Introduction to alcohols
1/6
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
7 Terms
What is the general formula for alcohols
CnH2n+1OH
Why are alcohols polar
They contain hydrogen bonds, which form due to electronegativity difference in oxygen and hydrogen
Why are alcohols soluble in water
Alcohols are more soluble in water due to their -OH can form hydrogen bonds with water molecules
What is a primary alcohol
Where the carbon which the -OH group is attached to is attached to one other carbon
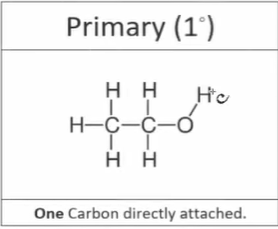
What is a secondary alcohol
Where the carbon which the -OH group is attached to is attached to two other carbons
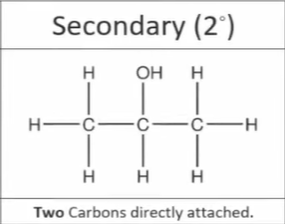
What is a tertiary alcohol
Where the carbon which the -OH group is attached to is attached to two other carbons. Tertiary alcohols are always branched

Why are tertiary alcohols the most stable
They have an alkyl group on the OH group. They have no hydrogen bond to the carbon.