BIO 81 Exam 1 Study Guide
1/192
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
193 Terms
What are the criteria for being a living organism?
complexity and organization at different levels
highly regulated
abilities to respond to the environment
grow and develop
metabolize
reproduce
capacity to evolve
Compare and contrast a hypothesis vs. a theory and give an example of each.
Hypothesis - a potential explanation made from an observation, can be supported or falsified by data, and a good hypothesis makes predictions that can be tested
ex: "If a student studies with music, then their test scores will decrease." (This is a prediction that can be tested through a controlled experiment).
Theory - a general explanation of a natural phenomenon supported by a body of experiments
ex: The Theory of Evolution by Natural Selection (This is a widely accepted, extensively verified explanation for the diversity of life on Earth)
Explain the purpose and steps of the Scientific Method.
The Scientific Method is a systematic, logical approach used by scientists to discover how things work and to solve problems.
Its primary purpose is to minimize human bias and ensure that results are reliable, reproducible, and based on objective evidence rather than intuition or guesswork.
Steps:
make observations
ask a question
formulate a hypothesis
make predictions
design experiment
collect and interpret data
draw conclusions
possibly peer review and publish the study
What are the first and second laws of thermodynamics, and how do they apply to living organisms?
First law of thermodynamics - Energy can be neither created nor destroyed; it can only be transformed from one form to another
Living organisms transform energy from the environment into chemical energy that cells can utilize. They give off heat as a by-product.
Second law of thermodynamics - The degree of entropy (disorder) in the universe tends to increase
Living organisms are highly organized and need a lot of energy to remain that way. The cell gives off energy as heat, causing small molecules in the environment to move more. This increase in movement increases the entropy (disorder) of the entries system.
What does it mean to say that a cell is life’s functional unit?
A cell is the simplest entity that can exist as an independent unit of life. While many organisms are multicellular, life can take on a single-celled form as well. All cells have a discrete boundary that separates their interior from the external environment, and they also have the ability to harness materials and energy from that environment.
How does the central dogma help us to understand how mutations in DNA can result in disease?
DNA directs the formation of proteins that do the cell’s work. Mutations in DNA can be transcribed and translated into proteins with altered structure and, therefore, different functions. Changes in protein function can cause the cell to work improperly, or fail althogether, resulting in some cases in disease. In some cases, however, altered proteins improve the function of the cell.
What was Stanley Miller’s experiment, and what is its significance in understanding the origins of life?
Experiment: Stanley Miller’s experiment was designed to simulate Earth’s early atmosphere. He began by heating liquid water to produce water vapor. This water vapor then mixed with other gases thought to be present on early Earth, including methane, ammonia, and hydrogen gas. To simulate lightning, Miller passes a spark through the mixture of gases. The gas mixture was then cooled, allowing liquid water to form again and dissolve any molecules that were synthesized. These were collected at the bottom of the apparatus and obtained through a sampling valve for analysis.
Results: As the experiment proceeded, reddish material accumulated at the bottom of the apparatus. Analysis showed that this material included a number of amino acids, the building blocks that make up proteins, which are the key structural and functional molecules that do much of the work of the cell.
What are atoms made up of? Describe each component.
Protons - positively charged particles, in the nucleus
Neutrons - neutral particles, in the nucleus
Electrons - negatively charged particles, orbit around the nucleus in regions called orbitals
What are isotopes?
Atoms of the same element that have different number of neutrons
Describe the 4 different types of bonds, and give examples of each.
Polar covalent - shared pair of electrons between atoms with very different electronegativities
ex: water
Non-polar covalent - shared pair of electrons between atoms with similar electronegativities
ex: hydrocarbons, gases (O2, H2, and N2)
Ionic - two ions with opposite charges associate with each other due to their difference in electronegativity
ex: electrolytes such as K+, Na+, and Cl-
Hydrogen - a weak attraction between a partial positive hydrogen and a partial negative electronegative atom
ex: base pairing in DNA, alpha-helices and beta-sheets in proteins, cohesive properties of water
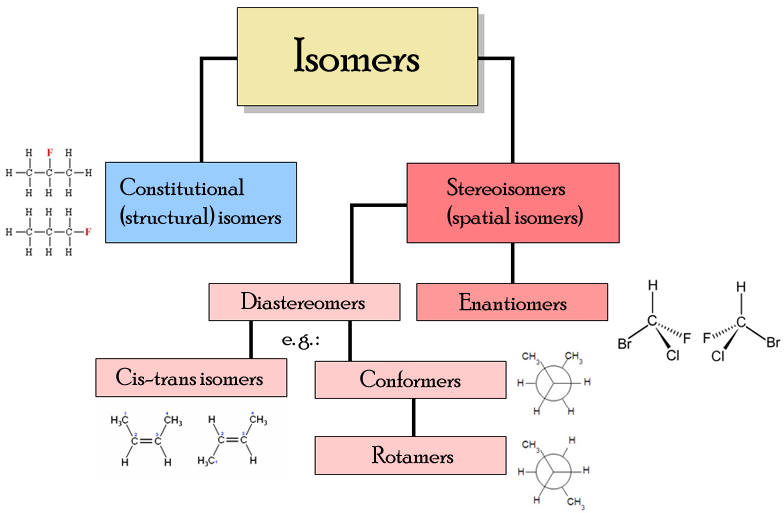
What are the differences between structural isomers, Cis-trans isomers, and Enantiomers?
Structural isomers - Atoms are connected in a completely different order (e.g., branching vs. straight chain)
Cis-trans isomers - Geometric isSame connectivity, but rigid bonds (like \(C=C\)) prevent rotation, forcing groups to be on the same side (cis) or opposite sides (trans)
Enantiomers - Non-superimposable mirror images, usually involving a chiral center (carbon with four different groups)
Why do salts dissolve in water?
The partial positive charge on H+ associates with the negative charge on the Cl-.
The partial negative on the O- associates with the positive charge on the Na+.
What are the four emergent properties of water that contribute to Earth’s suitability and explain why those properties do so?
Cohesive behavior - due to hydrogen bonding, water molecules stick together (cohesion) and to other substances (adhesion). This allows for capillary action, enabling plants to transport water and nutrients against gravity from roots to leaves.
Ability to moderate temperature - water has a high specific heat, meaning it resists changes in temperature by absorbing or releasing large amounts of heat with little change in its own temperature. This stabilizes ocean temperatures for marine life and keeps terrestrial climates moderate.
Expansion upon freezing - water is less dense as a solid (ice) than as a liquid. Ice floats, insulating the liquid water below, which prevents oceans and lakes from freezing solid and allows aquatic life to survive winter.
Versatility as a solvent - due to its polarity, water is the "universal solvent" capable of dissolving many substances. This allows water to act as a transport medium for nutrients in biological fluids (blood, sap) and facilitates chemical reactions necessary for metabolism.
What would be the effect on the water molecule if oxygen and hydrogen had equal electronegativity?
The molecules would become nonpolar, losing its charge separation and the ability to form hydrogen bonds.
water would lose properties like cohesion, surface tension, and its ability to act as a solvent
would make it unsuitable for life
Why does water make a great solvent?
Its polarity and ability to form hydrogen bonds.
The water molecule (H2O) has a partial negative charge near the oxygen atom and a partial positive charge near the hydrogen atoms, allowing it to attract, break apart, and surround charged or polar substances like salts and sugars.
What is cohesion? Give an example.
The tendency of molecules of the same type to stick together (due to hydrogen bonds)
ex: surface tension, water transport in plants
What is adhesion? Give an example.
The tendency of two different molecules to stick together (due to hydrogen bonds)
ex: water transport in plants (water molecules adhere to plant wall)
How does water regulate temperature?
Hydrogen bonds make water resist changes in temperature
high specific heat capacity and high heat of vaporization
Why is table salt iodized?
Some trace elements are required to prevent diseases. A lack of iodine causes the thyroid gland to swell (this is cqalled goiter)
What is pH and how is it measured?
pH is a measure of the concentration of protons in a solution
Low H+ concentration means basic
High H+ concentration means acidic
What are the four most common elements in organic molecules?
Carbon, oxygen, hydrogen, and nitrogen
What features of carbon allow it to form diverse structures?
Behaves as if it has 4 unpaired electrons
Carbon atoms can bond with other carbon atoms to form large carbon chains that branch or form rings
Can form double bonds (limits the flexibility of the molecules and their structural options)
What are hydrocarbons? What are some of their properties?
a molecule composed entirely of carbon and hydrogen atoms
are nonpolar/hydrophobic
can exist in many forms (e.g., linear chains, rings, branched chains)
are the primary components of fats and fossil fuels
What are the four major classes of biological macromolecules and what are their functions?
carbohydrates (sugars + starches)
provide a source of energy and make up the cell wall in bacteria, plant, and algae cells
proteins (structural molecules, enzymes)
act as catalysts to facilitate chemical reactions and also provide structural support of the cell
nucleic acids (DNA, RNA)
encode and transmit genetic information
lipids (fats, oils, waxes)
store energy, act as signaling olecules, and make up the membranes of the cell
How is diversity achieved in polymers? Use proteins as an example.
Through numerous combinations of subunits
proteins for example, are a polymer of amino acid subunits
any combination of subunits can be made, each resulting in a different protein
in this way, polymers are capable of displaying virtually limitless diversity
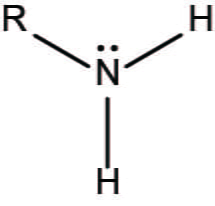
What is the amino group, its properties, and the macromolecule(s) it’s found in?
-NH2
properties:
polar, positively charged at the pH of the cell, behaves as a base, hydrophilic
commonly found in:
amino acids, proteins
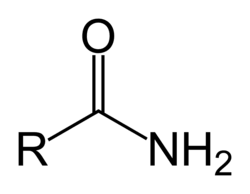
What is the amide group, its properties, and the macromolecule(s) it’s found in?
-C(=O)NH-
properties:
polar, hydrophilic
commonly found in:
proteins
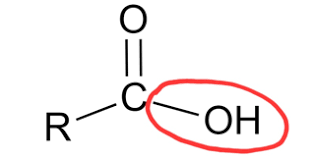
What is the carboxyl group, its properties, and the macromolecule(s) it’s found in?
-COOH
properties:
polar, negatively charged at the pH of a cell, behaves as an acid, hydrophilic
commonly found in:
fatty acids, amino acids, proteins
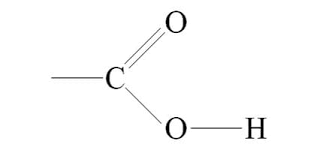
What is the carbonyl group, its properties, and the macromolecule(s) it’s found in?
>C=O
properties:
polar, hydrophilic
commonly found in:
carbohydrates, proteins
What is the hydroxyl group, its properties, and the macromolecule(s) it’s found in?
-OH
properties:
polar, hydrophilic
commonly found in:
carbohydrates, proteins, nucleic acids
What is the sulfhydryl group, its properties, and the macromolecule(s) it’s found in?
-SH
properties:
polar, forms S-S disulfide bonds
commonly found in:
the amino acid cysteine, proteins
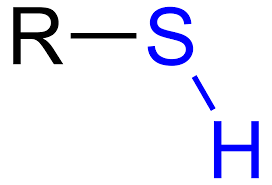
What is the methyl group, its properties, and the macromolecule(s) it’s found in?
-CH3
properties:
nonpolar
commonly found in:
amino acids, proteins, nucleic acids
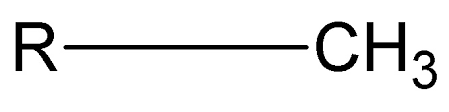
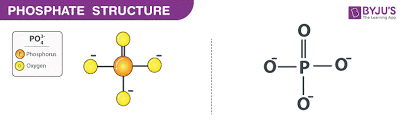
What is the phosphate group, its properties, and the macromolecule(s) it’s found in?
-OPO3H2
properties:
polar, negatively charged at the pH of a cell, hydrophilic
commonly found in:
phospholipids, nucleic acids, ATP
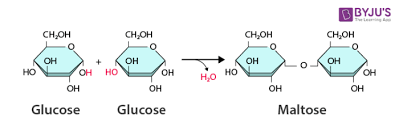
What is a dehydration synthesis reaction? Anabolic or catabolic? Give an example.
builds polymers by releasing a water molecule (anabolic)
ex: formation of maltose (glucose + glucose)
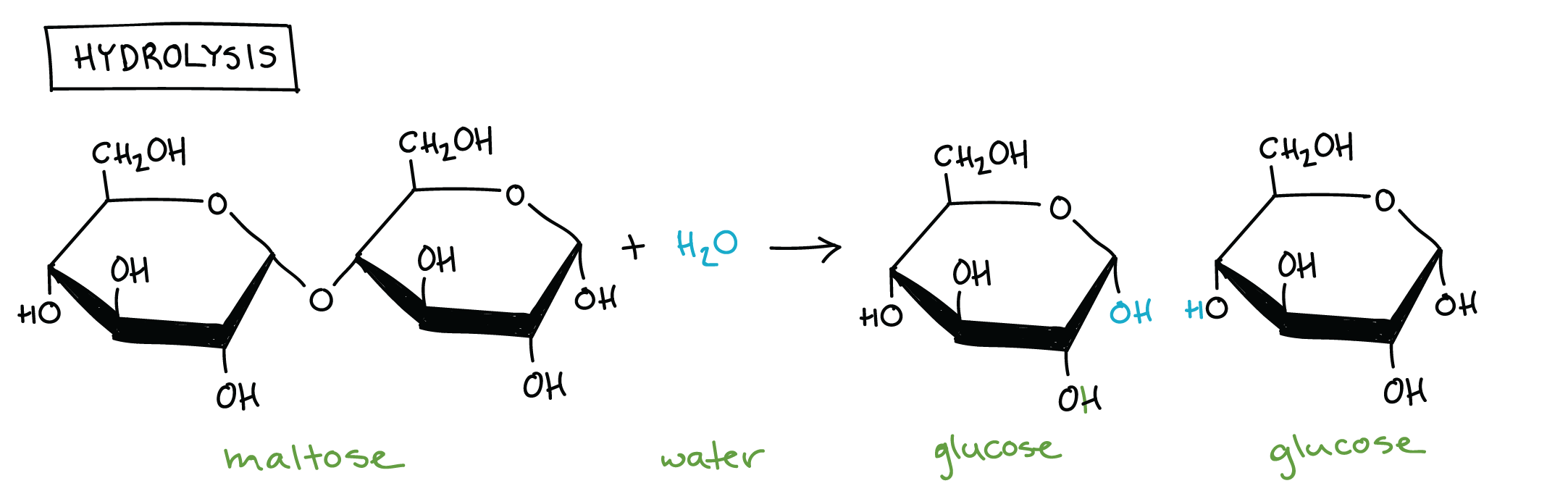
What is a hydrolysis reaction? Anabolic or catabolic? Give an example.
breaks polymers into monomers by adding a water molecule (catabolic)
ex: breaking down maltose (glucose + glucose)
What are saccharides, monosaccharides, disaccharides, and polysaccharides? Give examples of each.
Saccharides - the simplest carbohydrate molecule (sugar)
Monosaccharides - a simple sugar; the monomers that make up large carbohydrates
ex: ribose, glucose, fructose
Disaccharides - a double sugar
ex: lactose in milk (glucose + galactose), maltose (glucose + glucose), sucrose (glucose + fructose)
Polysaccharides - a polymer of simple sugars; provide long-term energy storage or structural support
ex: starch, cellulose, chitin, glycogen
What is the bond that attaches one monosaccharide to another?
Glycosidic bond
covalent
formed between carbon 1 of one monosaccharide and a hydroxyl (-OH) in a different monosaccharide molecule
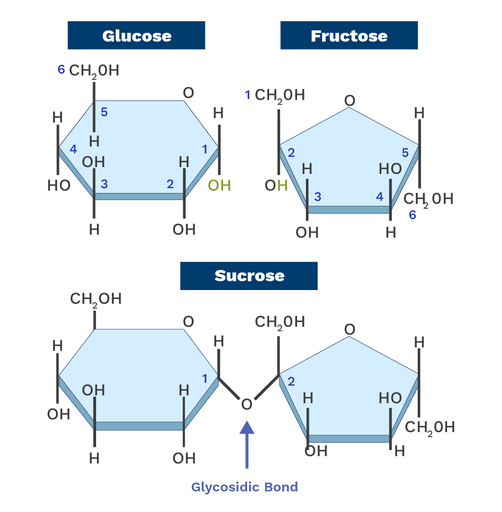
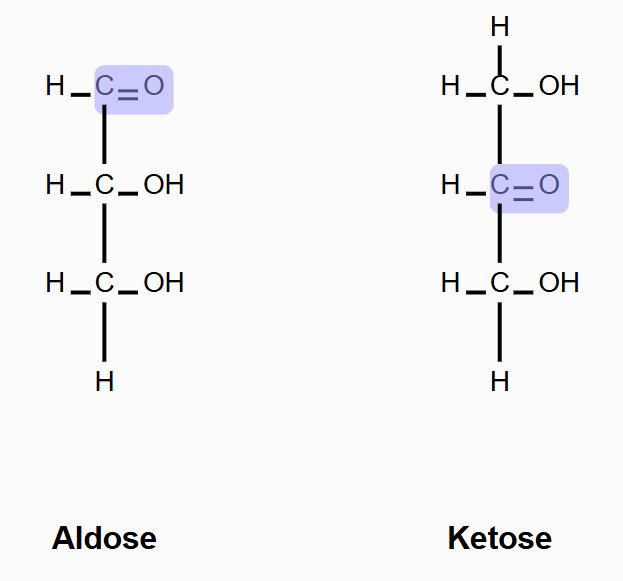
What are the two classifications of monosaccharides based on the location of the carbonyl group and number of carbons?
Aldose - contains an aldehyde group (-CHO) at the end of the carbon chain
Ketose - contains a ketone group (C=O) typically at the second carbon
What are the four classifications of polysaccharides? Which are for storage and which for support?
starch (storage)
glycogen (storage)
cellulose (support)
chitin (support)
What is starch? Found in plants or animals?
A storage polysaccharide of plants and consists entirely of glucose monomers
The simplest form of starch is amylase
What is glycogen? Found in plants or animals?
A storage polysaccharide in animals
Vertebrates store glycogen mainly in liver + muscle cells
What is celulose?
A major component of the tough wall of plant cells, a polymer of glucose w/ different glycosidic linkages
major source of insoluable fiber
What is chitin?
Found in the exoskeleton of arthropods, also provides structural support for cell walls of fungi
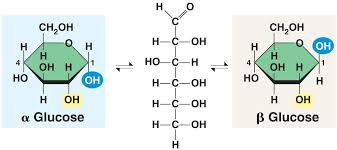
What are alpha and beta glucose ring structures? What are their oritentations?
Alpha and beta glucose are cyclic isomers of glucose that differ only in the orientation of the hydroxyl (-OH) group on the first carbon (C1)
alpha are helical polymers, are more reactive and easily broken down by enzymes
beta are straight polymers, less reactive due to structure, and require different enzymes for breakdown
What are lipids, their types, and their properties?
fatty, oily, waxy compounds that are soluble in organic solvents and insoluble in polar solvents such as water
types: triglycerides, phospholipids, sterols
properties:
most hydrophobic
mostly made of hydrocarbons
What are triglycerides?
composed of 1 glycerol molecule and 3 fatty acid molecules
assembled via dehydration synthesis
What is the difference between saturated and unsaturated fats?
Saurated
no double bonds
maximum # of hydrogens attached to each carbon
solid at room temperature
primarily animal fats
Unsaturated
has one or more double bonds
fewer than a maximum of 3 hydrogens
liquid at room temperature
plant-based oils, nuts, seeds, fish
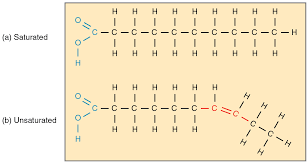
How are saturated fats solid at room temperature while unsaturated fats are liquid?
Saturated fats are solid at room temperature because their straight molecular chains pack tightly together, allowing for strong intermolecular attractions.
have greater van der Waals forces cause they’re straight and can pack together tightly
Conversely, unsaturated fats remain liquid because double bonds create "kinks" in their chains, preventing dense packing and keeping the molecules fluid.
What are Van der Waals Forces and how do they relate to fatty acids?
the attraction of temporarily polarized molecules that is formed from opposite charges
the hydrocarbon chains in fatty acids have nonpolar covalent bonds
the longer the hydrocarbon tial, the greater teh strength of van der Waals forces
What is hydrogenation?
converts unsaturated fats to saturated fats
creates trans fats, a type of unsaturated fat that is even less healthy than saturated fats
What are steroids and their properties? Give examples.
The precursor molecule for cholesterol and steroid hormones
composed of a carbon skeleton with 4 rings and varying functional groups
ex: cholesterol, synthetic anabolic steroids, estrogen, testosterone
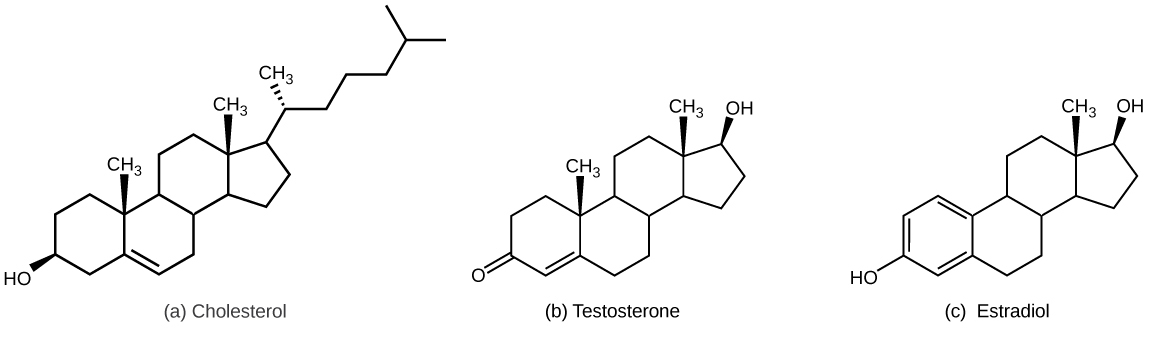
What is cholesterol and its functions?
a sterol
component of membrane
the “base steroid” from which your body produces other steroids
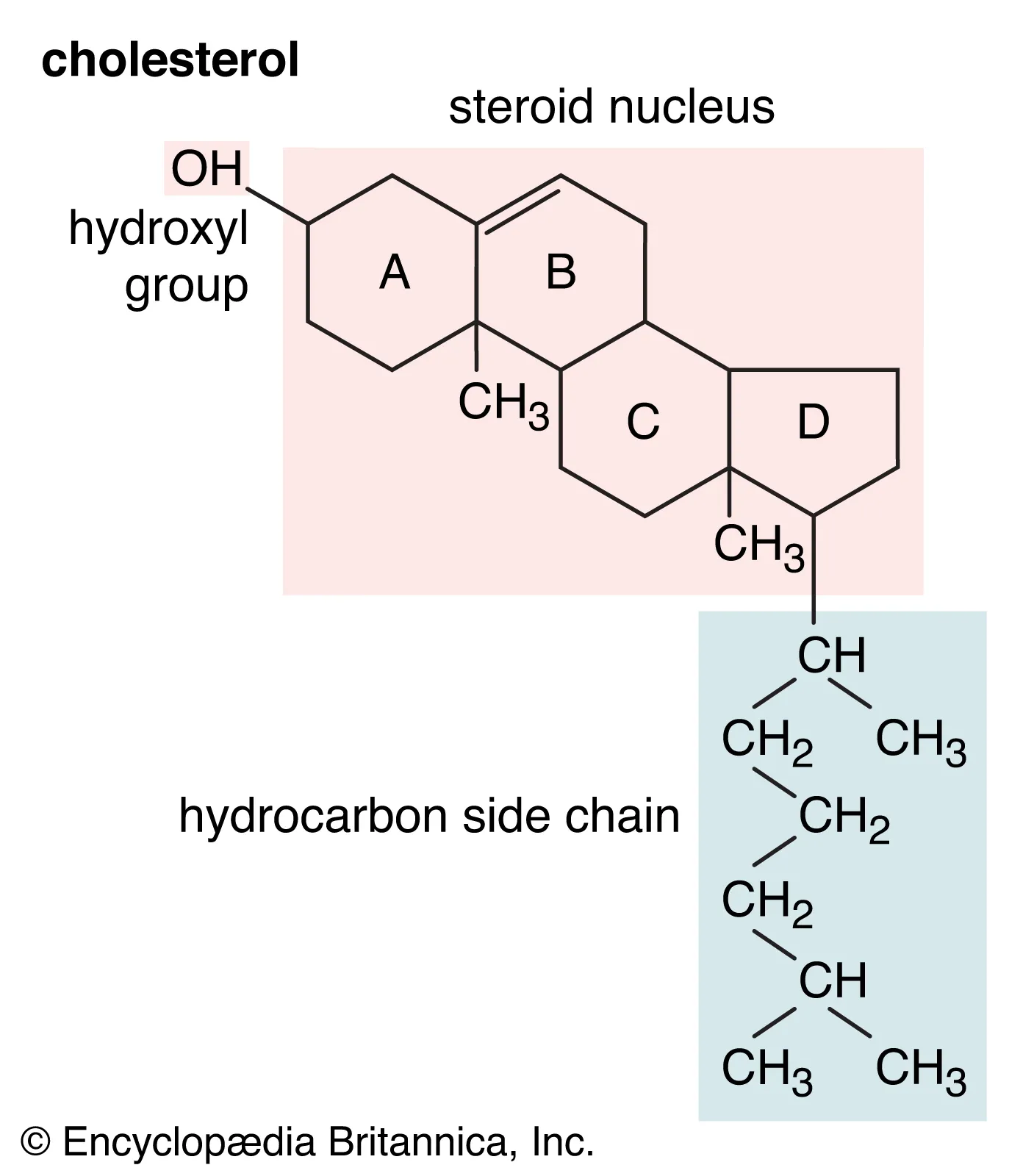
What are phospholipids and their functions?
a major component of all cell membranes
amphipathic
structure:
one glycerol molecule
two fatty acids
one phosphate
one choline molecule
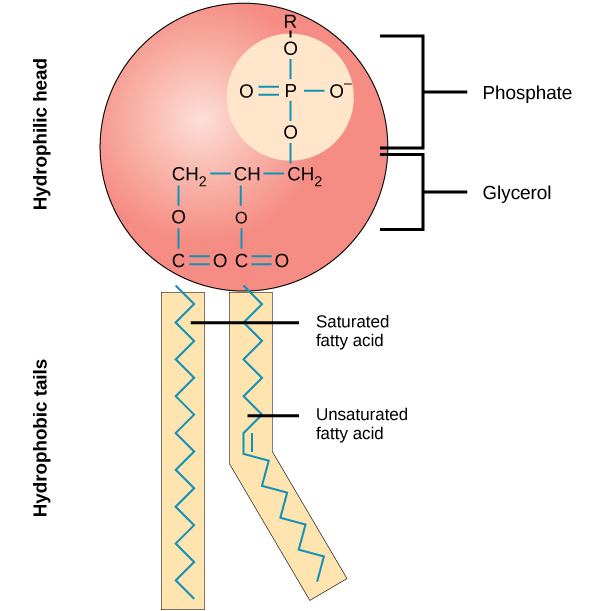
What are proteins and their functions? Give examples.
the key structural and functional molecule that does the work of the cell, providing structural support and catalyzing chemical reactions
functions:
structural support, storage, transport, cellular communications, movement, defense against foreign substances
examples:
transport proteins, enzymatic proteins, contractile and motor proteins, cell membrane proteins
Describe the structure of a protein.
apha carbon (central C)
animo group (-NH2)
carboxyl group (-COOH)
hydrogen atom
R group (side chain)
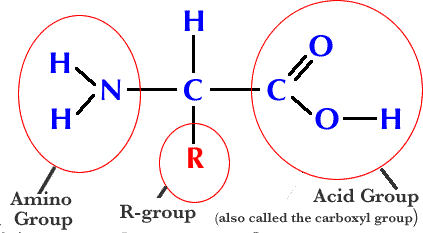
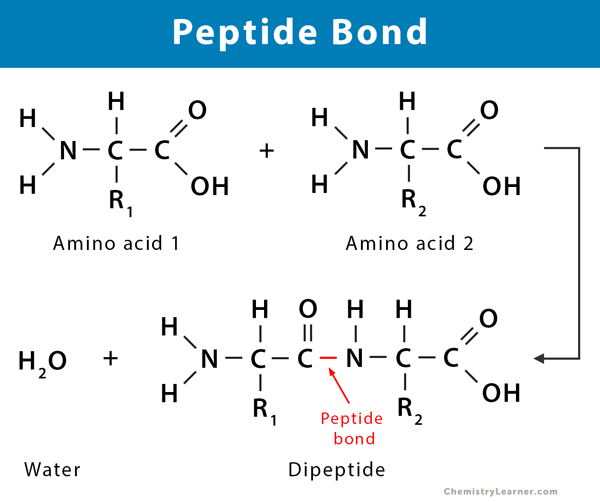
What is the type of bond that connects amino acids together to form a protein?
peptide bond
covalent bond
alpha carboxyl chain attaches to the next alpha-amino region
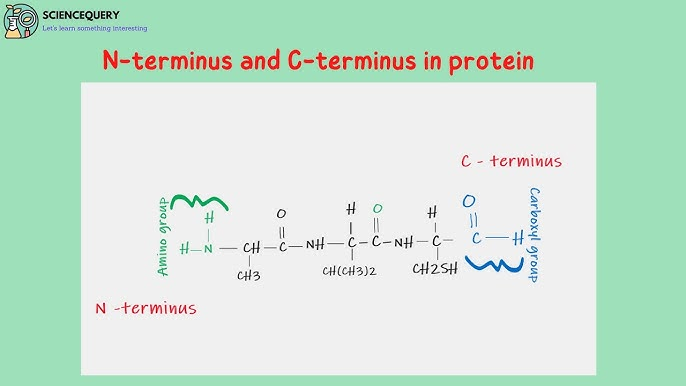
What are the two ends of a protein?
amino end (N-terminus)
carboxyl end (C-terminus)
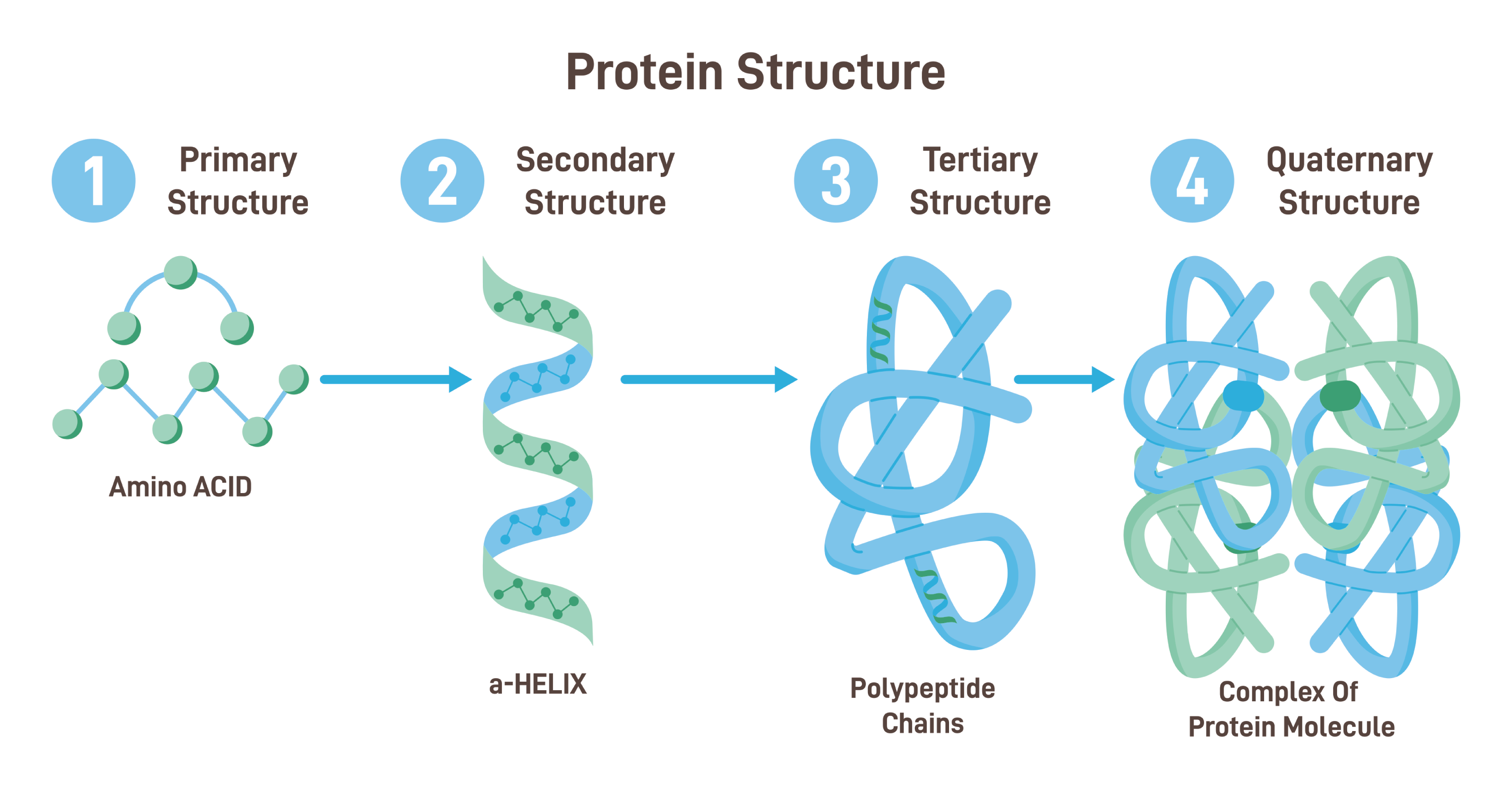
Describe the four levels of structure of a protein (include the bonds at each level).
primary structure
sequence of amino acids in a protein
held together by covalent peptide bonds
secondary structure
results from repeating hydrogen bonding interactions between atoms of the polypeptide backbone
hydrogen bonds form between the amino hydrogen and carboxyl oxygen in the peptide backbone
alpha-helix structure:
polypeptide backbone is twisted in a right-hand coil
beta sheet structure:
polypeptide chain folds back and forth on itself, forming a pleated sheet
tertiary structure
determined by interactions between R groups, rather than interactions between backbone constituents
interaction between R groups: hydrogen bonds, ionic bonds, hydrophobic interactions, and van der Waals interactions
the 3D shape enables the protein to carry out its specific function in a cell
has disulfide bridges (a strong covalent bond between the sulfur atoms of two cysteine amino acid residues within a protein)
quaternary structure
results when several polypeptide chains form one macromolecule
held together by hydrogen bonding and London dispersion forces
ex: collagen and hemoglobin
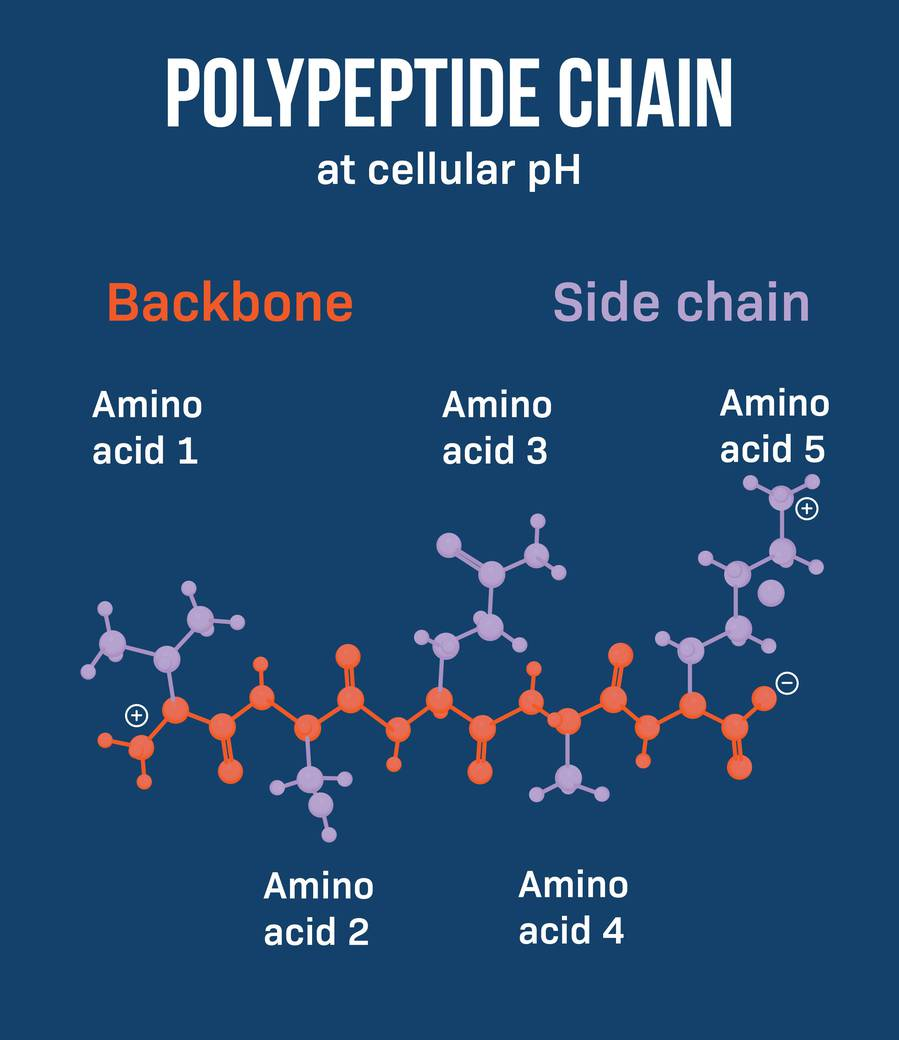
What is a protein or peptide backbone?
The protein backbone, also known as the peptide backbone, is the fundamental structural framework of proteins. It consists of a continuous chain of amino acid residues linked together by peptide bonds, forming the primary structure of a protein molecule.
The protein backbone is composed of a repeating pattern of nitrogen, carbon, and oxygen atoms, with the side chains of the amino acids protruding from the backbone.
What is a disulfide bridge?
a strong covalent bond between the sulfur atoms of two cysteine amino acid residues within a protein
these bonds play a crucial role in stabilizing the tertiary and quaternary structures of proteins, holding them in their functional 3D shapes
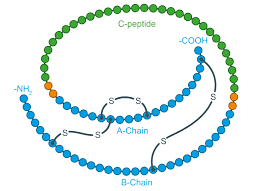
How can proteins become denatured? What is denaturation of a protein?
definition: the unfolding of proteins by chemical treatment or high temperatures
the separation of paired, complementary strands of nucleic acids
a denatured protein is biologically inactive
can become denatured through alterations in pH, salt concentration, temperature, or other environmental factors
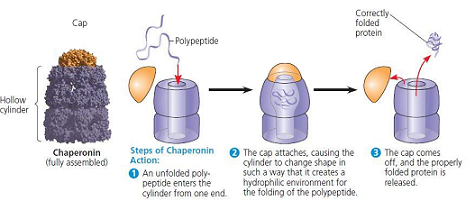
What are chaperonins?
a protein that shield a slow-folding protein ntil it can attain proper three-dimensional structure
What are nucleic acids and their properties? Give some examples.
a polymer of nucleotides that encodes and transmits genetic information
nucleic acids determine the primary structure of each protein in a cell
roles:
DNA and RNA
the monomers of nucleic acids are nucleotides
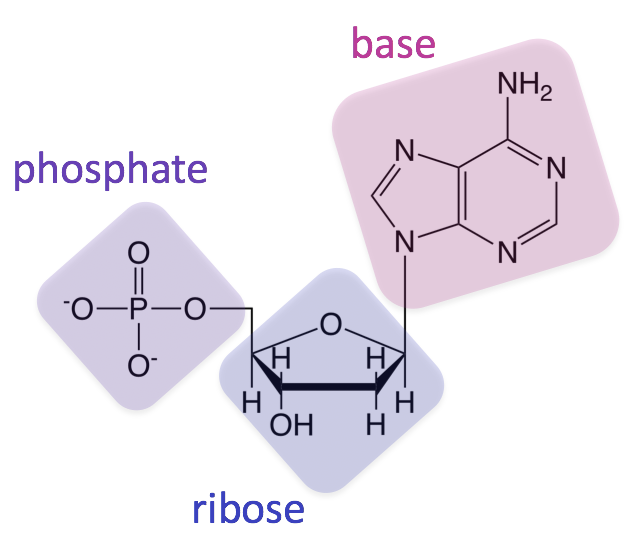
What are nucleotides and their two classifications?
monomers of nucleic acids
composition:
one phosphate group (or more)
one pentose sugar (deoxyribose or ribose)
one nitrogenous base
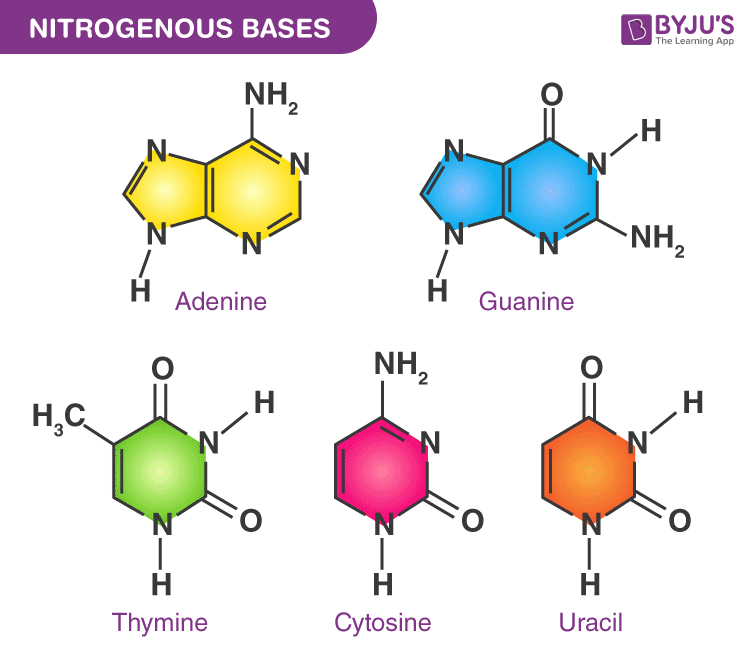
pyrimidine bases (have a single ring)
cytosine, thymine, uracil
CUT the Pie (CUT = Pyrimidines)
purine bases (have a double ring)
guanine, adenine
Pure As Gold (AG = Purines)
What is the bond that forms the sugar-phosphate backbone of DNA and RNA?
phosphodiester bond
forms when a phosphate group in one nucleotide is covalently joined to the sugar unit in another molecule
ocurs via dehydration reaction
links 5’ phosphate to 3/ hydroxyl to form covalent bonds
forms a sugar-phosphate backbone
How does base pairing stabilize the double helix of DNA?
the arrangement of atoms allows precise hydrogen bonding
Define a cell.
the smallest unit of life that can function independently
every living thing is made up of one or more cells
biochemical processes take place inside cells to carry out basic functions of life
What is cell theory?
Cell theory states:
All organisms are made of cells
The cell is the fundamental unit of life
Cells come from preexisting cells
What are the common features of all cells?
They all have genetic material, ribosomes, cytoplasm, and cell membrane
Compare prokaryote and eukaryote cells.
Prokaryotes (Archaea & Bacteria)
single-celled, no nucleus, no membrane-bound organelles, reproduce asexually, the most ancient form of life
Archaea: more similar to eukaryotes than bacteria, can survive in extreme environments
Bacteria: cell walls containing peptidoglycan, found in nearly every environment
Eukaryotes (Eukarya)
has a nucleus, has membrane-bound organelles, larger, more complex
Compare plant and animal cells.
In animal cells, but not in plant cells:
lysosomes
centrioles
flagella (in some plant sperm)
In plant cells, but not in animal cells:
cell wall
chloroplasts
central vacuole
Similarities:
bound by a membrane
contains membrane-bound organelles
How do metabolic requirements set upper limits on the size of a cell?
surface-to-volume ratio
as a cell grows, its volume increases faster than its surface area
Small cells have a high surface area relative to volume, enabling efficient nutrient uptake and waste disposal.
What are vesicles and how do they work?
small membrane-enclosed sac that transports substances within the cell
how vesicles work:
bud off an organelle, taking a piece of the membrane and internal contents of the organelle from which they derive
they then fuse with another organelle or the cell membrane, reforming a continuous membrane and unloading their contents
exocytosis
a vesicle from the cytoplam fuses with the cell membrane and empties its contents into the extracellular space or delivers protein to the cell membrane
endocytosis
a vesicle buds off from the cell membrane, bringing material from outside the cell into that vesicle
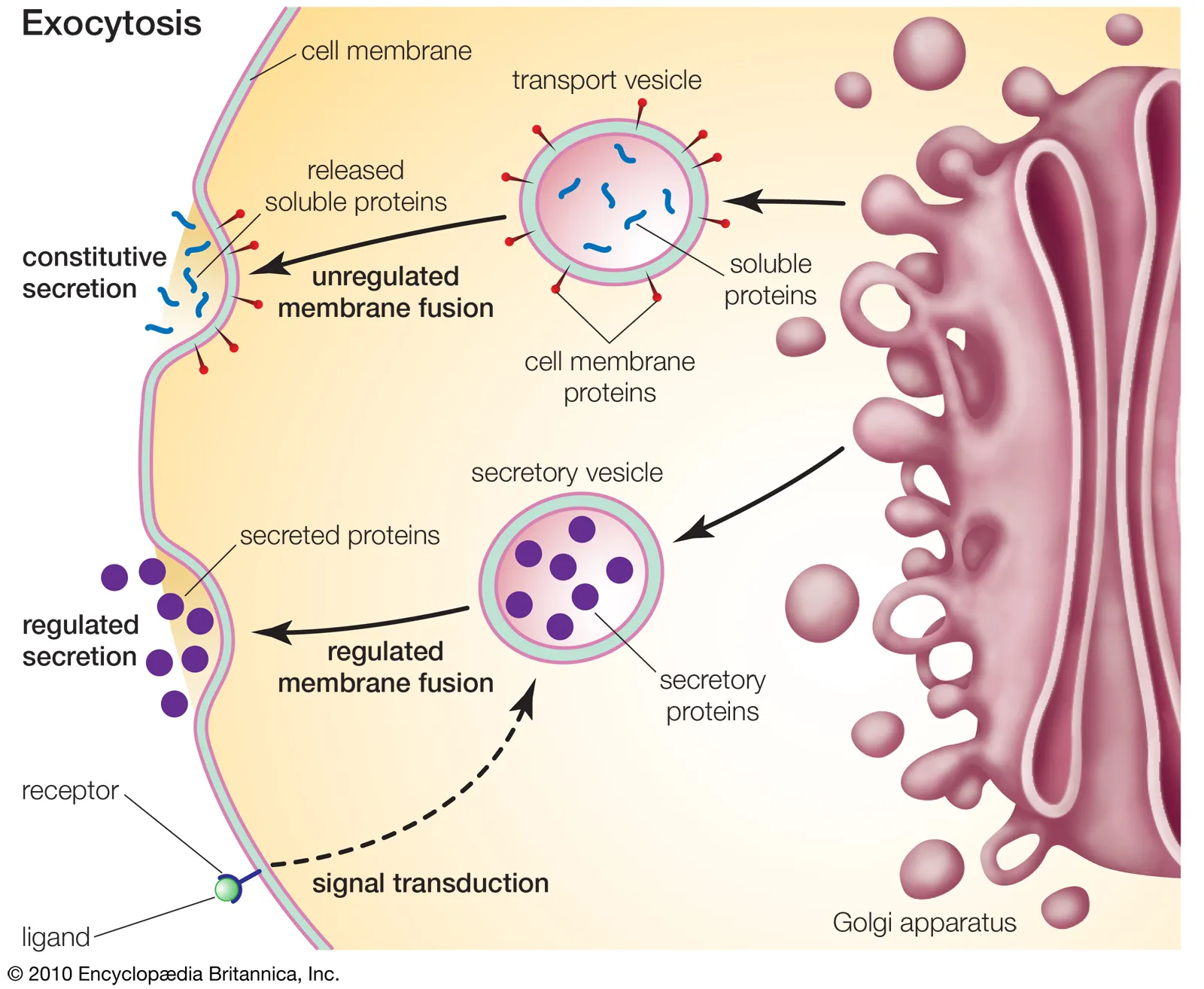
What is the nucleus, its structures, contents, and functions?
contains most of the cell’s genes and is usually the most conspicuous organelle
site of transcription (DNA → RNA)
RNA is exported from the nucleus
produces ribosomes necessary for protein synthesis
coordinates the replication of DNA during cell division
Nuclear Envelope - encloses the nucleus, separating it from the cytoplasm
is a double-membrane; each membrane has a lipid bilayer
Nuclear Pores - protein channels in the nuclear envelope that allow molecules to move into/out of the nucleus
essential for the nucleus communicating w/ the rest of the cell
(mRNA out & ribosomal proteins in/out)
Nuclear Lamina - a meshwork of intermediate filaments (lamins) and proteins lining the inner nuclear envelope
Describe DNA and RNA in the nucleus.
DNA
organized into discrete units called chromosomes
each chromosome is composed of a single DNA molecule associated w/ proteins
chromatin - a complex of DNA and proteins that gives chromosomes their structure
condenses to form discrete chromosomes as a cell prepares for division
RNA
nucleolus - a distinct, dense, non-membrane-bound structure within the nucleus that contains the genes and transcripts for ribosomal RNA
RNA is synthesized in the nucleus
mRNA leaves the nucleus through a nuclear pore
binds to a ribosome so protein synthesis can start
What is the endomembrane system?
an interconnected network of lipid bilayer membranes within eukaryotic cells that works together to modify, package, and transport lipids and proteins
It facilitates cellular traffic and compartmentalization but excludes mitochondria and chloroplasts
Key components include the nuclear envelope, endoplasmic reticulum (ER), Golgi apparatus, lysosomes, vesicles, and the plasma membrane
What organelles are in the endomembrane system and which are not?
IN Endomembrane System
nuclear envelope
endoplasmic reticulum (ER)
Golgi apparatus
lysosomes
vesicles and vacuoles
plasma membrane
NOT in Endomembrane System
*These operate independently and do not exchange vesicles
mitochondria (Responsible for ATP production)
chloroplasts (Found in plants for photosynthesis)
peroxisomes (Metabolic compartments that break down fatty acids, which do not receive vesicles from the Golgi)
nucleolus (Located inside the nucleus but distinct from the membrane system)
What are the names and function of the major organelles of the endomembrane system in eukaryotic cells?
Nucleus - encloses the cell’s genetic information and is the site of transcription
Endoplasmic Reticulum (ER) - an organelle in which proteins and lipids are synthesized
Golgi apparatus - modifies proteins and lipids produced by the ER and acts as a sorting station as those molecules move to their final destinations
Lysosomes - contain enzymes that break down macromolecules
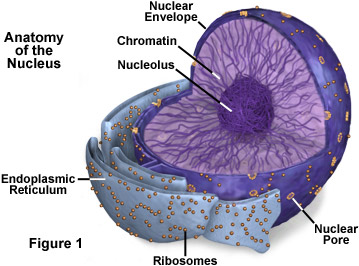
What are ribosomes and their functions?
Particles made of ribosomal RNA and protein that synthesize proteins
Note: they are not membrane-bound organelles
They carry out protein synthesis in two locations:
in the cytosol (free ribosomes)
on the outside of the ER or nuclear envelope (bound ribosomes)
Consists of a small subunit (reads mRNA) and a large subunit (joins amino acid)
What are antibiotics and how do they work?
Antibiotics are powerful medications that treat bacterial infections by either killing bacteria directly
They work by targeting specific structures or processes in bacteria—such as cell wall formation, protein synthesis, or DNA replication—that human cells do not have, or inhibiting their growth and reproduction
Many antibiotics bind to bacterial ribosomes or cell walls
Penicillin prevents bacteria from building cell walls, causing them to burst
Some antibiotics interfere with the machinery bacteria use to create essential proteins, stopping them from multiplying
Specific drugs break down the bacterial cell membrane, causing essential components to leak out
Other antibiotics prevent bacteria from copying their DNA, stopping reproduction.
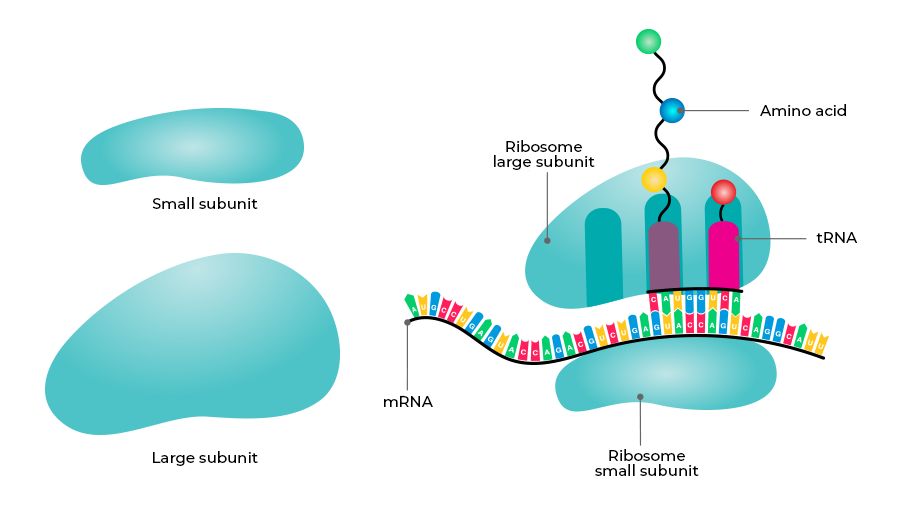
What is the endoplasmic reticulum and its functions?
An organelle composed of a network of membranes that is involved in the synthesis of proteins and lipids
Structure:
has a complex network of interconnected tubules and flattened sacs
cisternae - flattened, sac-like, membrane-bound compartments
lumen - fluid-filled inner compartment inside the cisternae
Rough ER
has attached ribosomes that secrete glycoproteins
primarily involved in protein synthesis and modification
distributes transport vesicles
all cells have at least some rough ER for the production of transmembrane and organelle proteins
Smooth ER
lacks ribosomes
extension of rough ER
important for lipid and steroid synthesis
large proportion in specialized cells
involved in metabolic processes like lipid synthesis, carbohydrate metabolism, and detoxification of drugs and poisons
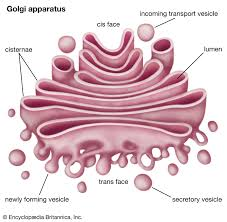
What is the Golgi apparatus and its functions?
An organelle that modifies proteins and lipids produced by the ER and acts as a sorting station as they move to their final destination
Structure:
flattened membranous sacs
cisternae - flattened sacs that are the internal space of the Golgi
cis face - receiving side (closest to ER)
trans face - shipping side (closest to plasma membrane)
Different regions of the Golgi have different enzymes that catalyze specific reactions
Functions:
Modifies proteins and lipids received from the ER
Packages/sorts proteins and lipids into vesicles for transport
Synthesizes carbohydrates that’ll be added to proteins and lipids
The edges of cisternae bud off as vesicles carrying proteins or modified sugars to the cell membrane
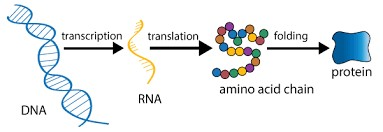
Describe the protein production pathway.
Transcription
In the nucleus, RNA polymerase converts a DNA gene sequence into messenger RNA (mRNA), which is then processed.
Export
The mature mRNA molecule exits the nucleus via nuclear pores to the cytoplasm.
Translation
Ribosomes read the mRNA, and tRNA brings amino acids to build the polypeptide chain.
Protein Folding & Modification
The ribosome moves to the endoplasmic reticulum and “reads” the mRNA instructions
Protein moves to the Golgi complex for additional processing and sorting
Transport & Secretion
Protein moves to the plasma membrane to be secreted
What are lysosomes and their functions?
A vesicle derived from the Golgi apparatus that contains hydrolytic enzymes that break down macromolecules
These macromolecules are either damaged or unneeded
Maintains a pH of 5
Digestion through phagocytosis:
forms a food vacuole
autophagy - Use enzymes to recycle the cell’s own organelles and macromolecules
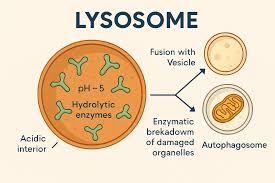
How do lysosomes illustrate the importance of compartmentalizing certain reactions in different spaces in the cell?
They have an acidic environment (pH of about 5), and this environment works well for degrading macromolecules, which is one of the functions of lysosomes.
These reactions must be confined to lysosomes because many of the cell’s proteins required for cellular function would unfold or degrade at such a low pH.
What are some genetic disorders associated with lysosomal disorders?
Tay Sachs Disease - A neurodegenerative condition resulting in physical and mental deterioration
missing an enzyme in lysosomes that breaks down lipids
Result: lipids accumulate in the brain, which leads to mental and physical deterioration
Niemann-Pick Disease - Causes lung, liver, and spleen dysfunction, alongside neurological symptoms
also causes a lipid accumulation, but in the spleen, liver, lungs, bone marrow, and brain
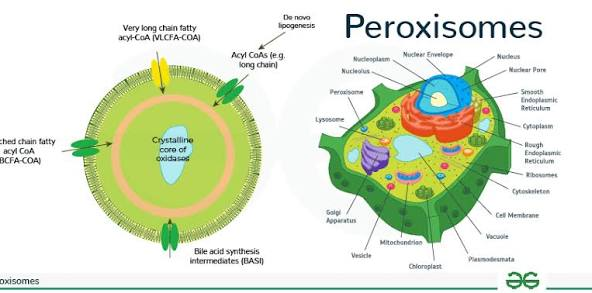
What are peroxisomes and their functions?
Self-replicating oxidative organelles
aid in digestion
originate at the ER
contain enzymes that digest and then oxidize certain toxic molecules
break down fatty acids
produce cholesterol and some lipids
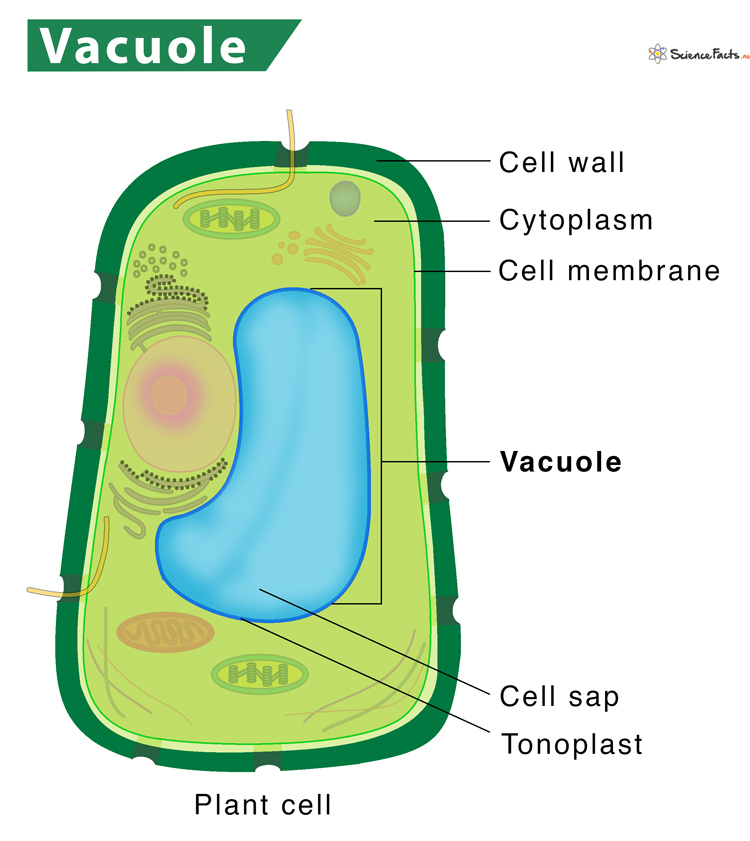
What are vacuoles and their functions?
Membrane-bound, fluid-filled organelle found in the cytoplasm of plant, fungal, and some animal and bacterial cells
Structure:
space within a cell, enclosed by a membrane
Function:
storage (holds water, ions, nutrients, proteins, pigments)
turgor pressure for structural support in plants
Why are mitochondria and chloroplasts are not considered in the endomembrane system
They are self-replicating organelles with their own DNA and double-membrane structures, likely originating from engulfed prokaryotes (endosymbiotic theory).
Unlike the endomembrane system (ER, Golgi, vesicles), they function independently and are not part of the cell's membrane trafficking, protein synthesis, or transport network.
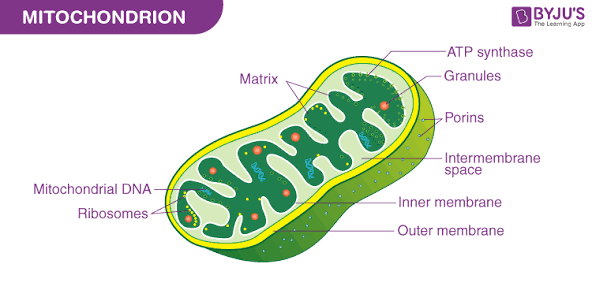
What are mitochondria, their structure, and their functions?
Harness energy from chemical compounds (such as sugars) and turns it into ATP
found in almost all eukaryotic cells
grow and reproduce inside the cell
contain their own DNA and ribosomes
Structure:
has a double membrane
Outer membrane: a highly permeable membrane containing proteins called porins that allow the movement of ions and small molecules
Inner membrane: less permeable and highly folded into structures called cristae
Cristae increase the surface area for biochemical reactions
Inner membrane space: the space between the outer and inner membranes, which is critical for creating a proton radient for ATP production
Matrix: fluid-filled inner compartment containing enzymes for the citric acid cycle, mitochondrial DNA, and ribosomes
Function:
Energy production (cellular respiration)
Metabolic signaling and synthesis (involved in the synthesis of heme groups (for hemoglobin) and steroids, & the production of reactive oxygen species (ROS) for signaling)
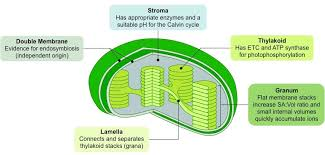
What are chloroplasts, their structure, and their functions?
Organelles that convert energy from sunlight into energy stored in sugar molecules
contain their own DNA and ribosomes
sugar made in chloroplasts travels to the mitochondria, which extracts the energy to use in cellular process
Structure:
Double membrane envelope
Stroma: space inside the chloroplast, surrounding the tylakoids
Thylakoid system: a collection of membranous sacs containing chlorophyll
Grana: tightly packed stacks of thylakoids, designed to maximize light absorption
Lumen: space inside the thylakoid membranes
Function:
Photosynthesis (converts light energy, CO2, and water into glucose and oxygen)
Produces glucose that can then be used to produce ATP for energy
Are mitochondria present in animal cells, plant cells, or both?
Both
What are four similarities between mitochondria and chloroplasts?
not part of the endomembrane system
have a double membrane
have proteins made by free ribosomes
contain their own DNA
Describe the endosymbiont theory, discuss the evidence.
proposes that eukaryotic organelles—specifically mitochondria and chloroplasts—originated as free-living prokaryotes (bacteria) that were engulfed by a larger host cell
The theory suggests that an anaerobic host cell engulfed an aerobic proteobacterium, which evolved into the mitochondrion, giving rise to all eukaryotes. Later, one of these aerobic, nucleated cells engulfed a photosynthetic cyanobacterium, which evolved into the chloroplast, giving rise to plants and algae.
Evidence:
Mitochondria and chloroplasts contain their own circular DNA, similar to bacterial chromosomes
These organelles have their own ribosomes (70S) that closely resemble bacterial ribosomes in size and structure, rather than the 80S ribosomes found in the surrounding eukaryotic cytoplasm.
Mitochondria and chloroplasts reproduce independently within the cell through a process similar to binary fission, the same method used by bacteria.
Both organelles are surrounded by two membranes—an inner membrane belonging to the original bacterium and an outer membrane derived from the host cell's vesicle.
They are roughly the same size as prokaryotic cells.
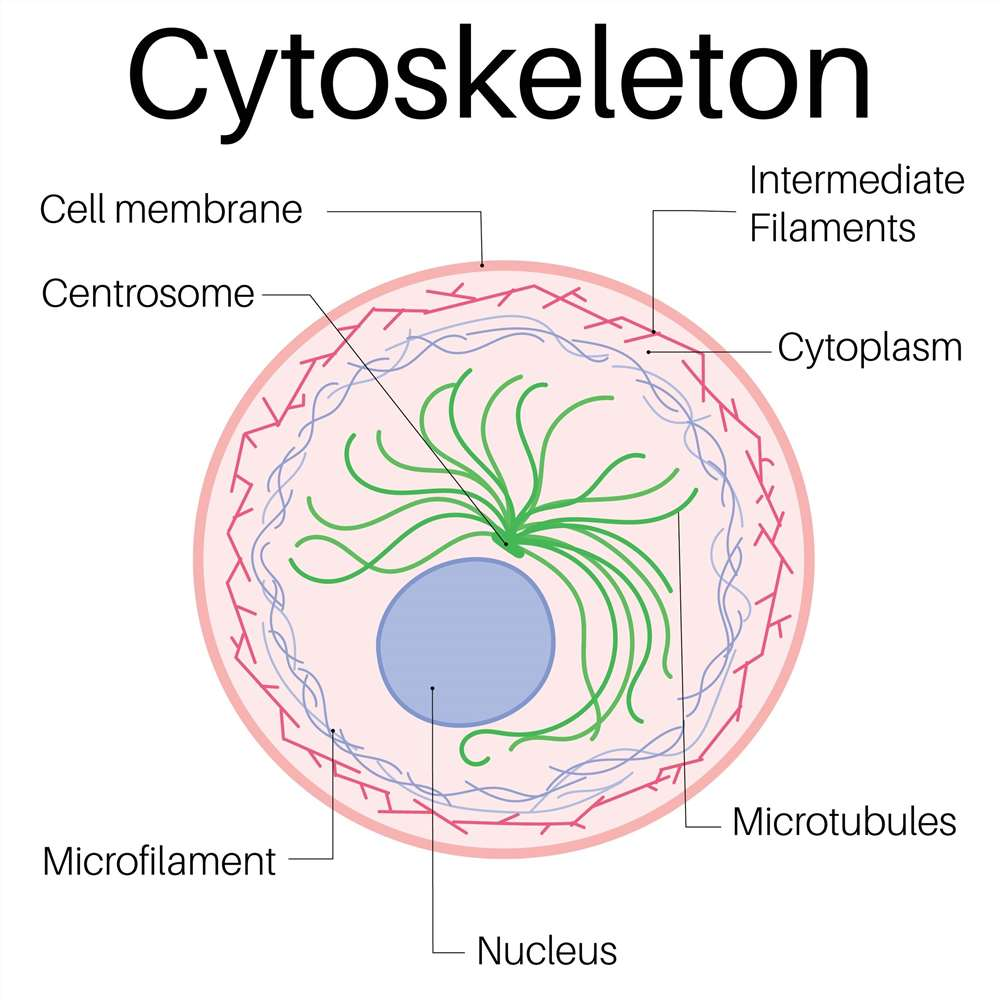
What is the cytoskeleton, what is it composed of, and what are the functions?
The cytoskeleton provides a structural framework for the cell
Extends through the cytoplasm
Organizes the cell’s structures and activities
Anchors organelles
Structural support
Cell movement
Aids in digestion
Organelle transport
Composed of 3 molecular structures:
microfilaments
microtubules
intermediate filaments
What is polymerization and depolymerization?
Polymerization is the chemical process of linking small molecules (monomers) together to form long, complex chain molecules called polymers
exothermic (releases heat)
synthesis of new material
Depolymerization is the reverse process, breaking down polymers back into their constituent monomer units or oligomers, often used in chemical recycling to recover raw materials
endothermic(requires energy input)
recovery of raw materials
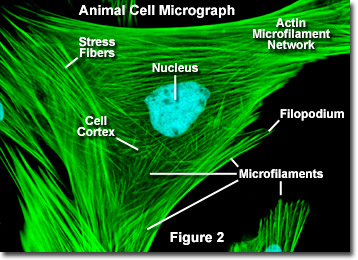
What are microfilaments, their structure, function, and importance in the cytoskeleton?
Microfilaments (also called actin filaments) are the thinnest components of the eukaryotic cytoskeleton that are dynamic, polar structures that enable cell motility, maintain cell shape, facilitate muscle contraction, and divide cells during cytokinesis
Structure:
Size: the thinnest and shortest (abt 7nm in diameter)
Composition: made of globular protein subunits called G-actin (globular actin)
Arrangement: G-actin monomers polymerize to form long, flexible, helical polymers called F-actin (filamentous actin)
Helix Structure: two parallel F-actin strands twist around each other
twisted double helix of actin subunits
Polarity: they have distinct plus (+) and minus (-) ends
allows them to grow (polymerize) and shrink (depolymerize) rapidly, often utilizing ATP
Function:
movement and muscle contraction
Cell shape and structure:
form a 3D network called the cortex
area of cytoplasm just beneath the cell membrane
bundles of microfilaments make up the core of microvilli of intestinal cells
Cell movement:
Enable movement by lengthening and shortening (amoeboid, pseudopod extension, cytoplasmic streaming)
Muscle contraction:
Working with motor protein myosin, they slide past each other to cause muscle fiber contraction
Cell division:
Form the contractile ring (cleavage furrow) that pinches an animal cell into two during cytokinesis
Intracellular transport:
Act as tracks for myosin-based movement of organelles and vesicles within the cytoplasm.
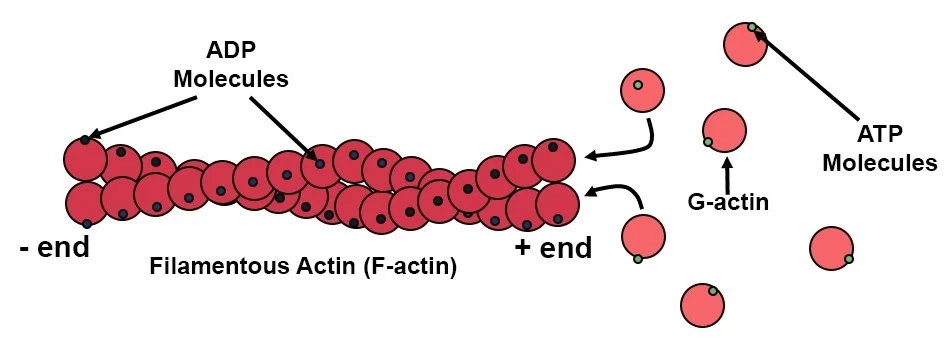
What are actin and myosin? What are the purposes/functions?
The primary motor proteins that are responsible for muscle contraction and cell motility
Actin - globular proteins that link together to form long, spiralling chains
thin and smooth
serve as the “tracks” along which myosin travels
Role: acts as the track along which myosin moves during contraction
Additional functions: part of the cytoskeleton, maintains cell shape, aids in endocytosis, facilitates cell motility
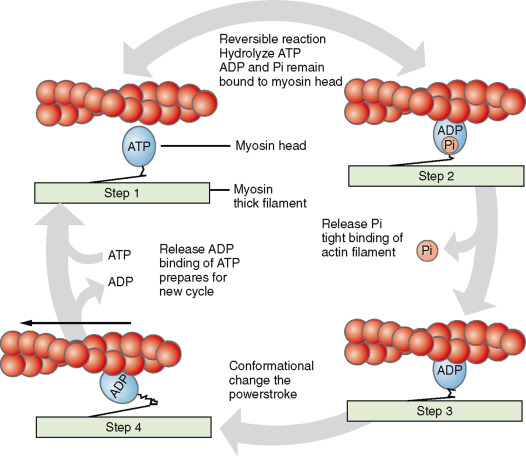
Myosin - motor proteins that convert ATP to mechanical force
thick and rough
acts like a motor, using its heads to “walk” along actin filaments
Role: convert chemical energy (ATP hydrolysis) into mechanical energy
Interactions:
The myosin head binds to actin, forming cross-bridges to pull the actin filaments, known as the sliding filament mechanism
Play a role in:
muscle contraction
cellular mobility
intracellular transport
cell division
What makes up the core of microvilli of intestinal cells?
Bundles of microfilaments
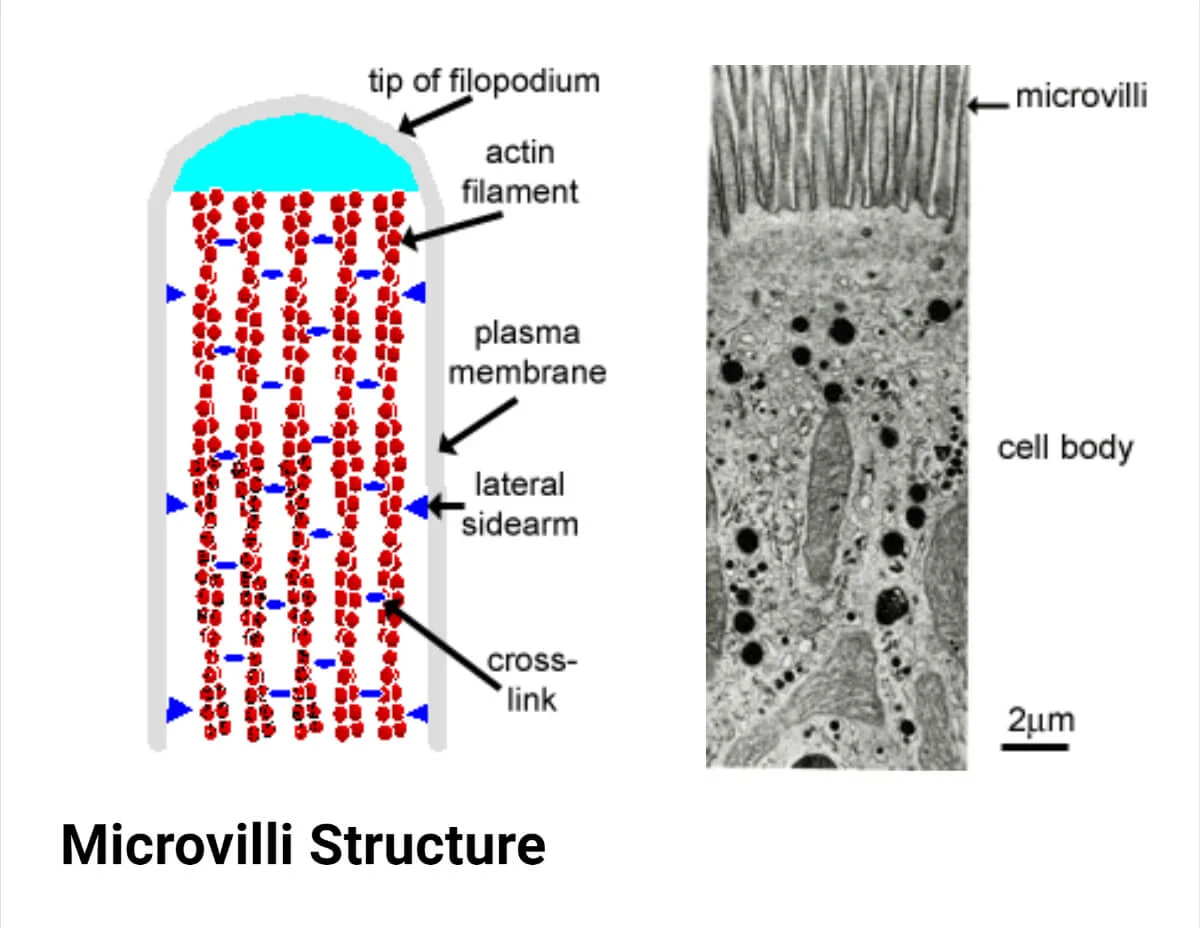
What is the cortex in a cell?
The cell cortex (or actin cortex) is a specialized, thin network of actin filaments, myosin motors, and actin-binding proteins located directly beneath the plasma membrane of most animal cells. It acts as a structural scaffold to maintain cell shape, regulate surface tension, and facilitate movement, division, and structural integrity.
It acts as a mechanical shell that regulates cell shape, surface tension, and rigidity while enabling cell division (cytokinesis) and migration.