How far how fast + errors
1/14
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
15 Terms
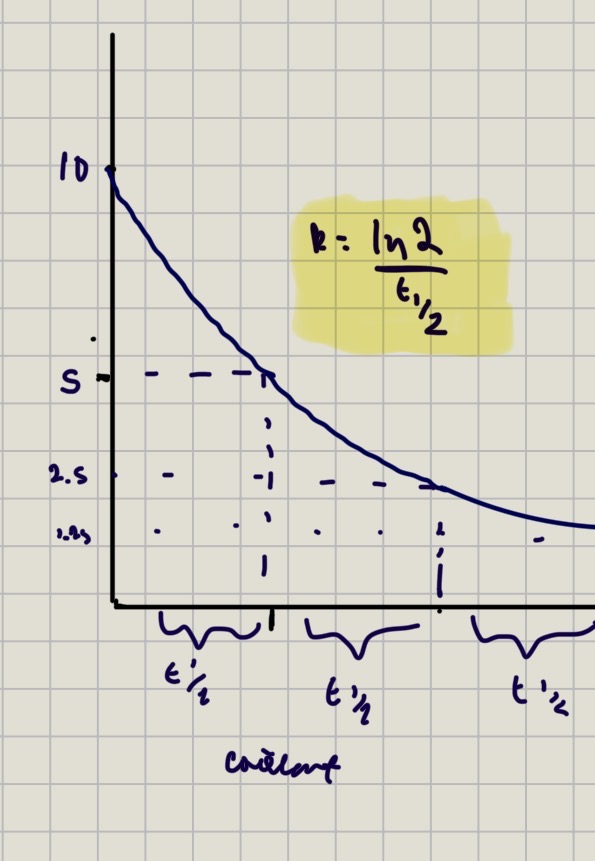
First order’s rate constant from graph
Ln(2) / half time
In first order, half life…
half life stays constant
In zero order, half life..
Half life decreases
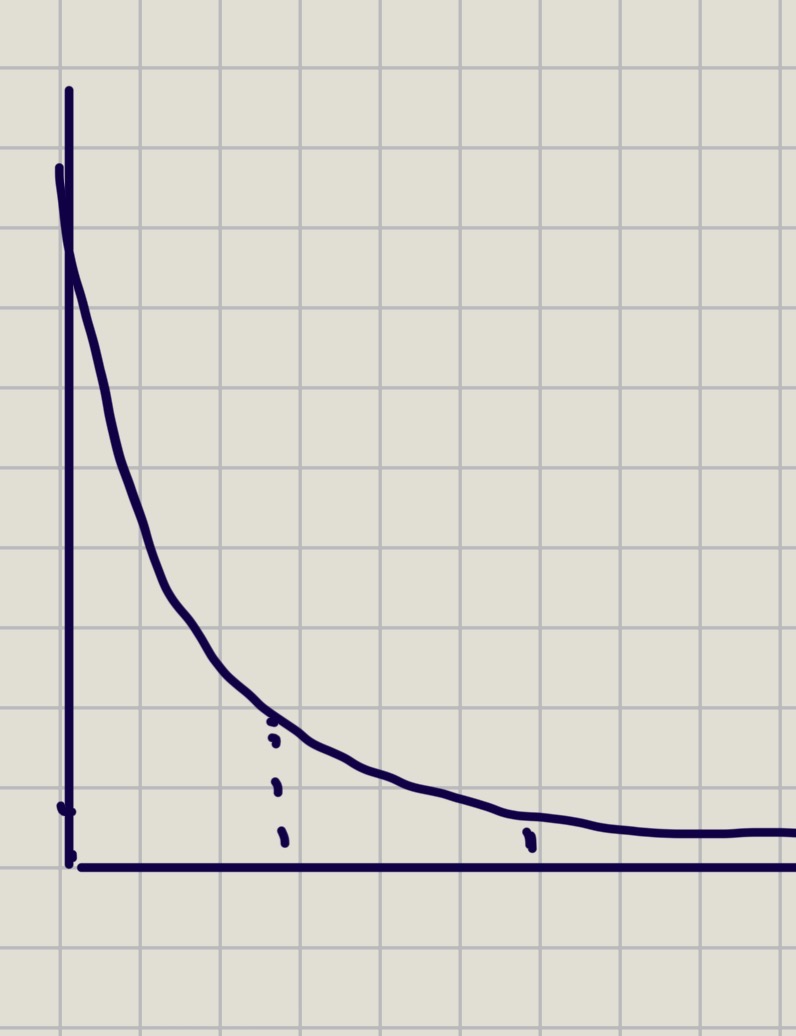
In 2nd order, half life…
Half life increases
Overall order
Sum of orders of reactants
First order concentration graph
Zero order concentration graph
Second order concentration graph
Kp only changes when…
Temperature changes
Why would you add a large excess of a reactant you are not investigating the rate of in an experiment
So that the order of reaction to that reactant is (essentially) zero
how to find activation energy using a converted arhemmius equation graph
( change in ln k as ln k / changee in 1/t as 1/t) x negative gas constant
how to know if reaction is zero order with respect to reactant from concentration / time graph (2)
straight line graph will show that the rate is constant throughout / concentration does not affect rate of reaction
half life will be constant
how to control concentrations by dilution (4)
mix (constant volume) of reactnats
dilute each solution in turn with water
only one solution changed at a time
total volume remains the same
How do we know something ISNT a catalyst
are consumed / used up
are in overall reaction
are not regenerated / reformed
Reasons a reaction may take place in more than one step
Rate equation does not match overall equation
Collisions unlikely with more than 2 ions / species / particles