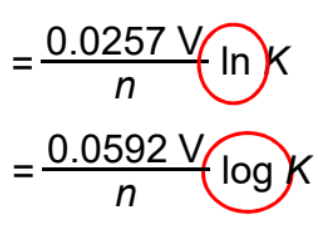Chapter 19 | Electrochemistry
1/11
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
12 Terms
Oxidation-Reduction (Redox) Reactions
Chemical reactions that involve a transfer of electrons
Oxidation number
The charge the atom would have in a molecule (or an ionic compound) if electrons were completely transferred
Known oxidation numbers
Free elements have an oxidation number of 0
Monatomic ions have an oxidation number equal to the charge of the ion
Oxygen = -2
Hydrogen = +1
Group IA metals = +1
Group IIA metals = +2
Fluorine = -1
Oxidation (Anode)
The loss of electrons and an increase in oxidation number
Reduction (Cathode)
The gain of electrons or a decrease in oxidation number
Reducing agent
A substance that reduces something else
Oxidizing agent
A substance that oxidizes something else
Balancing redox equations
Separate the equation into two half-reactions
Balance the atoms other than O and H in each half reaction
For reactions in acid, add H2O to balance O atoms and H+ to balance H atoms
Add electrons to one side of each half-reaction to balance the charges of the half-reaction
If necessary, equalize the number of electrons in the two half-reactions by multiplying the half-reactions by appropriate coefficients
Add the two half-reactions together and balance the final equation by inspection. The number of electrons on both sides must cancel
Verify that the number of atoms and the charges are balanced
For equations in basic solutions, add OH- on both sides of the equation for every H+ that appears in the final equation
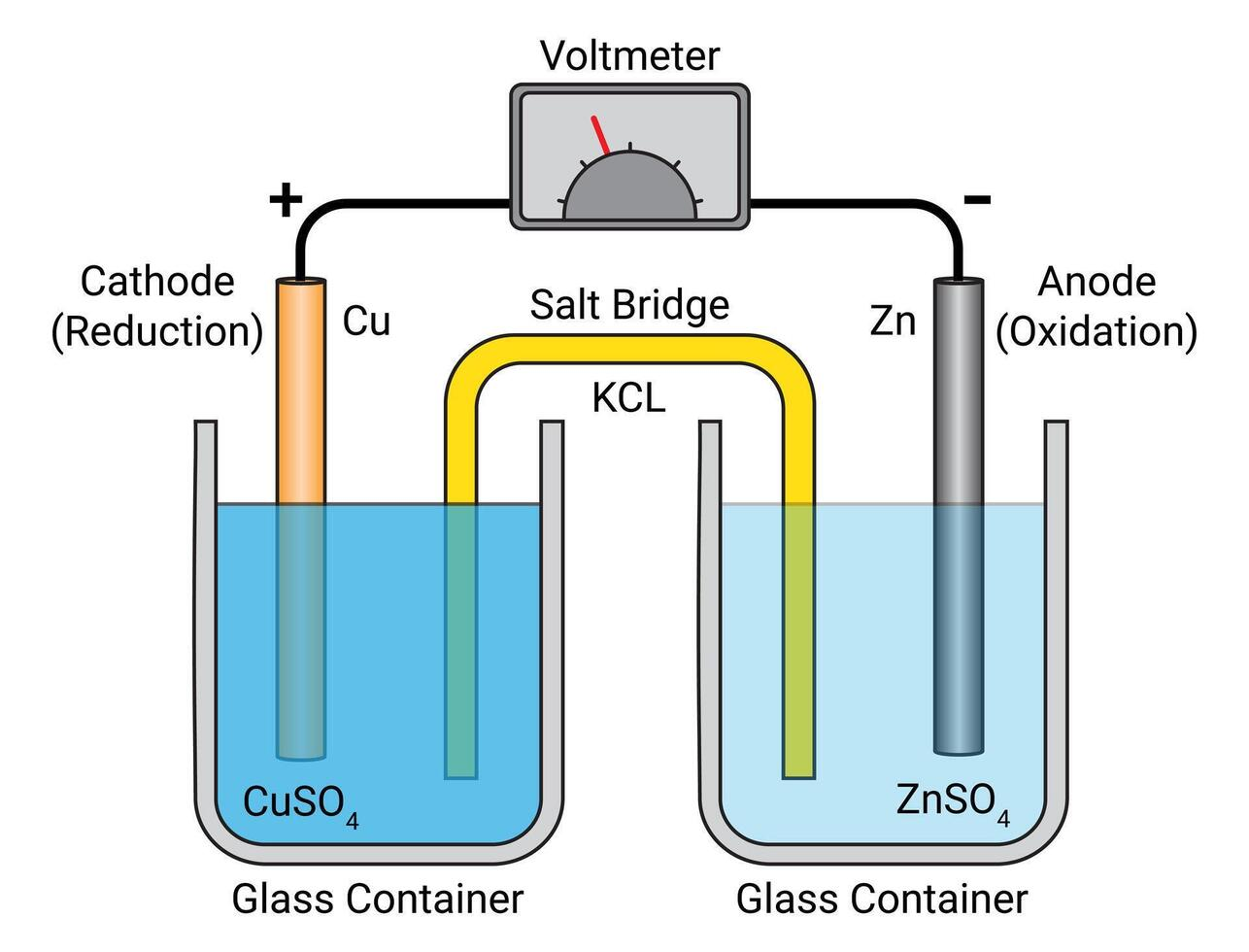
Salt Bridge
A pathway that lets ions flow between cell parts to keep balance and help electricity move
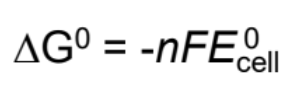
Free energy equation
ΔG = free energy
n = number of moles of electrons in reaction
F = Faraday Constant = 96500 C/mol
E°cell

E°cell and equilibrium