Balancing Equations and Reactions Chemistry Study Guide
1/17
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
18 Terms
What are subscripts?
Subscripts are numbers written on the lower right to indicate more than one of an element.
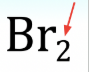
What are coefficients?
Coefficients are large numbers written in front of the entire chemical formula. Coefficients multiply every atom.

What are reactants?
Chemicals present at the beginning of the reaction.
What are products?
Chemicals created by the end of the reaction.
The law of Conversation of Mass states that the mass of the products must equal to…
The mass of the reactants.
Chemical equations are balanced when what quantity or quantities are the same?
The sum of masses of all substances involved.
The number of atoms of each kind.
What are a part of chemical formulas due to bonding and may not be changed?
Subscripts
What acts as multipliers of each substance and may be changed to balance an equation?
Coefficients
When counting certain atoms you…
Multiply Example: Answer-10

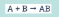
What reaction type is this?
Synthesis

What reaction type is this?
Decomposition

What reaction type is this?
Single Replacement

What reaction type is this?
Double Replacement

What reaction type is this?
Combustion
Which type of reaction requires oxygen as a reactant?
Combustion (Fire)
Which type of reaction results in the creation of one product when it is complete?
Synthesis
Which type of reaction involves a single substance being broken down into (or more) simpler chemicals?
Decomposition
What happens during a chemical reaction?
Atoms are rearranged.