All bio mols so far
1/197
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
198 Terms
what happens before a cell divides?
each chromosome makes an identical copy of itself so that the daughter cell will contain exactly the same DNA as the parent cell
how is a template formed for DNA replication?
the two strands of the double helix separate and each strand acts as a template for the formation of a new strand
what is semi conservative DNA replication?
when DNA replicates, 2 new molecules are produced, each consisting of one original strand and one new strand
what are the two new molecules like in semi conservative replication
the two new molecules are identical to each other and to the original DNA parent molecule
what separates the 2 strands of DNA by breaking the hydrogen bonds between the bases?
DNA helicase
what does the enzyme DNA helicase do?
it breaks the hydrogen bonds between the complimentary bases, separating the 2 strands of DNA
(unzips DNA)
What does the enzyme RNA primase do?
synthesises very short sections of RNA complementary to the relevant exposed bases
this acts as a starting point for the enzyme DNA polymerase
what does the enzyme DNA polymerase do?
joins up the sugar phosphate backbone
What happens during the joining stage of DNA replication?
Free nucleotides line up with the exposed bases on each strand, and the enzyme DNA polymerase joins the nucleotides together to form the new DNA strands
which way can DNA polymerase move along the DNA?
it can only move along the DNA in the 3’ to 5’ direction
what is the name of the strand which can be replicated continuously and which way does it go?
leading strand
5’ to 3’
what is the name of the strand which is replicated discontinuously and is what direction?
the lagging strand
3’ to 5’
what are the fragments called that DNA is replicated in on the lagging strand?
Okasaki fragments
what seals the Okasaki fragments together?
the enzyme DNA ligase
why is there one discontinuous/lagging strand in DNA replication?
the enzyme DNA polymerase can only work 3’ to 5’ and as the strands are antiparallel it means there is one continuous strand and the other is discontinuous as it works as it unwinds

how are mutations formed?
when random errors occur in DNA replication, it leads to a change in the sequence bases
what were the 3 alternative models for DNA replication that were suggested?
conservative
semi-conservative
dispersive
what is the conservative method of replication?
suggested that parental DNA strands remain together and newly formed daughter strands are also together
what is the dispersive method of replication?
suggested that after replication the 2 daughter DNAs had alternating segments of both parental and newly synthesised DNA interspersed on both strands
what were the scientists called who did an experiment to discover which DNA model of replication were correct - and what year?
Meselsohn and Stahl
1958
what was the aim of Meselson and Stahl’s experiment?
to determine which model of DNA replication is correct
what bacteria did Meselson and Stahl use?
E. coli
what was Meselson and Stahl’s method?
• Bacteria were grown in a medium containing heavy nitrogen (¹⁵N)
• DNA became labelled and heavy
• Bacteria were transferred to a medium containing light nitrogen (¹⁴N)
• DNA was extracted after each round of replication
• DNA was separated by centrifugation based on density
what were Meselson and Stahl’s results after one replication?
One intermediate band was observed
Conservative replication was ruled out because it would produce two bands (one heavy parental DNA and one light new DNA)
what were Meselson and Stahl’s results after 2 replications?
Two bands were observed: one light and one intermediate
Dispersive replication was ruled out because it would produce only one band, which would become progressively lighter after each replication, not two distinct bands
Results supported semi-conservative replication
This model predicts one hybrid molecule after first replication and a mixture of hybrid and light DNA after second replication
Each new DNA molecule contains one original strand and one newly synthesised strand
what were Meselson and Stahl’s conclusion?
• DNA replication is semi-conservative
• Each daughter DNA molecule contains one original strand and one newly synthesised strand
how does the structure of RNA differ from DNA?
the pentose is ribose not deoxyribose
there is no thymine; uracil is present instead and binds to adenine
it is single stranded not double
it is shorter than DNA with a lower molecular mass
what are 3 types of RNA?
ribosomal RNA
messenger RNA (mRNA)
transfer RNA (tRNA)
what is messenger RNA (mRNA)?
single polynucleotide strand formed in the nucleus during transcription, using a specific section of the DNA molecule (gene) as a template
carries a copy of the genetic info of the gene to the ribosomes in the cytoplasm
mRNA is used in translation to determine the sequence of amino acids in a protein (it’s primary structure)
what is transfer RNA (tRNA)?
tRNA molecule is a single strand folded into a clover leaf shape
different types of tRNA molecule in the cytoplasm, each with a binding site for the attachment of a specific amino acid
during protein synthesis each tRNA molecule carries its specific amino acid to a ribosome
a specific sequence of three bases on the molecule is known as an anticodon
what is central dogma?
describes the flow of genetic information from DNA to RNA to protein, outlining the processes of transcription and translation.
what is a gene?
a section of DNA that codes for a particular protein
what is a triplet code?
3 bases code for 1 amino acid (4^3 different combinations = 64) - for 20 amino acids
how does transcription occur?
DNA on the template strand is converted into a complementary sequence
how does translation occur?
the sequence of bases in mRNA determine the order of amino acids in the polypeptide chain
tRNA molecules bring the amino acids to the mRNA in the right order
where does transcription occur?
inside the nucleus
where does translation occur?
in the cytoplasm (attached to a ribosome)
what are nucleic acids?
large molecules which make up the genetic material of all living things
are nucleotides monomers or polymers?
polymers
what monomers make up nucleic acids?
nucleotides
what are 2 examples of nucleic acids?
DNA
RNA
what are 3 differences between DNA and RNA?
DNA is has a deoxyribose sugar, RNA has a ribose sugar
DNA has Thymine (T), RNA has Uracil (U)
DNA has 2 polynucleotide strands, RNA has 1 polynucleotide strand
what 3 parts is a nucleotide made up of?
pentose sugar (deoxyribose for DNA, ribose for RNA)
phosphate group
nitrogenous base
what is the structure of ribose?
5 carbon sugar (pentose) with the molecular formula C5H10O5, existing in both linear and ring forms

how are individual nucleotides formed?
condensation reactions between components
how are nucleotides linked to form polymers?
the phosphate of one nucleotide can join to the sugar of another
what bond is formed between 2 nucleotides?
a phosphodiester bond
what type of reaction is it when nucleotides join?
a condensation reaction
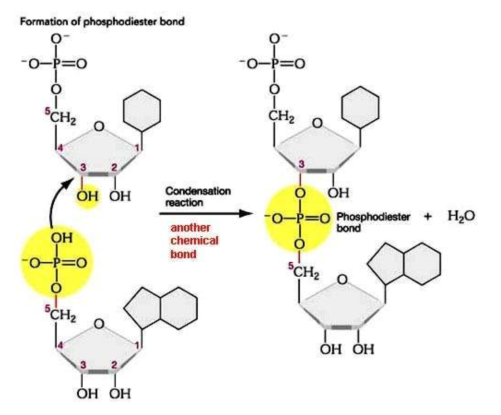
what is formed when many nucleotides join together?
polynucleotide chain or nucleic acid
they from a sugar-phosphate backbone, with the bases protruding to the side
which bases have a single carbon ring, and which bases have a double carbon ring in their molecular structure?
Cytosine and Thymine are single carbon ring
Guanine and Adenine are double carbon rings
which bases are complementary to each other?
A - T
C - G
what is the structure of DNA?
DNA forms a double helix
DNA is double stranded - made up of 2 strands of polynucleotides held together by H bonds between the bases
Each strand has a phosphate group (5’) at one end and a hydroxyl group (3’) at the other end. The two strands are arranged that they run in opposite directions - said to be antiparallel
what is the term to describe how the strands in DNA run in opposite directions to each other?
the strands are antiparallel
what does the term antiparallel mean in DNA?
the two strands in DNA run the opposite way to each other
is it 3’ or 5’ strand if the phosphate group is the one sticking out on the end?
5’
is it 3’ or 5’ strand if the sugar group is the one sticking out on the end?
3’
is RNA single or double stranded?
single
what are the 5 nitrogenous bases?
adenine
thymine (not present in RNA)
cytosine
guanine
uracil (only present in RNA)
what are the 2 groups the nitrogenous bases can be sorted into?
purines
pyrimidines
what are pyrimidines?
they contain a single carbon ring
what are purines?
they contain a double carbon ring
what is the difference between pyrimidines and purines?
pyrimidines have a single carbon ring whereas purines have a double carbon ring
what are some purines?
adenine
guanine
what are some pyrimidines?
thymine
cytosine
uracil
do purines always bond with another purine or a pyrimidine?
purines always bond with a pyrimidine
pyrimidines always bond with purines
this happens in their complimentary pairs
how many H bonds is there between adenine and thymine?
2 H bonds
how many H bonds are there between guanine and cytosine?
3 H bonds
what is ATP?
an energy carrying molecule released in respiration
it is a phosphorylated nucleotide - this means that it is a nucleotide with a phosphate group added
what is a phosphorylated nucleotide and what is an example of one?
a nucleotide with phosphate groups added
e.g. ATP
what does DNA stand for?
deoxyribonucleic acid
what is the method for extracting DNA and why do you do each step?
Mash up strawberries → breaks up cell walls/increase in surface area
Add extraction liquid including detergent, salt and protease
Then filter → removes insoluble structures, filtrate contains dissolved DNA
Add ice cold ethanol → precipitates out DNA strands and makes them visible
Properties of water
Polar
High boiling point
High specific heat capacity
Less dense in solid state
Cohesive properties
Adhesive properties
Not easily compressed
What does polar mean?
There is an uneven distribution of electrons in a molecule, resulting in an uneven charge across the molecule.
In a water molecule what are the slight charges on the oxygen and hydrogen atoms?
Oxygen = slightly negative
Hydrogen = slight positive
What is a hydrogen bond?
The weak interaction between a slightly negatively charged atom of a molecule and a slightly positively charged atom on another molecule.
How strong is a hydrogen bond?
Hydrogen bonds are relatively weak (weaker than a covalent bond)
Why is water a good solvent?
Water is polar, so any other polar molecule will dissolve very easily
Why is water a transport medium for living organisms?
It is an excellent solvent - minerals and ion dissolve very easily
Why does water have a high boiling point?
Contains many hydrogen bonds
Requires lots of energy to break the hydrogen bonds
Therefore is a good coolant
Why does water have a high specific heat capacity?
Many hydrogen bonds require lots of energy to overcome
Why is ice less dense than water?
When water is cooled below 4 degrees maximum number of hydrogen bonds form
Bonds fix molecule in an open lattice structure
Molecules are held further apart than in the liquid state
Why does water have adhesive and cohesive properties?
It is polar
Why is it important for life that water has a high specific heat capacity?
Temperature remains fairly constant - provides a stable environment (in cells and for aquatic organisms)
Why is it important for life that water has a high boiling point?
It is a good coolant
Why is it important for life that water is more dense than ice?
Ice floats
Water below is insulated
Aquatic animals do not freeze and currents can move
What does water’s cohesive properties create?
Surface tension
Why is cohesion necessary for water to act as a transport medium?
Water is attracted to other polar surfaces
Allow for capillary action
eg. in xylem of plants
what is chromatography?
a method of separating components of a solution based on differences in solubility
what phases does chromatography use?
a stationary phase (paper or thin gel)
a mobile phase (a solvent that moves up the stationary phase, carrying the components of a solution with it)
what is amino acid chromatography?
a mixture if amino acids can be separated by chromatography
to visualise the amino acids on the paper, the chromatogram needs to be sprayed with Ninhydrin
how do we use Rf values to identify amino acids?
the distance moved by each amino acid relative to teg distance moved by the solvent is called the rf value
different amino acids have specific rf values, so this method can be used to separate and identify amino acids
What elements are carbohydrates made up of?
Carbon
Hydrogen
Oxygen
Examples of monosaccharides
Glucose
Fructose
Galactose
Ribose
Deoxyribose
Disaccharides
Lactose
Maltose
Sucrose
Which monosaccharides is lactose made up of?
Glucose + galactose
Which monosaccharides is maltose made up of?
Glucose + glucose
Which monosaccharides is sucrose made up of?
Glucose + fructose
Polysaccharides
Starch
Glycogen
Cellulose
What is starch made of?
Alpha glucose molecules
What is glycogen made of?
Alpha glucose molecules