All of Unit 3 WJEC A-Level Chemistry
1/341
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
342 Terms
Oxidation
Loss of electrons/hydrogen, gain of oxygen
Reduction
Gain of electrons/hydrogen, loss of oxygen
Oxidising agent
Species that oxidises another species, and gains electrons in the process, becomes reduced itself
Reducing agent
Species that reduces another species, and loses electrons in the process, becomes oxidised itself
Oxidation number of elements in their standard state
0
Oxidation number of hydrogen
1 unless it’s with a Group 1 metal, then it’s -1
Oxidation number of oxygen
-2 unless it’s a peroxide when it’s -1, or reacted with fluorine, when it’s +2
Oxidation number of group 1
1
Oxidation number of group 2
2
Oxidation numbers of elements in a compound or ion
Sum = 0 or charge on the ion
Oxidation numbers and electronegativity
The most electronegative element is given the negative oxidation number.
Oxidation numbers and reduction
Decreases if reduced
Oxidation numbers and oxidation
Increases if oxidised
Standard Electrode Potential
p.d/EMF measured when a half-cell is connected to the standard hydrogen electrode
Standard conditions
298K temperature
1atm pressure
1 moldm^-3 comcentration
Standard hydrogen electrode components
Platinum electrode (s)
H+ solution (aq) 1.00 moldm^-3
H2 (g) at 1atm (100kPa)
298K
Standard hydrogen electrode conditions
H+ solution (aq) 1.00 moldm^-3
H2 (g) at 1atm (100kPa)
298K
Function of platinum electrode in SHE
Conducts electricity
Unreactive
Porous
Reaction between 2 SHEs
No redox reaction
Reaction between SHE and half-cell with another metal
Redox as circuit allows electrons to flow between them
How electrochemical cells are formed
2 metal/metal ion half-cells
Components of electrochemical cells
High resistance voltmeter and wire
Salt bridge
How electrochemical cells are connected
High resistance voltmeter
Salt bridge
High resistance voltmeter function in cells
Allows electrons to flow/move from the half-cell where oxidation occurs to the half-cell where reduction occurs. Measures p.d. between the two
Salt bridge function
Completes the circuit
Maintains electrochemical balance between the 2 half-cells
Composition of salt bridge
Usually made from filter paper soaked in saturated solution of KNO3 (aq)
Electrodes based on different oxidation states
Where two ions are in solution (Fe2+/Fe3+ or Cr3+/Cr2O7 2-)
Or where the OS of one species is zero (CL2/Cl-)
Components used when there are 2 ions in solution
Both solutions included in the same half-cell solution
Pt (s) as multiple oxidation states
Components used when the oxidation state of one species is zero
Pt (s) electrode
Direction of flow of electrons and SEP value
L —> R = +ve
R —> L = -ve
LOAN RRCP rule
Left, Oxidation, Anode, Negative electrode
Right, Reduction, Cathode, Positive electrode
Rule used for position (L/R), process and charge
LOAN RRCP
SEP values and oxidising/reducing power
Most positive = strongest OA
Most negative = strongest RA
Order of values written in the electrochemical series
OA + e- —> RA
Cell EMF definition
Electromotive Force. Difference between SEPs of the two half-cells
Cell EMF equation
EMF (cell) = E (most positive or reduced) - E (most negative or oxidised)
Cell EMF and feasibility
+ve = reaction is feasible
-ve = reaction is not feasible
Comparing standard electrode potentials
most +ve = reduced
most -ve = oxidised
Representing Cells using Cell Notation
X (s) (electrode) | X+ (aq) (solution) || Y2+ (aq) | Y(s)
LHS = oxidation from X to X+
RHS = reduction from Y2+ to Y
Single line (|) = change in state
Double line (||) = salt bridge
Cell notation for change in state
single line (|)
Cell notation for salt bridge
Double line (||)
Cell notation when half cell contains 2 ions in solution
Comma between ions as same state
Pt (s) electrode
Hydrogen fuel cell diagram
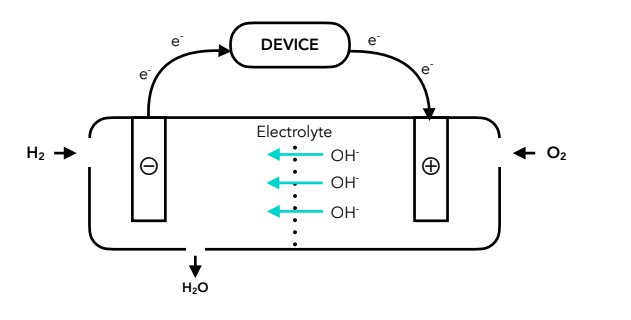
Principles and equations of the hydrogen fuel cell

Advantages of hydrogen fuel cells
more efficient release of energy from fuel
Use a renewable fuel source (hydrogen)
Only by-product formed is H2O
Maintain a constant voltage (compared to chemical cells)
Less heat energy is lost compared to the standard hydrogen combustion engine
Disadvantages of hydrogen fuel cell
Expensive to produce
Toxic chemical used in production
H2(g) is expensive to produce
One source of H2(g) is fossil fuels
Non-renewable
Storing H2(g) can be difficult & dangerous
Metal used in hydrogen fuel cells + why
platinum as it is unreactive
Constructing ion/electron half-equations
Write the reagents and products
Balance the atoms present
Add any extra hydrogen ions that can be used to form water
Find the difference in charge between the start and end to find the number of electrons needed
Combining half-equations
Write then with one beneath the other so the arrows align and are in the correct directions for the reactions
Multiply one or both of them to balance the electrons so both have the same number of electrons
Combine into one equation with all reactants on the left and all products on the left
Cancel the electrons
H+ or H2O can be cancelled too if needed
Need for acidic conditions in redox reactions
Oxidising agent contains oxygen
H+ needed to combine with the oxygen to produce H2O as a product
Number of H+ depends on number of water molecules
Equation for reduction of acidified dichromate
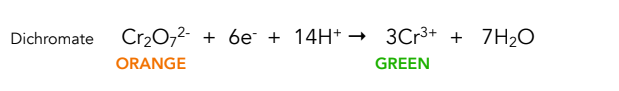
Colour change in reduction of acidified dichromate
Orange to green
Equation for reduction of acidifed manganate
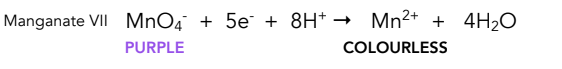
Colour change for reduction of acidified permanganate
Purple to pale pink/colourless
Equation for reduction of iodine
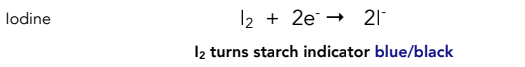
Colour change for reduction of iodine
Brown to colourless
Blue-black to colourless if starch indicator is added
Equation for oxidation of thiosulfate
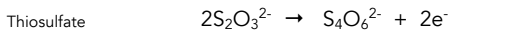
Colour change for reduction of thiosulfate
None
Equation for oxidation of oxalate
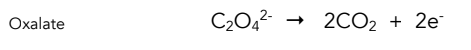
Equation for oxidation for Fe3+
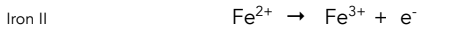
Colour change for oxidation of iron II
Pale green to pale yellow
Equations for redox reaction between Cu2+ and I-
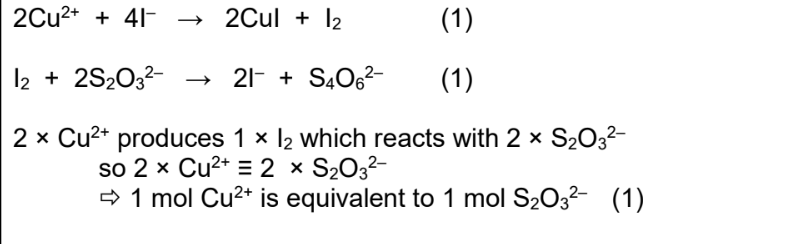
Reaction between aqueous thiosulfate ions and aqueous iodine
Thiosulfate oxidised by iodine
Thiosulfate in butte
Iodine pipetted into conical flask
Thiosulfate run into flask until colour due to iodine fades to pale yellow
Starch solution added as indicator
Colour change to dark blue
End point = blue colour decolourised
Thiosulfate and iodine equation

Reaction between aqueous iodide ions and aqueous copper (II) ions
Blue solution loses its colour
White ppt of CuI forms
Brown solution of I2
I2 titrated with thiosulfate ions
Iodide and copper (II) equation
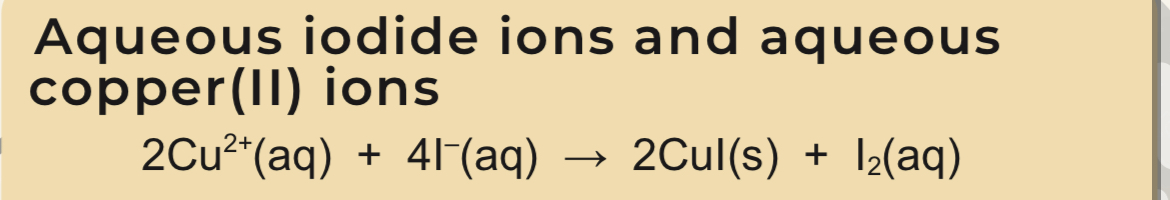
Aqueous dichromate ions and iron (II) ions reaction
Aqueous dichromate ions placed in burette
Added to conical flask
Oxidises aqueous Fe2+
Indicator required
Dichromate and iron (II) equation
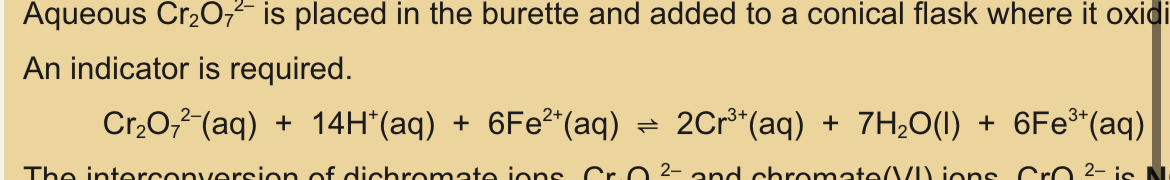
Dichromate to chromate(VI) ions reaction
Not redox as chromium does not change is oxidation number
Dichromate to chromate(VI) colour change
Orange to yellow
Dichromate to chromate(VI) equation
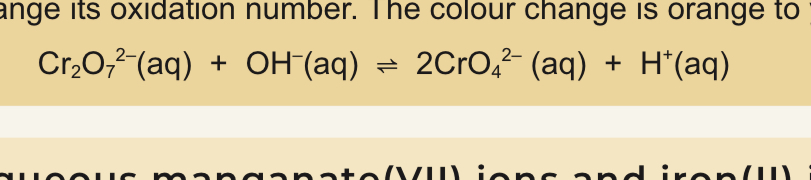
Aqueous manganate(VII) ions and iron(II) ions reaction
Aqueous MnO4^- in burette
Fe2+ solution pipetted into conceal flask with excess aqueous sulfuric acid
No indicator required
End point; all Fe2+ has been oxidised —> next drop of MnO4^- gives a pink colour
Manganate and iron(II) equation
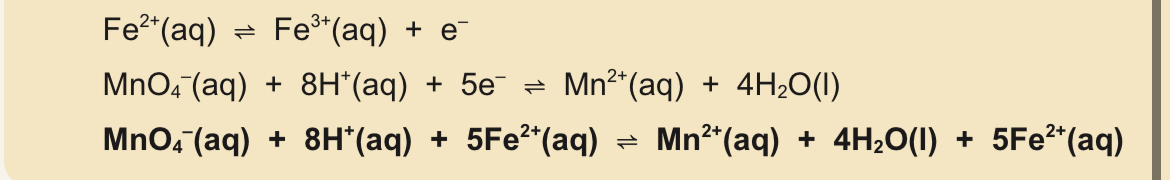
Redox titrations vs acid base
Similarity; use a solution of known concentration of ions to find the contraction of ions in another
Difference; redox involving transition metals usually involve a colour change to no need for indicator to be used
p block electron configuration
Outermost electrons in a p orbital
Oxidation states of p block elements
Maximum oxidation state = group number
Lower = group number-2
Amphoteric
Metals that react with/act as an acid and a base
Examples of amphoteric behaviour
Al³+/Al
Pb²+/Pb
Aluminium Oxide acting as a base
Al2O3 + 6HCl —> 2AlCl3 + 3H2O
Aluminium oxide acting as an acid
Al2O3 + 2NaOH + 3H2O —> 2Na+ + 2[Al(OH)4]- (2Na[Al(OH)4]
Aluminium Hydroxide acting as a base
Al(OH)3 + 3H+ —> Al3+ + 3H2O
OR Al(OH)3 + 3HCl —> AlCl3 + H2O
Aluminium Hydroxide acting as an acid
Al(OH)3 (s) + OH- —> [Al(OH)4]- (aq)
Lead Oxide acting as a base
PbO + 2HCl —> PbCl2 + 2H2O
OR PbO + 2HNO3 —> Pb(NO3)2 + H2O
Lead Oxide acting as an acid
PbO + 2NaOH + H2O —> 2Na+ + [Pb(OH)4]2- (Na2[Pb(OH)4])
Lead Hydroxide acting as an acid
Pb(OH)2 (s) + 2OH- —> [Pb(OH)4]2- (aq)
Lead hydroxide acting as a base
Pb(OH)2 + 3HCl —> PbCl2 + H2O
OR Pb(OH)2 + 2H+ —> Pb2+ + 2H2O
Amphoteric metal solutions and NaOH
Solid white precipitate of metal hydroxide
Precipitate then redissolves of excess to form a colourless solution
Al3+ and NaOH (aq) observations
White precipitate of aluminium hydroxide formed
Redissolves in excess to form a colourless solution
Al3+ and NaOH (aq) equation
Al3+ (aq) + 3OH- (aq) —> Al(OH)3 (s) white precipitate then + OH- (aq) —> [Al(OH)4]- (aq) colourless solution
Pb2+ and NaOH (aq) observations
White precipitate of lead hydroxide formed
Redissolves in excess to form a colourless solution
Pb2+ and NaOH (aq) equation
Pb2+ (aq) + 2OH- —> Pb(OH)2 (s) white precipitate then +2OH- —> [Pb(OH4)]2- (aq) colourless solution
Stable oxidation states of group 3
+3 and +1Stable ox
Stable oxidation states of group 4
+4 and +2
Stable oxidation states of group 5
+5 and +3
Trend in oxidation states down the group
Elements are more likely to form compounds in which they have the lower oxidation state so lower down the group you go, the more likely it is that the elements forms a compound with the lower oxidation state
Inert pair effect
the tendency of the outermost s2 pair of electrons in an atom to remain unshared in compounds, leading to a lower oxidation state.
Trend in inert pair effect
Occurs in groups 3, 4 and 5
Tendency increases/becomes more significant down the group
Cause of trend in inert pair effect down the group
The two outer electrons in the outer s orbital are less likely to take part in bonding
Octet expansion
the ability of some atoms to use d-orbitals to have more than 8 electrons in their valence (outer) shell.