ERTH1501 WK2
1/21
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
22 Terms
Most abundant elements in Earth and crust
Fe, O, Si, Mg
Fe, O, Si, Mg, Al, Ca, K, Na

Calcite (CaCO3)
Hardness = 3
Rhombohedral cleavage
Can form by direct precipitation from seawater or heated fluids in the Earth’s crust
a drop of diluted HCl on calcite produces an instant, obvious fizz

Dolomite CaMg(CO3)2
Hardness = 3
Rhombohedral cleavage
a drop on dolomite produces slow or no obvious bubbling

Aragonite CaCO3
Can form by direct precipitation from seawater or heated fluids in the Earth’s crust
Limestone + Dolostone
Calcite and aragonite
Dolomite
Oxide: Magnetite (Fe3O4)
contains both 2+ and 3+ Fe
alters easily to hematite in more oxygen-rich environments, so rocks that once contained magnetite may be altered to contain hematite instead.
a dark grey – black streak. is magnetic
Oxide: hematite (Fe2O3)
contains only the 3+ type of Fe.
forms in environments that are more oxygen-rich than magnetite
will have a red-brown streak
Examples of carbonates

Examples of halides

Examples of oxides

Examples of sulfides

Examples of sulfates

Examples of native elements

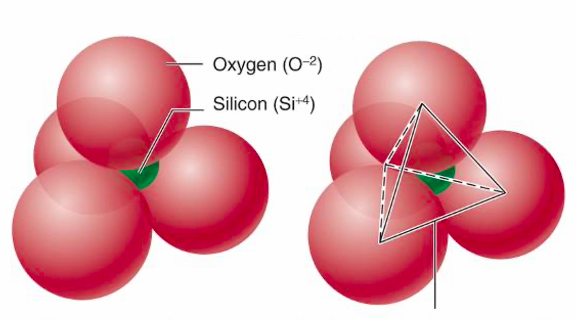
Silica tetrahedra
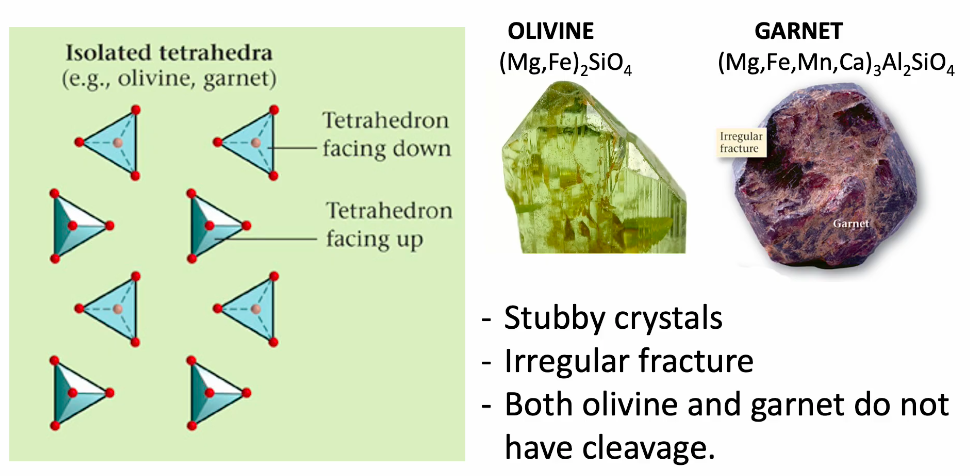
Isolated tetrahedra
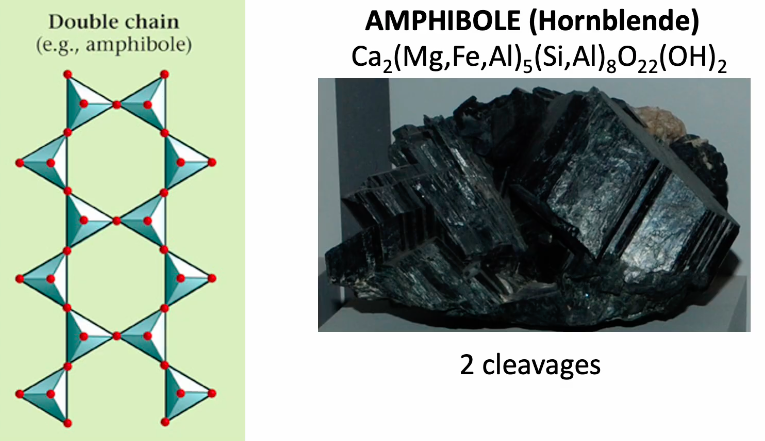
Single chain silicate

Double chain silicate
Sheet silicate

Framework or Network silicate
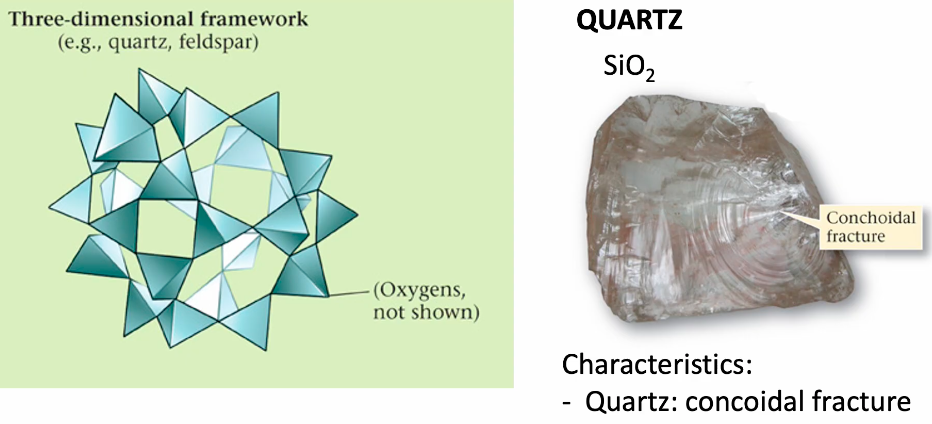
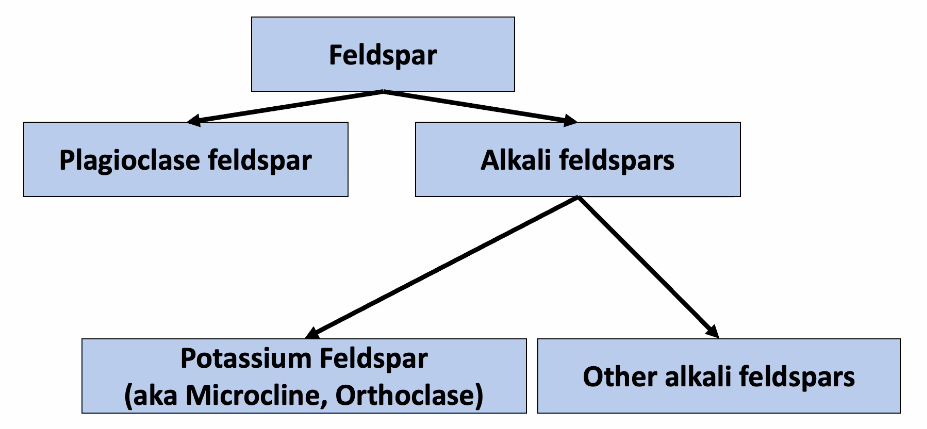
Framework or Network Silicate - Feldspars
PLAGIOCLASE FELDSPAR: NaAlSi3O8 to CaAl2Si2O8
POTASSIUM (K) FELDSPAR (aka Microcline Orthoclase) KAlSi3O8
Characteristics:
- Feldspar: 2 cleavages, striated appearance

Feldspar classification