Group 3: Boron and Aluminium
1/7
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No study sessions yet.
8 Terms
Boron and Aluminum are Group 3 elements. What does this mean?
They have 3 electrons in their outer shell.
How many covalent bonds can a boron and aluminum atom form?
3 covalent bonds- this gives a total of 6 electrons in the outer shell of the Group 3 atom.
Compounds of boron and aluminum are often described as ‘electron-deficient.’ What does this mean?
A species that has less than 8 electrons in its outer shell.
How may an electron-deficient boron/aluminum atom gain a stable octet?
Aluminum and boron are able to form co-ordinate bonds with a lone pair of electrons from another atom to gain a stable octet.
Therefore, they are called electron acceptors and can form dimers.
What is a dimer?
Formed when 2 of the same molecules join together.
What is a co-ordinate bond?
A covalent bond in which both of the bonding electrons come from the same species.
It is also known as a dative bond.
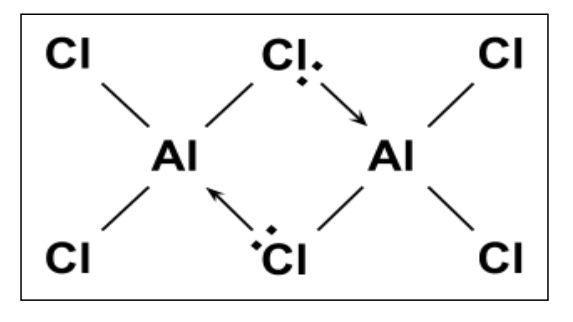
How is aluminum hexachloride, Al2Cl6 formed?
The aluminum in aluminum chloride is electron deficient.
Therefore, two monomers of aluminum chloride, AlCl3, are able to form a dimer of Al2Cl6. They are linked together by two co-ordinate bonds.
Two chlorine atoms share their lone pair of electrons with two atoms of aluminum to form the co-ordinate bonds.
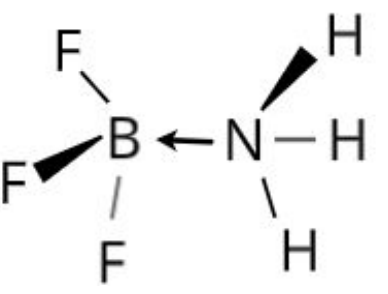
What is a donor acceptor compound and give one example of this type of compound.
Donor acceptor compounds form where a lone pair is donated by one compound and accepted by another, forming a co-ordinate bond.
An example of this is boron trifluoride accepting a lone pair from the nitrogen in ammonia to form NH3BF3.