Proteins
1/11
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
12 Terms
zwitterion
bear charged groups of opposite polarity
pI
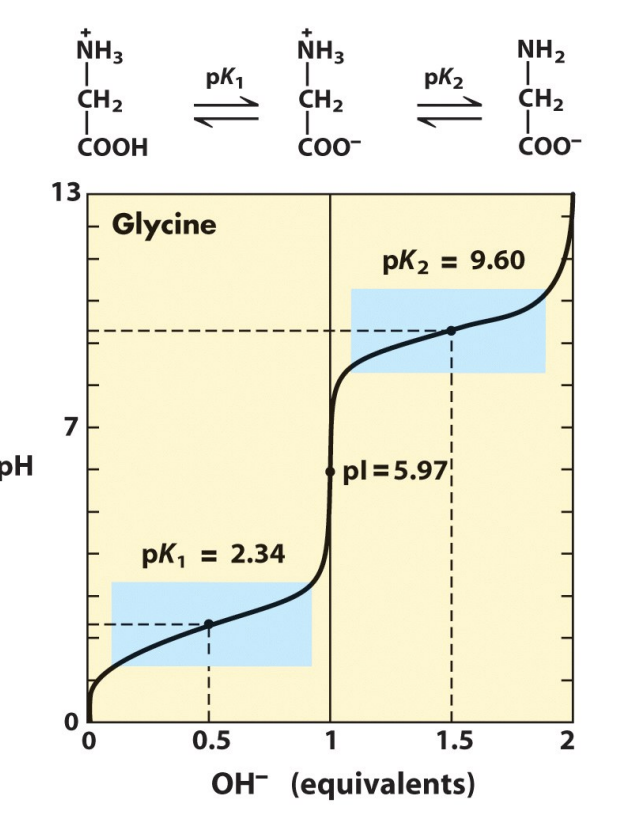
isoelectric point
aromatic amino acids
absorb at 280 nm
2.2
pka for cooh
9.3
pka for NH3+ side chain
ionize readily
disulfide bonds
oxidized to _______ in oxidizing conditions
reversible reactions
can happen between protein subunits
generally, exposed cysteines are in disulfide
6
histidine pKa
closest to neutral!
frequent acid-base catalysis because good at acid base chemistry
at pH 6- ½ neutral and ½ imidazole.
at physiological pH only 4% are ionized—>
free aa pkas
In the free amino acids, the pKa values are much lower, because the positively charged ammonium group electrostatically stabilizes the COO− group, in effect making it easier for the carboxylic acid group to ionize.
Similarly, the pKa values for α-amino groups in proteins range from 7.5 to 8.5. In the free amino acids, the pKa values are higher, due to the electron-withdrawing character of the nearby carboxylate group, which makes it more difficult for the ammonium group to become deprotonated.
exterior
buried cysteines are in disfuldies
exposed cysteins are not in disulfides because it’s energeticalyl expensive
salt bridge
form between positive and negative side chains