to working T cells
In thymus also undergo positive selection to ensure that immunocompetent lymphocytes are allowed to develop, but also rise to a form of self-tolerance called “central tolerance”
Sometimes autoreactive T cells escape negative selection
These cells become regulatory T cells that function to suppress immune responses directed against self antigens
Alternatively, autoreactive T cells may be inactivated (called anergy)
Due to activation of T cells requiring additional signals from other immune cells, which are not present unless the immune system is actively employed in fending off an infection
Together the development of regulatory T cells and induction of anergy help make “peripheral tolerance”
Peripheral: adaptive cells are regulated in the secondary or peripheral lymphoid tissues
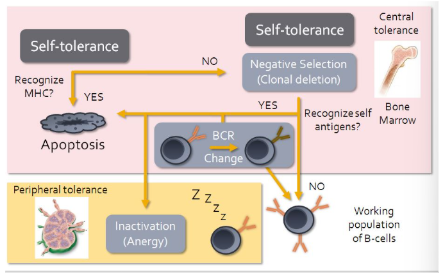
B cell development
Similar except recognition of MHC by B cell receptor will result in apoptosis
Recognition of MHC molecules would indicate that a B cell is autoreactive
Problem because all nucleated cells in body express MHC
While B cells do undergo limited negative selection within the red bone narrow, autoreactive B cells can be induced to change their BCR
In effect they undergo second round of somatic recombination
If successful, B cells contribute to working population
For most part anergy is main mechanism for preventing B cell mediated autoimmune responses if autoreactive B cells make their way out of the bone marrow
measures that prevent autoimmunity
negative selection (clonal deletion) leads to apoptosis
need for costimulatory molecules
anergy
regulatory T cells
need for costimulatory molecules
Need for T cells and B cells to become fully activated antigen presenting cells
Ex. CD80 surface molecule indicated on antigen presenting cell (APC)
Antigen presenting cells only express these costimulatory molecules when they receive danger signals released by innate immune cells or damaged tissues during an ongoing infection
Important to keep both humoral and cellular immunity under tight control
anergy
If an antigen presenting cell does not express the costimulatory signals, its interaction with a T cell or B cell in the lymph node will invariably induce state of anergy
Antigen presentation without costimulation will inactivate lymphocytes, making it even more difficult to activate them in the future
Sometimes anergic lymphocytes will undergo apoptosis and cease to be a burden on the immune system
regulatory T cells
Produced during thymic education
Function to inhibit or suppress activities of autoreactive lymphocytes, contributing to peripheral tolerance and ensuring that autoimmune diseases do not arise
reasons autoimmunity may arise
break-dwon of central and/or peripheral tolerance
molecular mimicry
appearance of new self antigens
genetic predisposition
gender
molecular mimicry
Ex. rheumatoid fever: antibodies produced during strep infection target bacterial M-proteins and may cross-react with self antigens expressed in the heart, resulting in long lasting damage to the heart muscle and valves (rheumatic heart disease)
Streptococcal antigens resemble or mimic self-antigens
The antibodies can cross react with self antigens in the joints and kidneys, leading to arthritis or glomerulonephritis respectively
3 situations where appearance of new self antigens occurs
When mutations is host genes result in the expression of new proteins on cell surface (cancers)
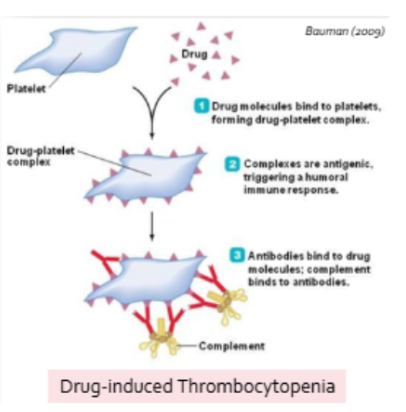
When self-antigens become altered by attachment of small molecules (like drugs or toxins) effectively rendering them foreign in the eyes of the adaptive response
Small nodules (haptens) are incapable of evoking an immune response on their own
Tissue trauma may cause release of self-antigens that were previously sequestered from the immune system
Ex. release of spermatozoa during vasectomy or testicular trauma, exposure of cornea or lens antigens following eye/contact lens trauma, exposure of cardiac muscle following MI
gender and autoimmunity
women are more susceptible (hormones play a role in regulation)
how are T cells identified
based on surface receptor expressed
what is CD
cluster of differentiation
act as co-receptors during immune activation
types of T cells
cytotoxic
helper
regulatory
cytotoxic T cells
CD 8+
targeted destruction of infected or abnormal cells
helper T cells
CD4+
regulates activities of T-cells and B cells
secretes cytokines that regulate innate and adaptive immunity
2 types
Th1: secretes IFN-y
Th2- secretes IL-4
*Th17- secretes IL-17
regulatory T cells
CD4+ and CD25+
mainly suppresses adaptive immune responses
prevents autoimmune disease
secretes IL-10