Cell Bio Unit 3 Study Guide
1/86
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
87 Terms
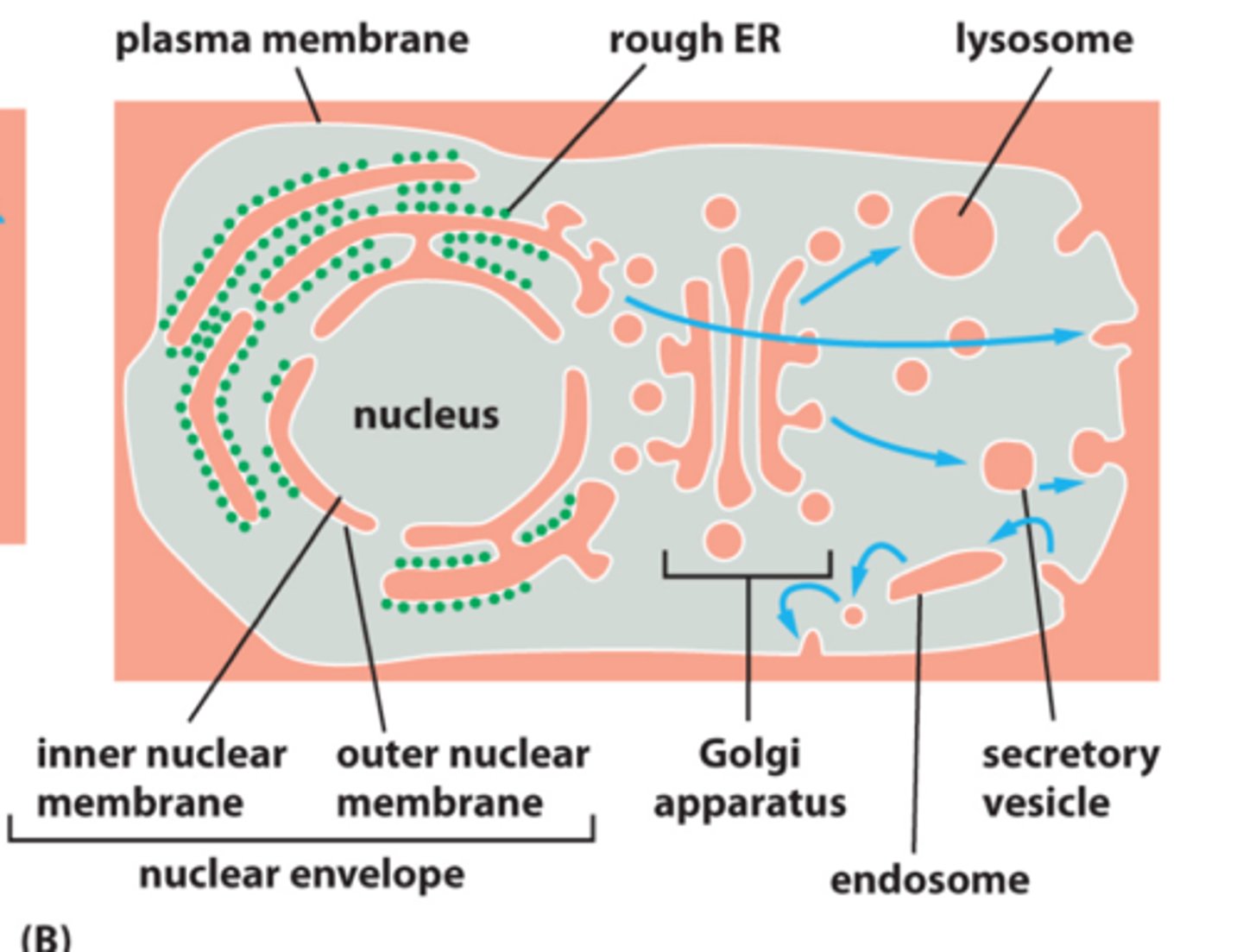
What are the basic functions of organelles listed in lecture?
Nucleus: DNA and RNA synthesis
Ribosomes: protein synthesis
Cytoplasm: synthesis and degradation
ER: lipid synthesis, protein synthesis
Golgi: protein modification and sorting
Endosome: endocytose material pass through before reaching lysosome (sorting and recycling)
Lysosome: intracellular digestion
Mitochondria: aerobic respiration
Peroxisome: oxidative reactions, breakdown of long fatty acid chains
What are the 3 mechanisms for sorting proteins to proper compartments? List examples.
1. Gated transport - proteins and RNA move between cytosol and nucleus (topologically equivalent) through nuclear pore complexes
2. Transmembrane transport (protein translocation) - transport of specific proteins across the membrane from cytosol into a space that is topologically DISTINCT
3. Vesicular transport - movement between topologically equivalent spaces (ER to Golgi); do not cross a membrane; selectivity of fusion
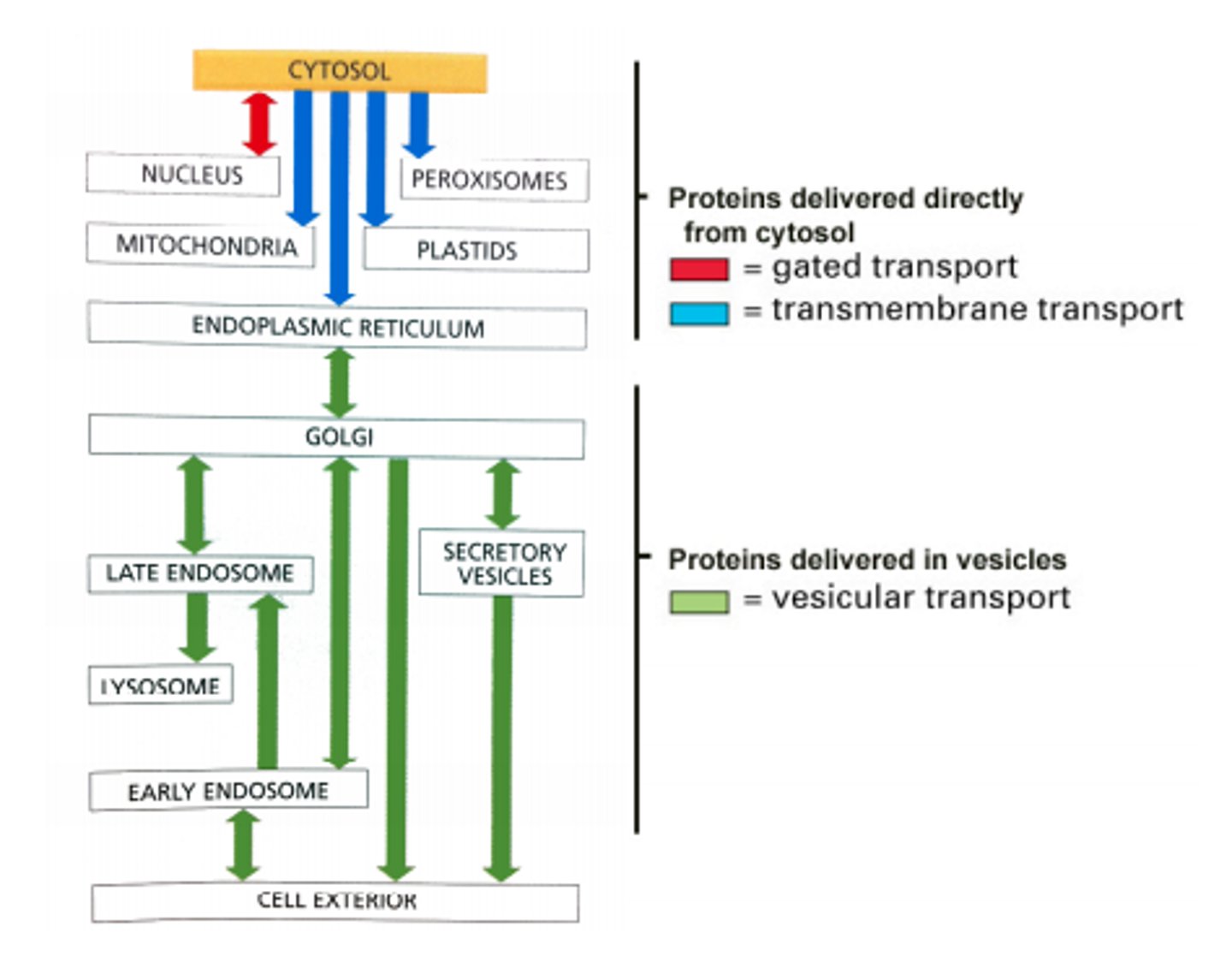
What is the relevance of "topologically equivalent" compartments with regards to transport?
Topologically equivalent compartments' molecules can get from one to the other without having to cross a membrane (nucleus is TE with cytosol; extracellular space is TE with ER, lysosomes, Golgi)
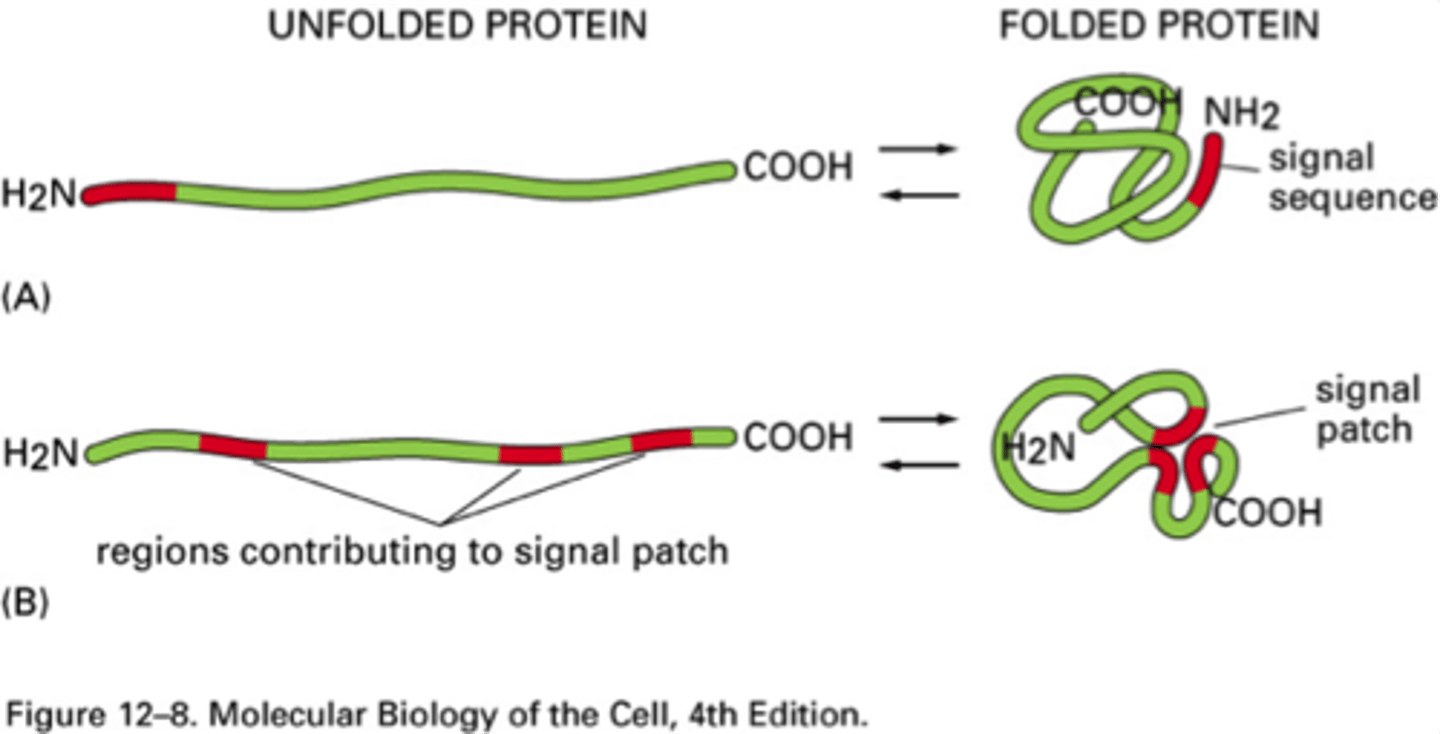
What are the two types of sorting signals?
1. Signal peptides (more common) - N-terminal; 15-60 AAs that direct proteins from cytosol to appropriate compartment (ER, mitochondria, chloroplasts, peroxisomes)
2. Signal patches - 3D arrangement of AAs on surface of a folded protein that direct enzymes and proteins to correct compartments (Golgi to lysosome transport)
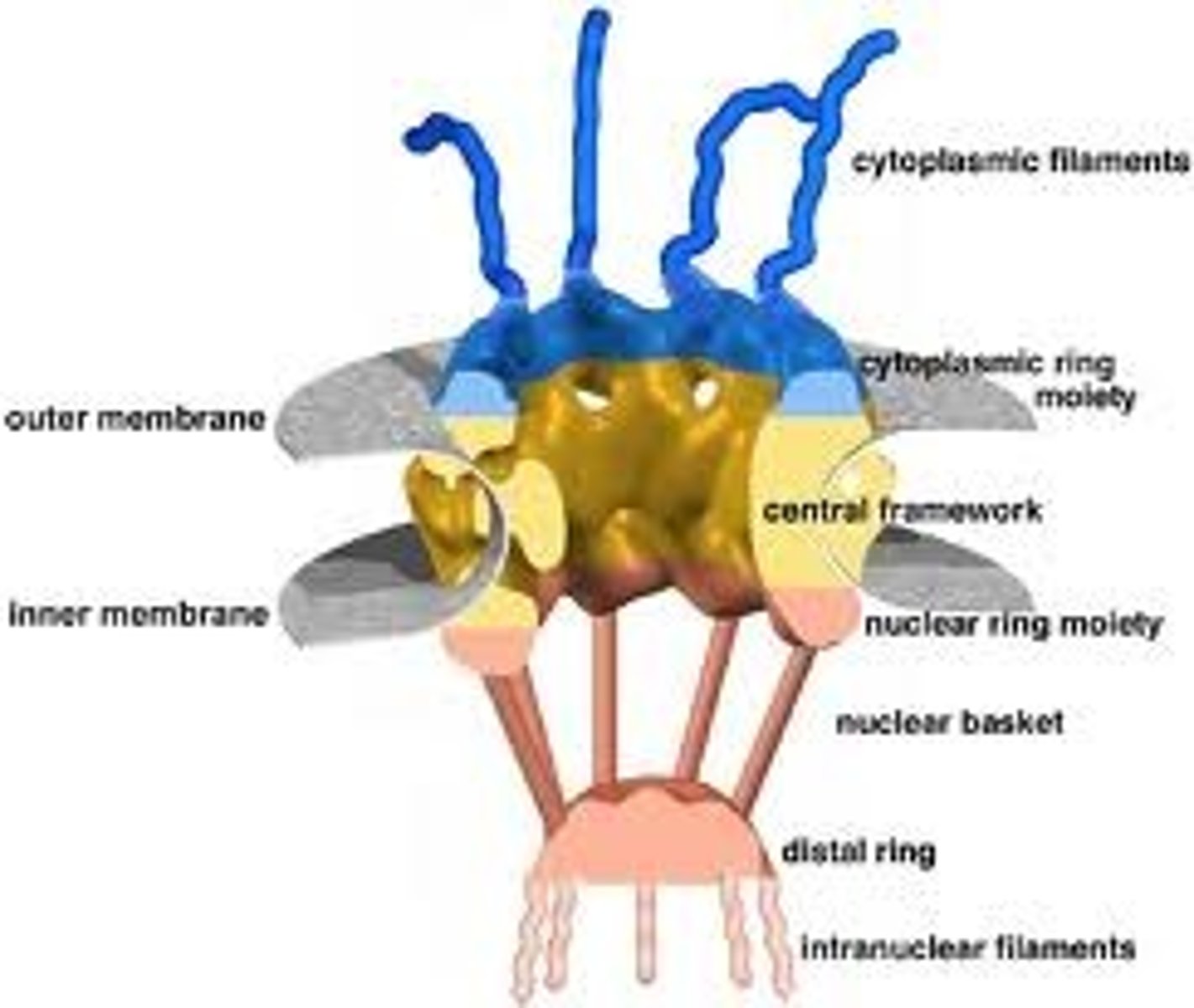
Identify components of the nuclear envelope.
Composed of two continuous membranes:
-Inner nuclear membrane (binds to chromosomes; nuclear lamina supports filaments)
-Outer nuclear membrane (studded with ribosomes, synthesized proteins are transported into the perindclear space) continuous with lumen of the ER
What is the function of nuclear pore complex? Describe movement through the pore. What are criteria for passive vs active transport through nuclear pore complex?
-Transport up to 1000 macromolecules/second (both directions at the same time!)
-Each NPC contains aqueous passages, so small water soluble molecules diffuse passively, but proteins larger than 60,000 daltons need active transport
What is nuclear localization signal?
Sequence of +++ charged AAs that form a patch and signal for a protein to enter the nucleus
What is nuclear import receptor?
Importin; recognize nuclear localization signal and binds to cargo protein
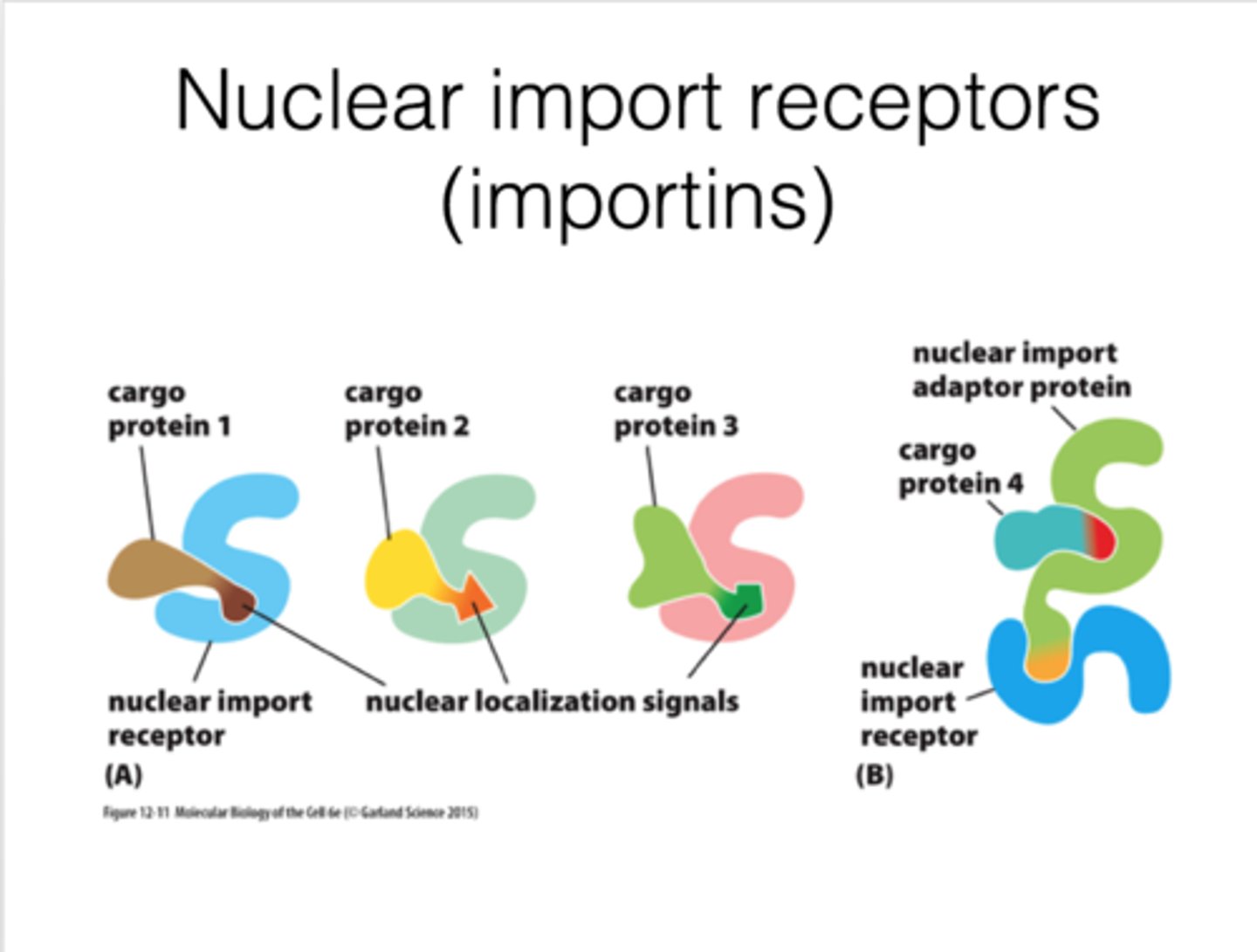
What is adaptor protein?
Helper; structurally related to nuclear import receptors and recognize nuclear localization signals
on cargo proteins
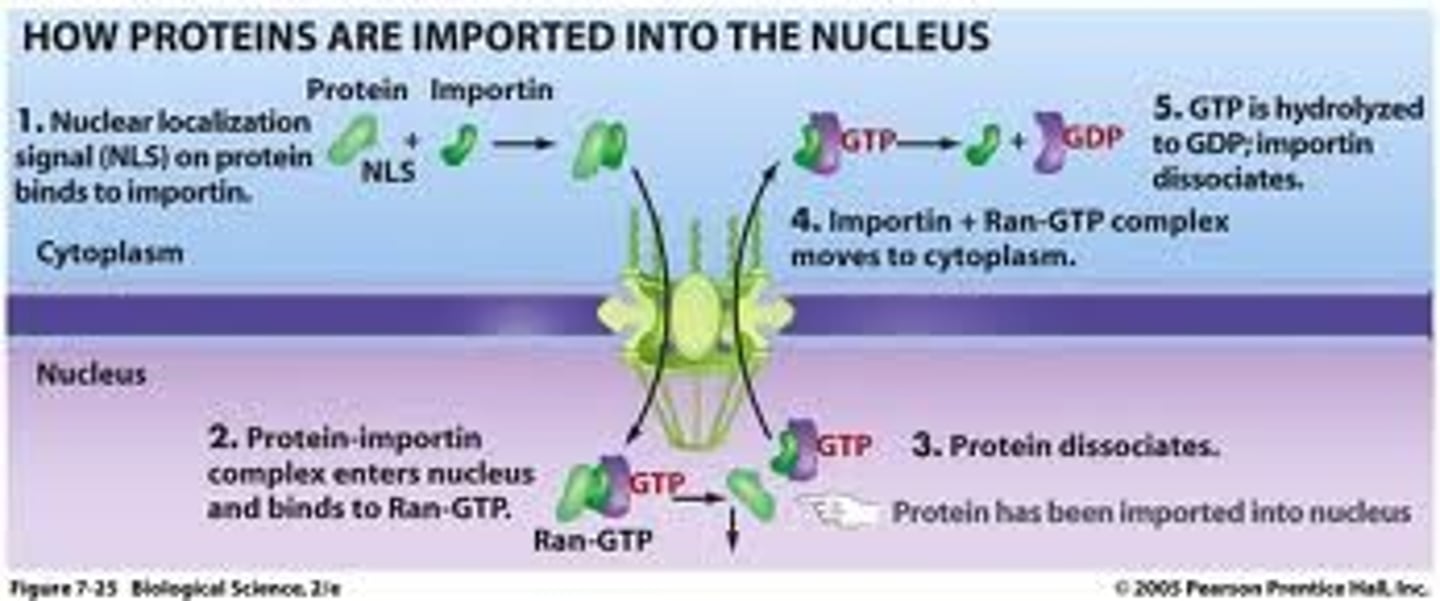
In detail, describe the Ran-GTPase cycle that controls nuclear import.
1. Importin binds to cargo
2. Importins bind to FG repeats in unstructured domain of channel nucleoporins; weak interactions referred to as "random walk"
3. Binding of cargo + receptor to Ran-GTP promotes release of cargo from receptor (Ran-GTP in higher concentration in nucleus)
4. Importin + Ran GTP move through NPC
5. Ran GAP: hydrolysis of Ran-GTP, Ran-GDP dissociates from receptor
6. Empty receptor (importin), ready for another round
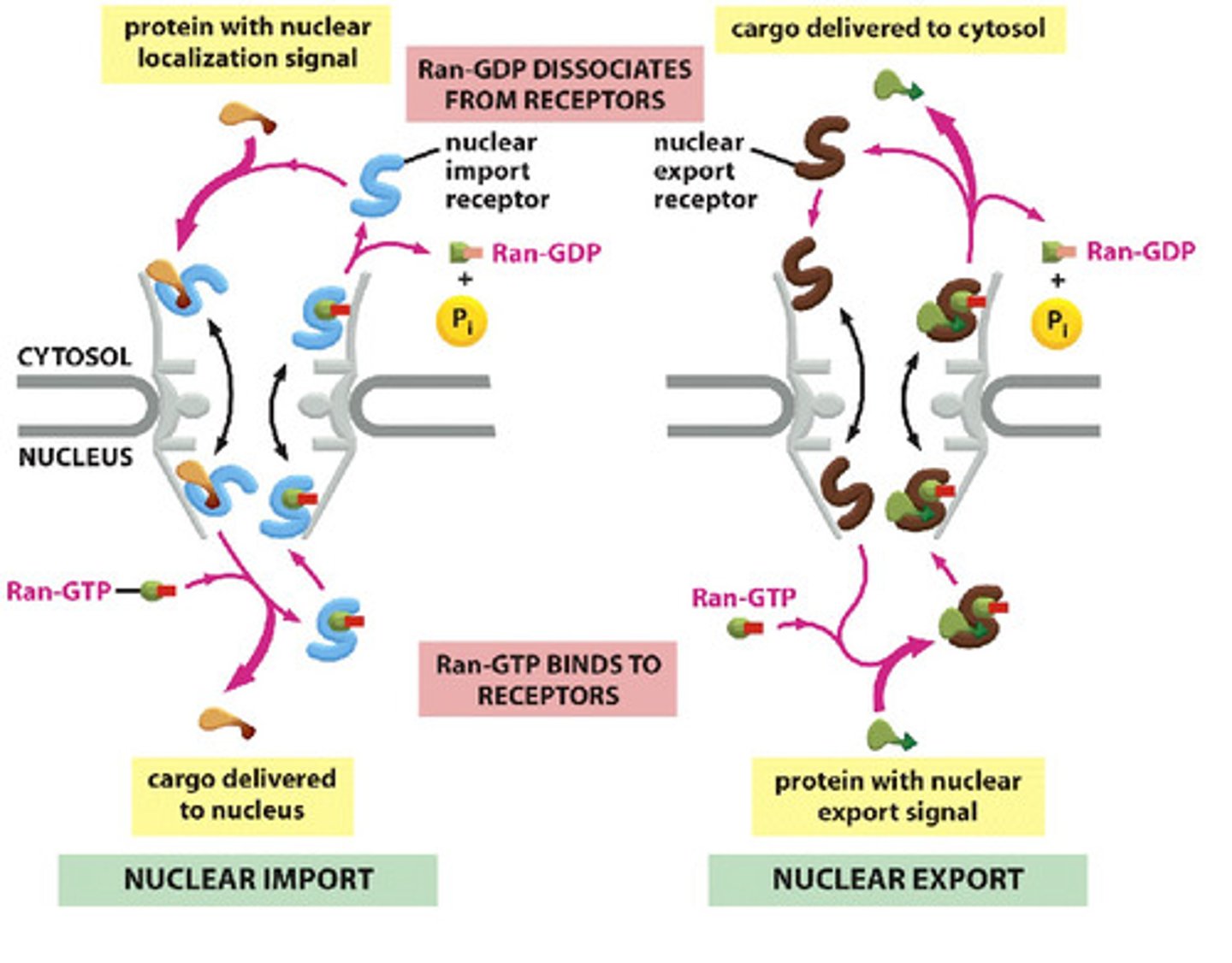
In detail, describe the Ran-GTPase cycle that controls nuclear export.
1. In nucleus, Ran-GTP promotes cargo binding to exportin (export receptor)
2. Ran-GAP binds, promotes GTP hydrolysis. Exportin releases its cargo and Ran-GDP
3. Exportin retruned to nucleus
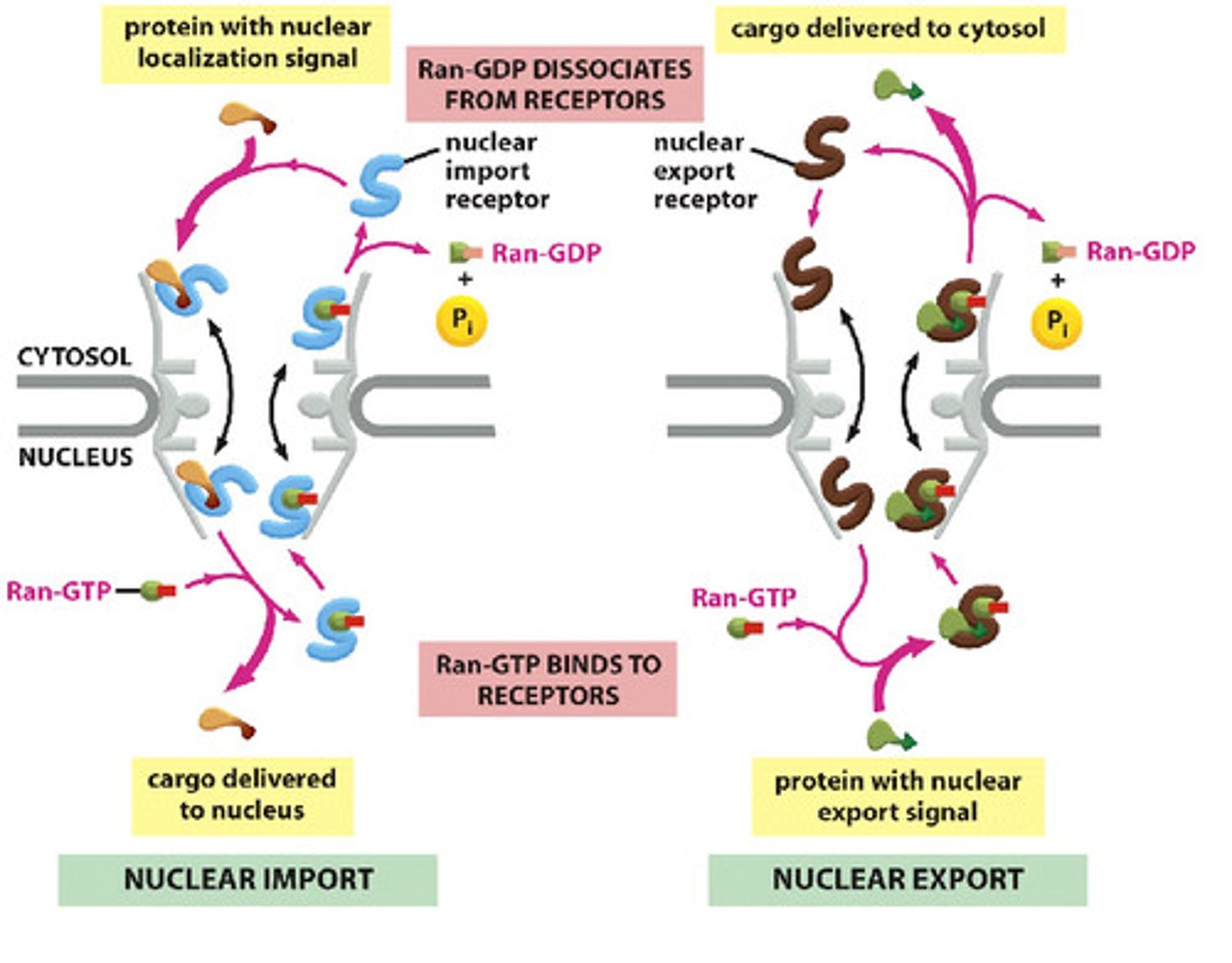
Why is the location of various components important for directional transport across the nuclear membrane?
Ran-GTP is in high concentration in the nucleus, Ran-GDP is in the cytosol (cannot bind receptor)
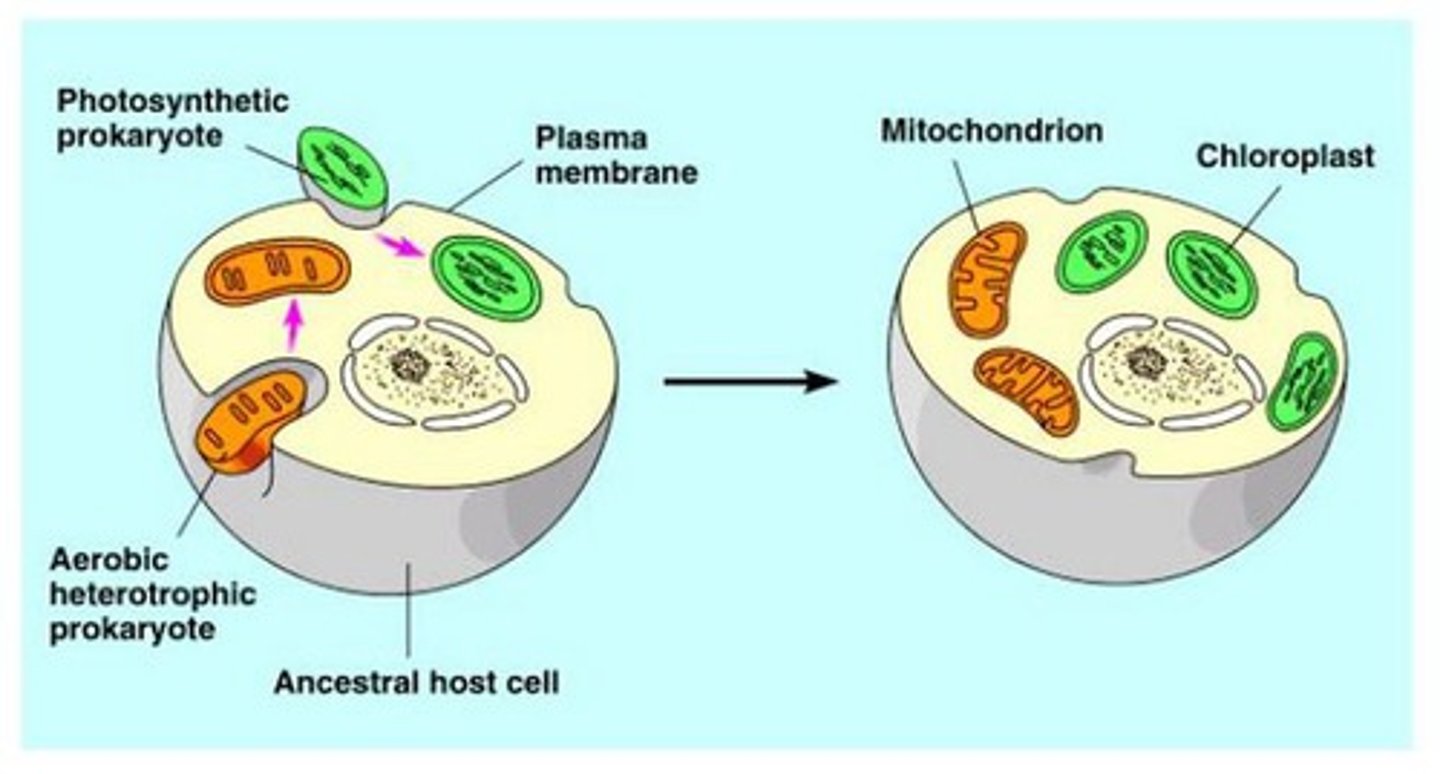
What is the origin of mitochondria as described by endosymbiosis theory? List support for this theory (ie structural/functional comparisons, evidence of endosymbiosis in other experiments...).
Ancient archaea engulfed aerobic bacteria that are protected and receive nutrients
-Both mitochondria and bacteria have circular DNA, binary fission, ribosomes, and inner mitochondrial membrane resembles plasma membrane of bacterial cell
-Jeon's Ameoba proteus culture became infected with rod-shaped bacterium (most died, some lived and ended up depending on the bacteria because of an enzyme it produced)
Describe structure and functions of the mitochondria.
Site of ATP synthesis
-Energy in H+ gradient powers ATP synthesis
Site of Fatty Acid Degradation
-Occurs in mitochondrial matrix
Does the mitochondria make most of its own proteins (since it has ribosomes)?
All mitochondrial compartments contain proteins derived from polypeptides synthesized by the cell
What is the significance of mitochondrial DNA?
Passed only along maternal line; been used to trace evolution and migration of human species; diseases associated with mutations in mitochondrial DNA
What are the cause and basic characteristics of Leigh Syndrome?
Cause: mutations in one of several mitochondrial genes that affect proteins required for ETC (failure of oxidative metabolism / ATP synthesis)
Characteristics: progressive neurological deterioration; failure to thrive, loss of mental and movement abilities, hyperintense lesions in basal ganglia
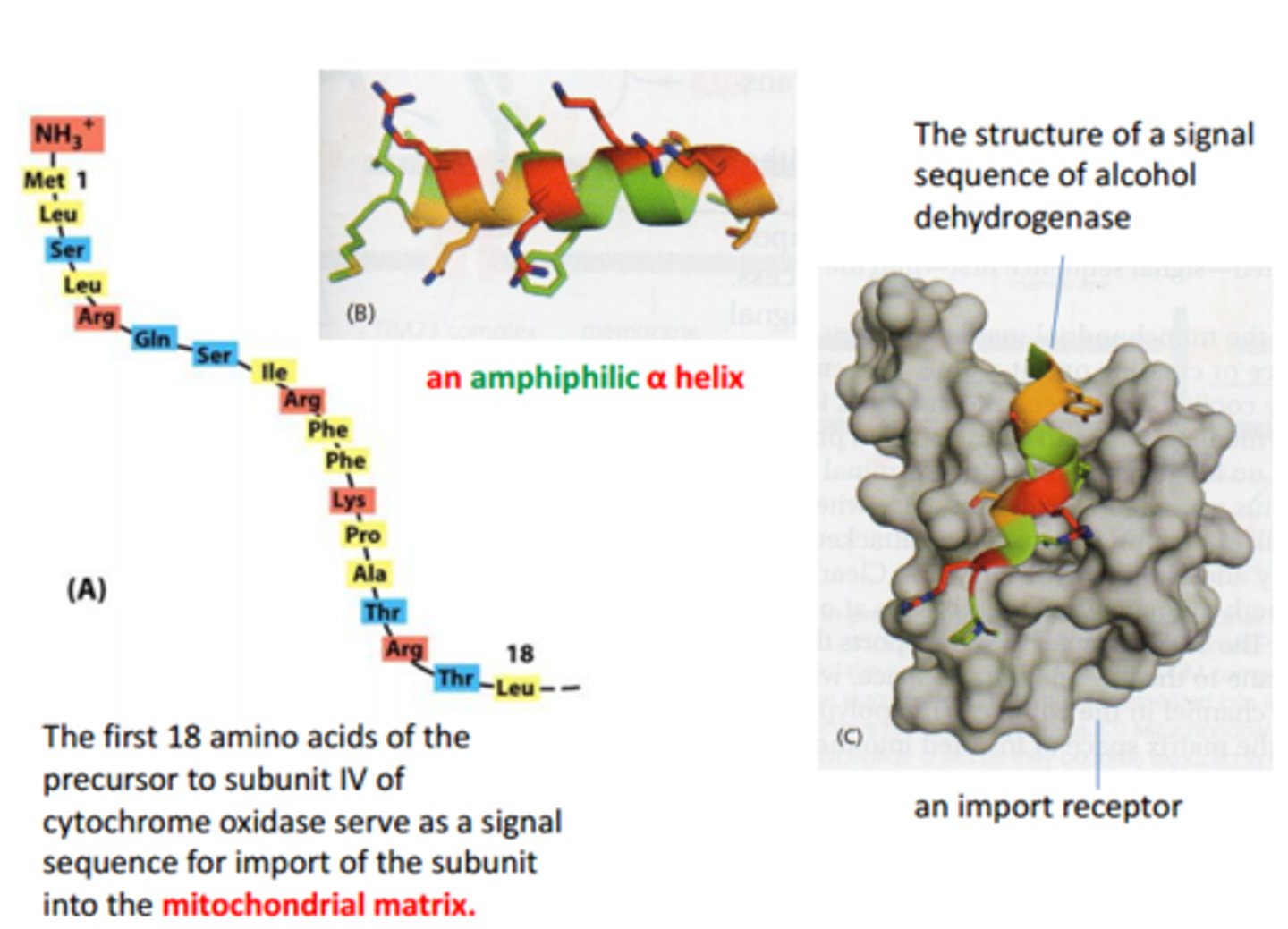
What are properties of mitochondrial signal sequence?
Structure: N-terminus signal peptide comprised of polar and non polar AAs arranged in amphipathic helix (one side + charges, one side nonpolar)
Function: direct translocation of most mitochondrial proteins to matrix
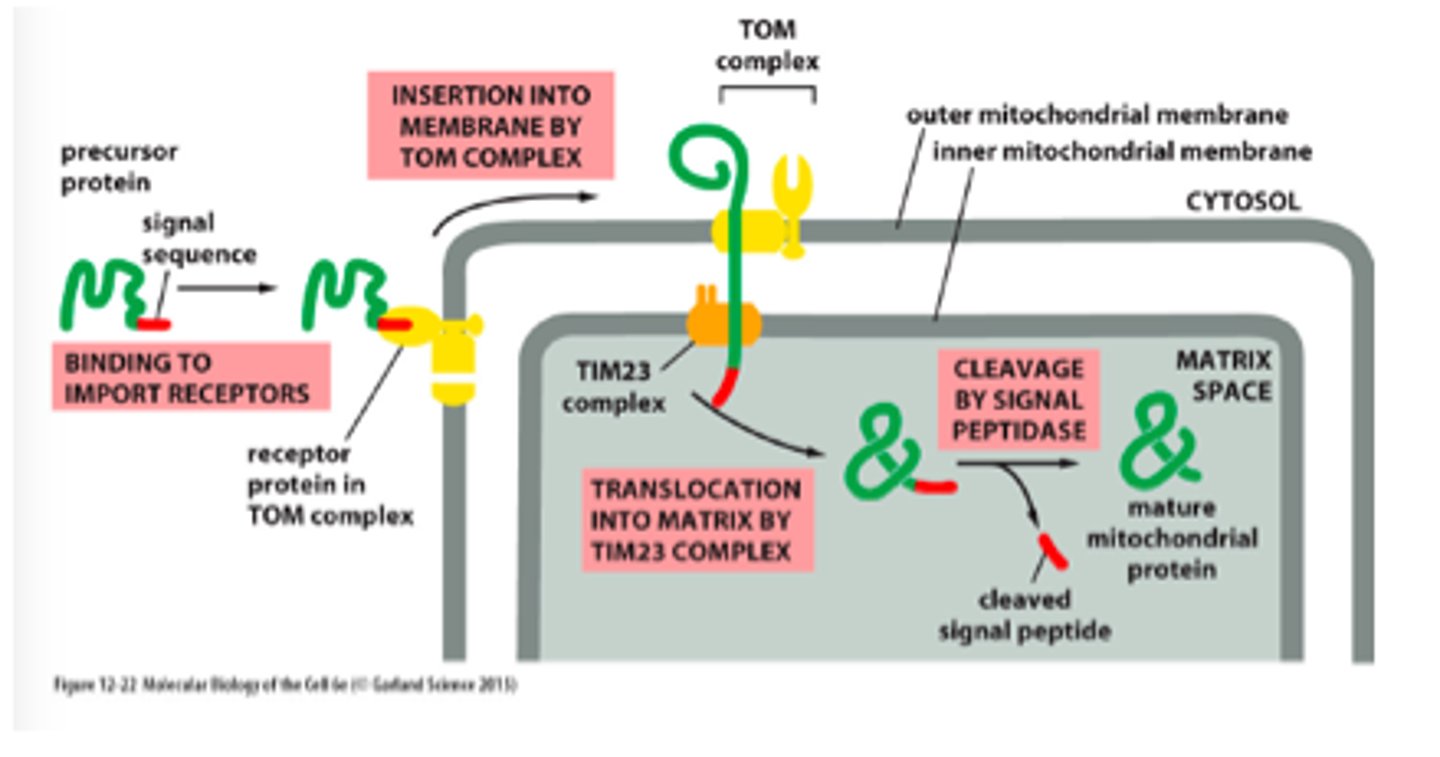
List the translocators, their location in the mitochondria, and main functions
-TOM (outer membrane) - import of all nucleus encoded mitochondrial proteins
-SAM (outer membrane) - help fold beta barrel proteins
-TIM22 and TIM23 (inner membrane) - mediate insertion of subclass of IM proteins (ATP synthesis precursors)
-OXA (inner membrane) - insertion of IM proteins synthesized within mitochondria
Describe the type of translocation involved in protein import to mitochondria.
Post-translational
Describe protein import (stepwise) through mitochondria into matrix space.
-N-terminal signal sequence is recognized by TOM
-Protein translocated through TIM23 complex so that it transiently spans both mitochondrial membranes
-Signal sequence cleaved by signal peptidase in matrix and degraded
What are 3 specific areas in protein import (protein translocated to matrix space) that require an input of energy?
1. Cytosolic hsp70 chaperone requires ATP hydrolysis to be released from precursor protein
2. Signal sequence translocated into membrane space needs energy from membrane potential (+ charge on signal sequence is attracted to - in inner membrane)
3. Mitochondrial hsp70 (part of import ATPase) binds to regions of polypeptide chain as they become exposed in matrix space and pulls the protein through the translocation channel using ATP hydrolysis
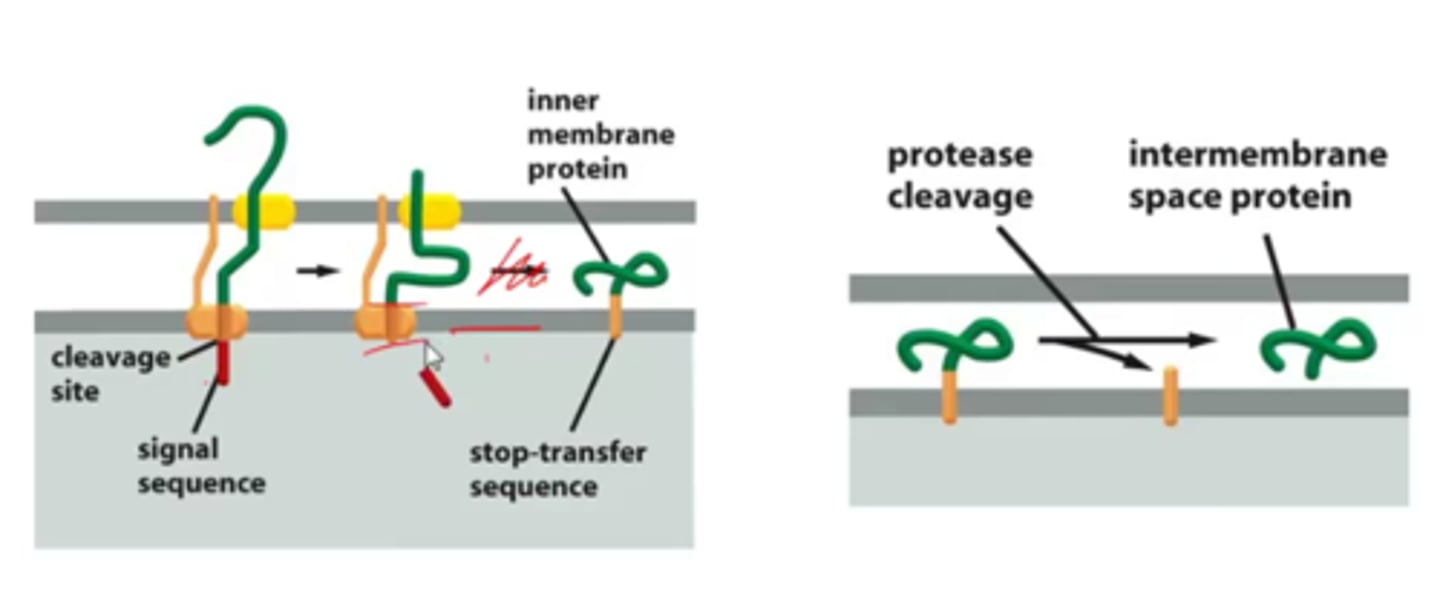
Describe translocation of an inner membrane protein and protein released to intermembrane space.
-N terminal sequence enters matrix space
-Stop transfer sequence prevents translocation across IM (hydrophobic to stay in bilayer)
-Protease cleaved and stays in inner membrane and IM space protein stays in IM space
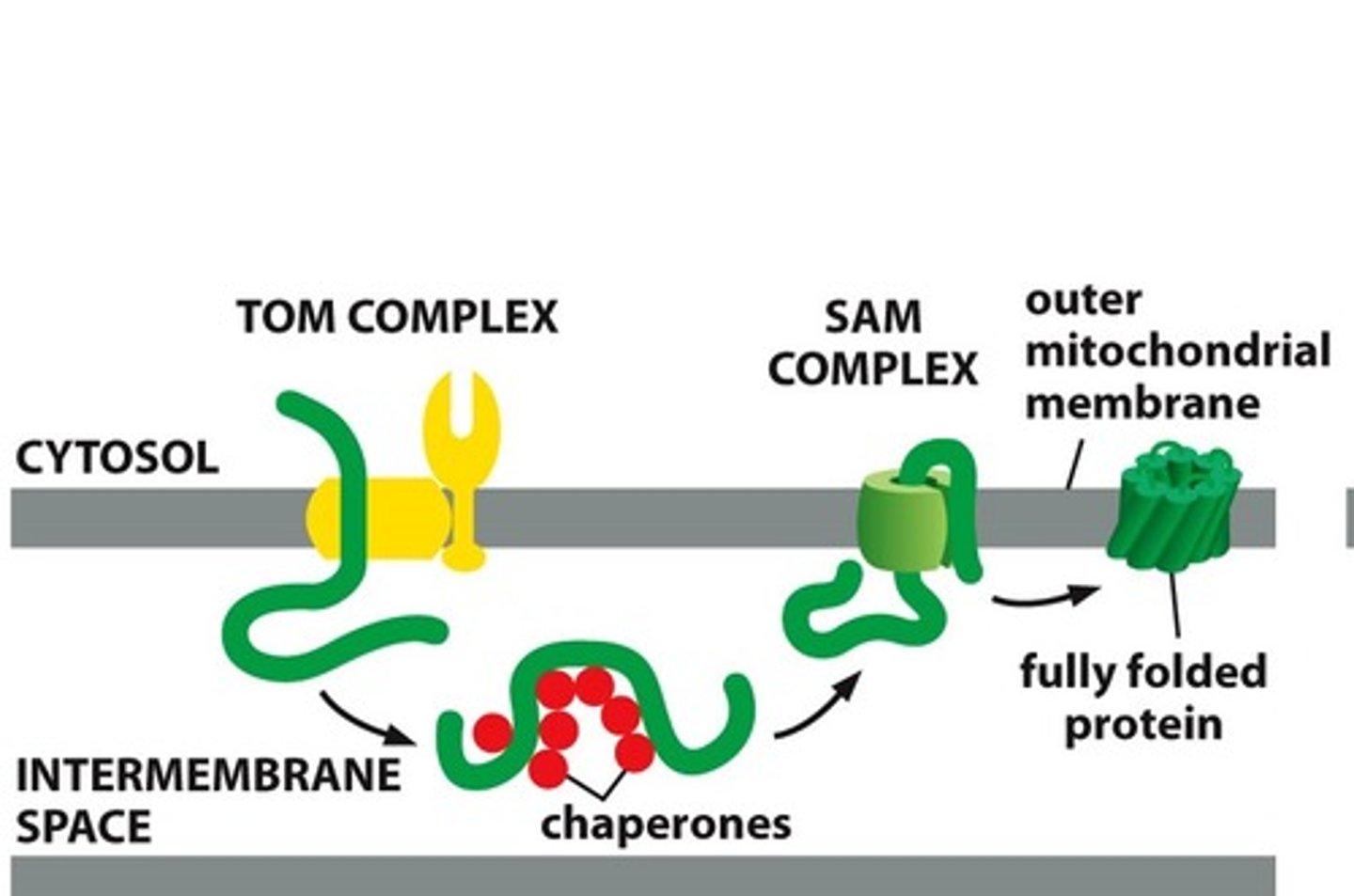
Describe insertion of porins.
-Porins transported unfolded from TOM into IM space
-Bind to chaperone proteins to prevent aggregating
-Bind to SAM in outer membrane, which inserts them into the outer membrane
What are the main functions of peroxisome in eukaryotic cell?
-Catalyze formation of plasmalogens (myelin)
-Lipid catabolism (breakdown of fatty acids)
-Breakdown toxic substances (use oxygen, detoxify, ethanol metabolism)
-Use H2O2 to make ethanol into acetaldehyde
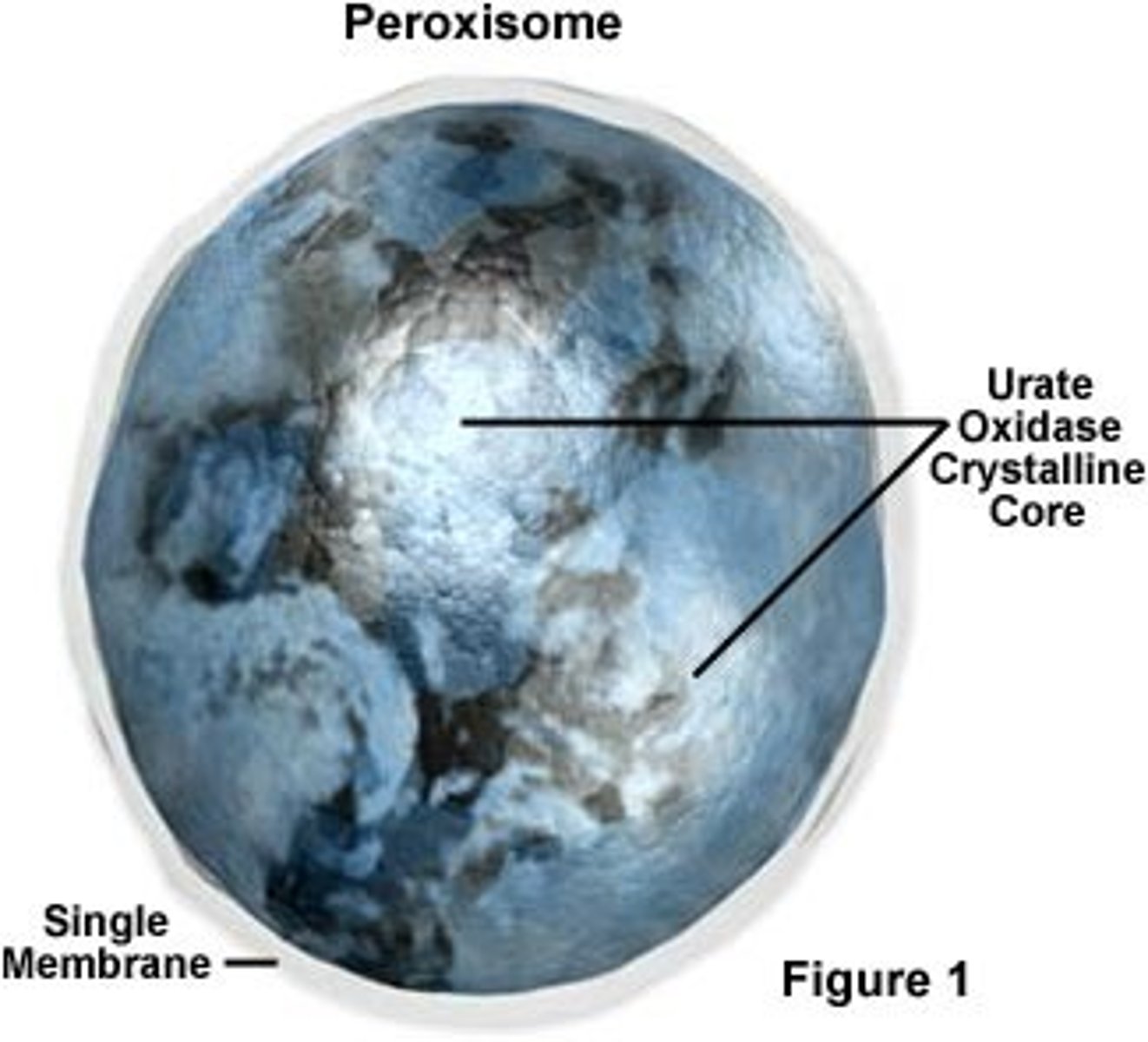
What are similarities and differences between peroxisome and mitochondria?
Peroxisomes have:
-Single membrane
-No DNA or ribosomes
-No genome
Both have:
-Oxygen utilization
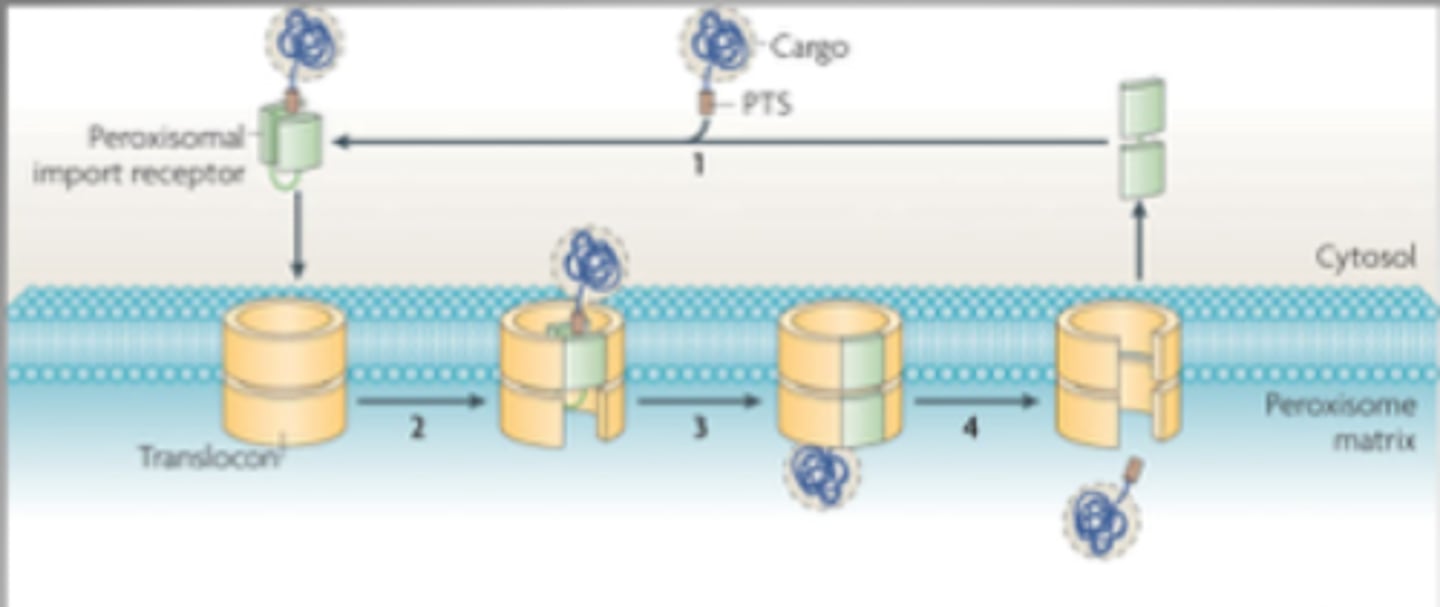
Describe protein import into peroxisomes.
-Energy required
-Signal sequence of Ser-Lys-Leu (SKL) at C terminus of peroxisomal protein is recognized by soluble receptor in cytosol (Pex 5)
-Proteins can remain folded during import
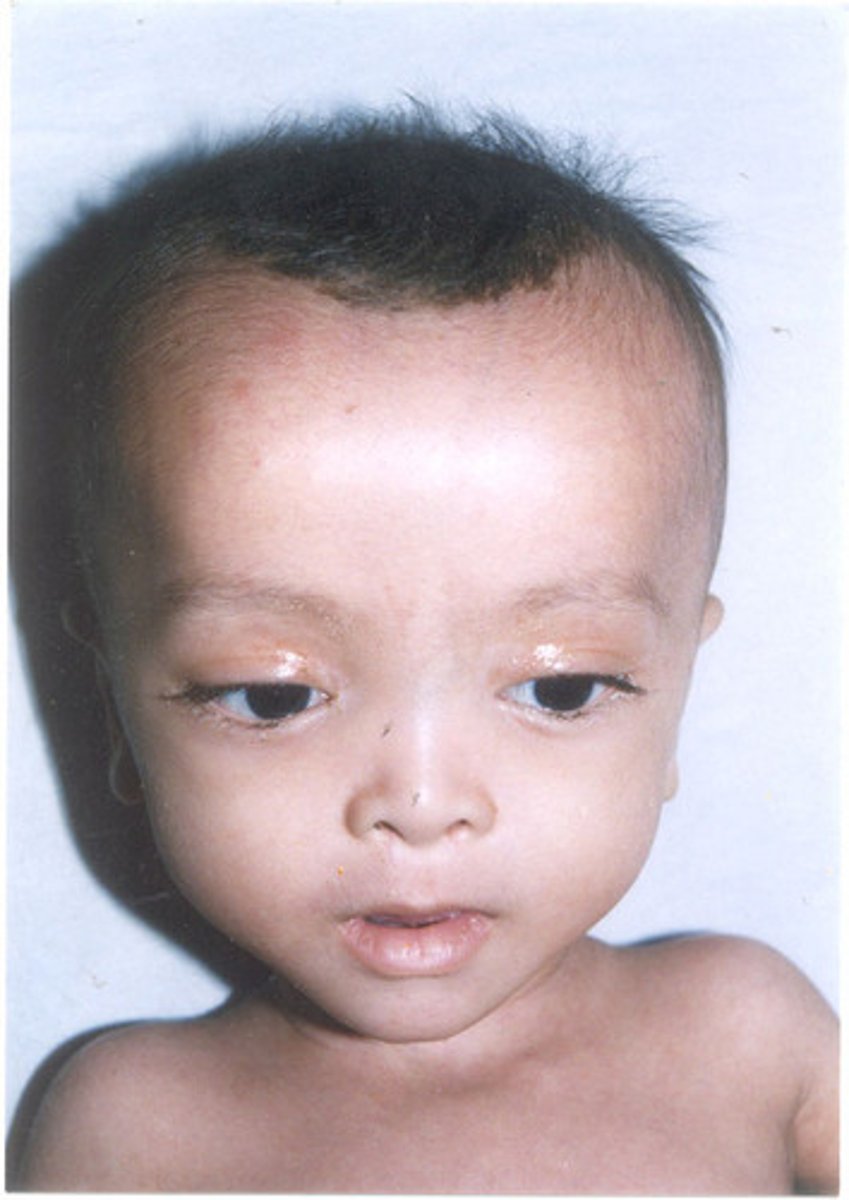
What is the cause and characteristics of Zellweger Syndrome?
Cause: mutation in PEX genes, defece in importing proteins into peroxisome "empty peroxisomes"
Symptoms: weak muscle tone, feeding problems, hearing/vision loss, and seizures for newborns, also distinct facial features including a flattened appearance to the face
What is the structure of the ER?
-Netlike labyrinth of branching tubules and flattened sacs that extend throughout cytosol
-ER and nuclear membranes form a continuous sheet enclosing the ER lumen
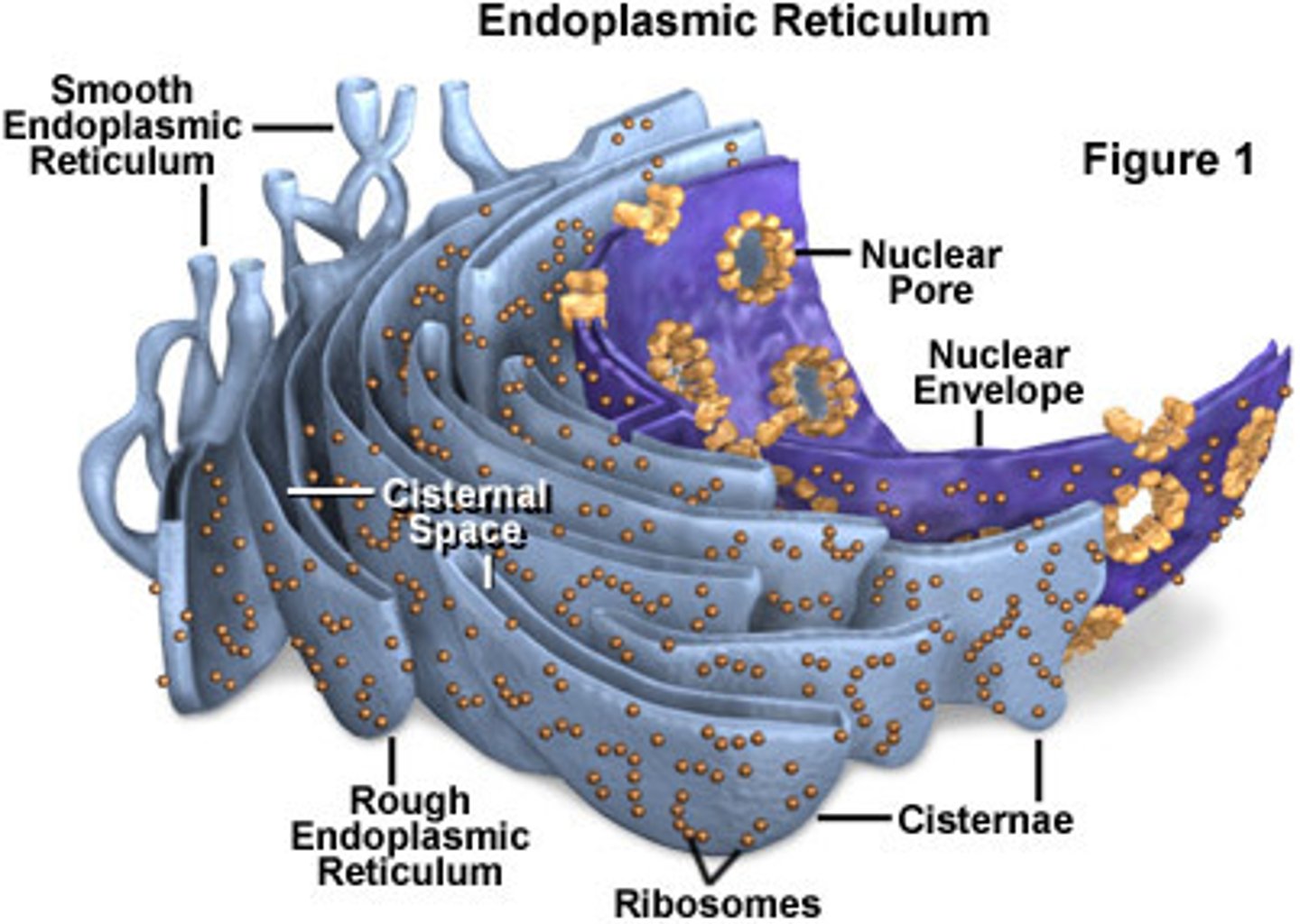
What are the functional roles of smooth ER, rough ER?
Smooth: primary site of ER vesicle budding, transitional zones, lipid metabolism (Ca2+ storage), steroid biosynthesis
Rough: primary site of protein synthesis of integral TM proteins and "soluble" secreted proteins (studded with ribosomes)
What is the transitional zone?
Areas of smooth ER where transport vesicles and new proteins/lipids bud off for transport to the Golgi

Describe key experiment (Blobel) that led to the discovery of a signal sequence.
SDS-page analysis shows that translation in the presence of microsomes (mini ER) results in a slightly smaller polypeptide
Conclusion: in presence of microsome, a leader sequence of the precursor protein directs it to the ER where it is cleaved off by a signal peptidase following transport into the ER
Describe the type of translocation involved in protein import into the ER.
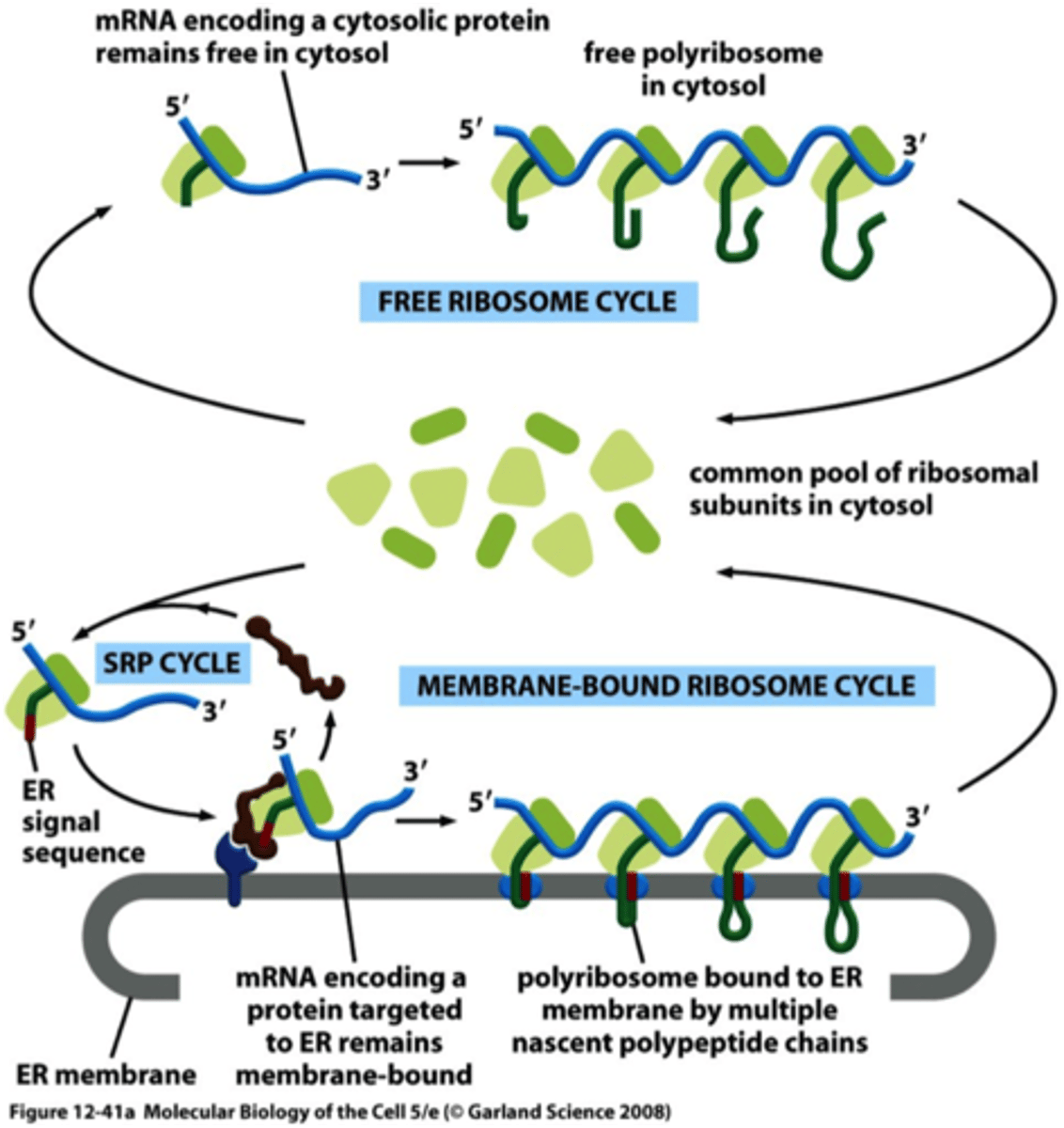
Co-translational: ribosome that is synthesizing the protein is attached directly to the ER membrane, so one end is translocated into the ER while the rest is synthesized
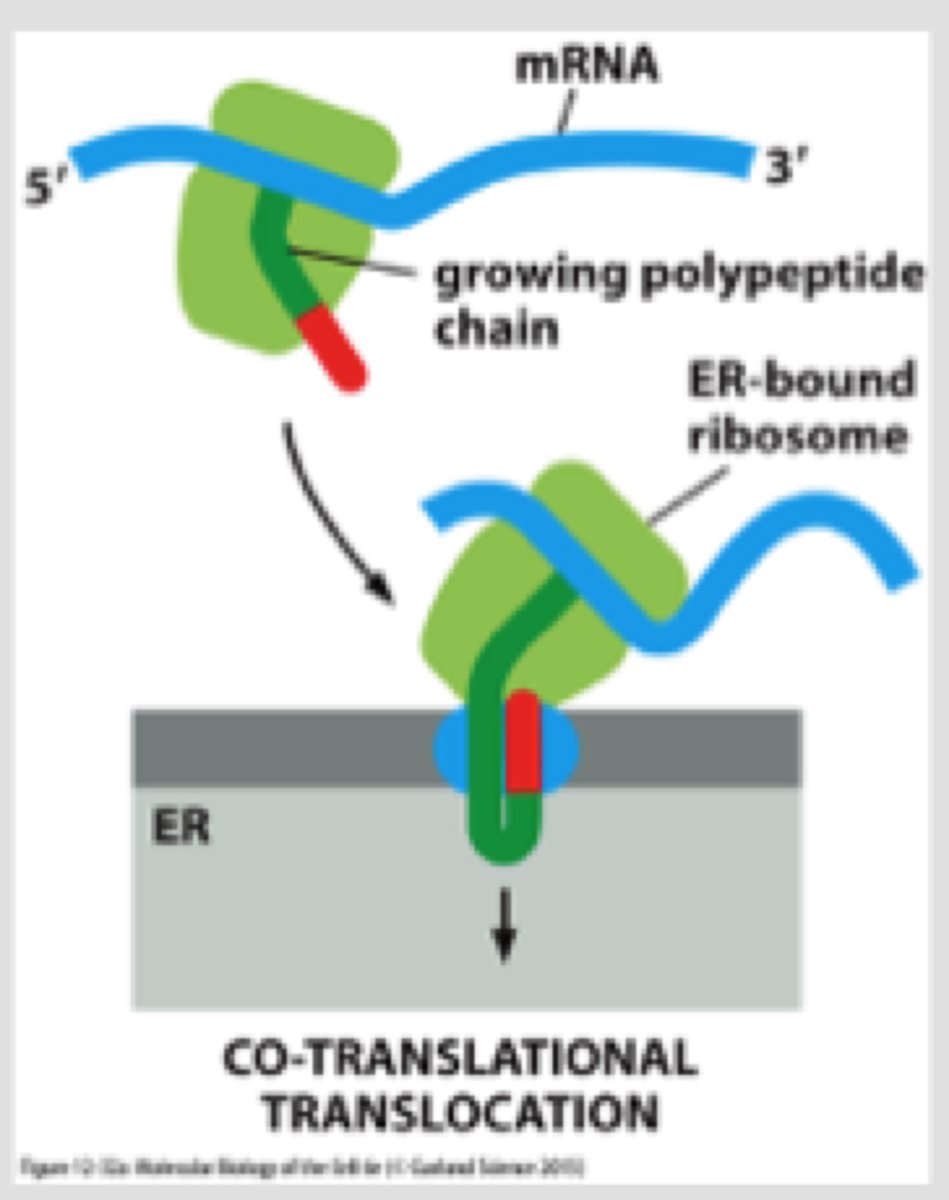
What is the SRP, what does it bind to?
-SRP (signal recognition particle made of protein + RNA) has large hydrophobic pocket lined with Met AA binds to ER signal sequence (hydrophobic) as it emerges from the ribosome
-SRP wraps around large ribosome subunit; one end binds to signal sequence and the other end blocks elongation factor binding site
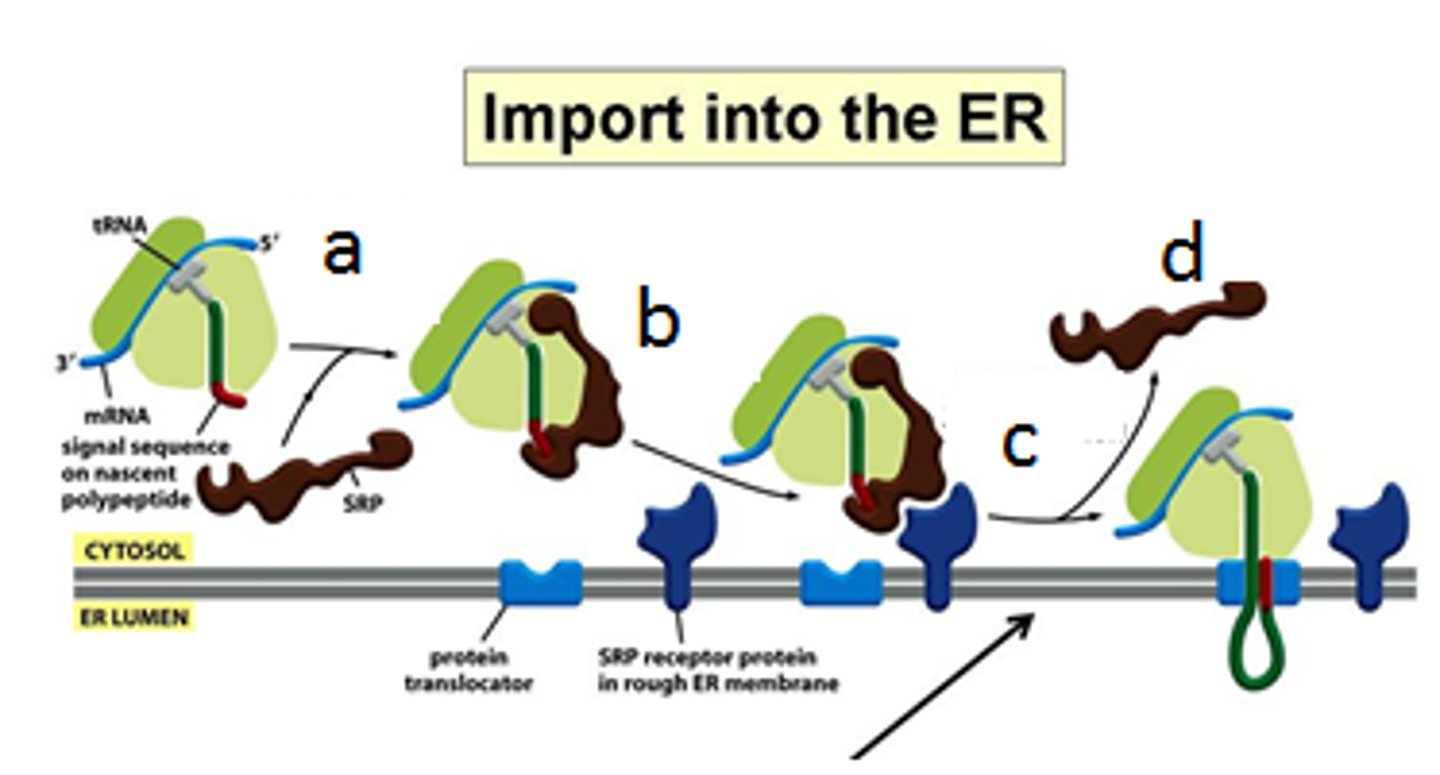
Why is a halt in protein synthesis necessary?
Don't want the protein to be already made/folded before it reaches the ER (co-translational)
When is SRP and its receptor released from ribosome complex?
After the SRP receptor in the ER membrane binds the SRP-ribosome complex and directs it to the translocator
Describe two populations of ribosomes.
Membrane-bound: attached to cytosolic side of ER membrane, engaged in synthesis of proteins that are being concurrently translocated into the ER
Free: synthesize all other proteins encoded by the genome
What is Sec61? Describe its functional role in the translocon (as a gated channel).
Heterotrimer that forms a dynamic gated channel; core of the translocator; lateral gating into lipid bilayer
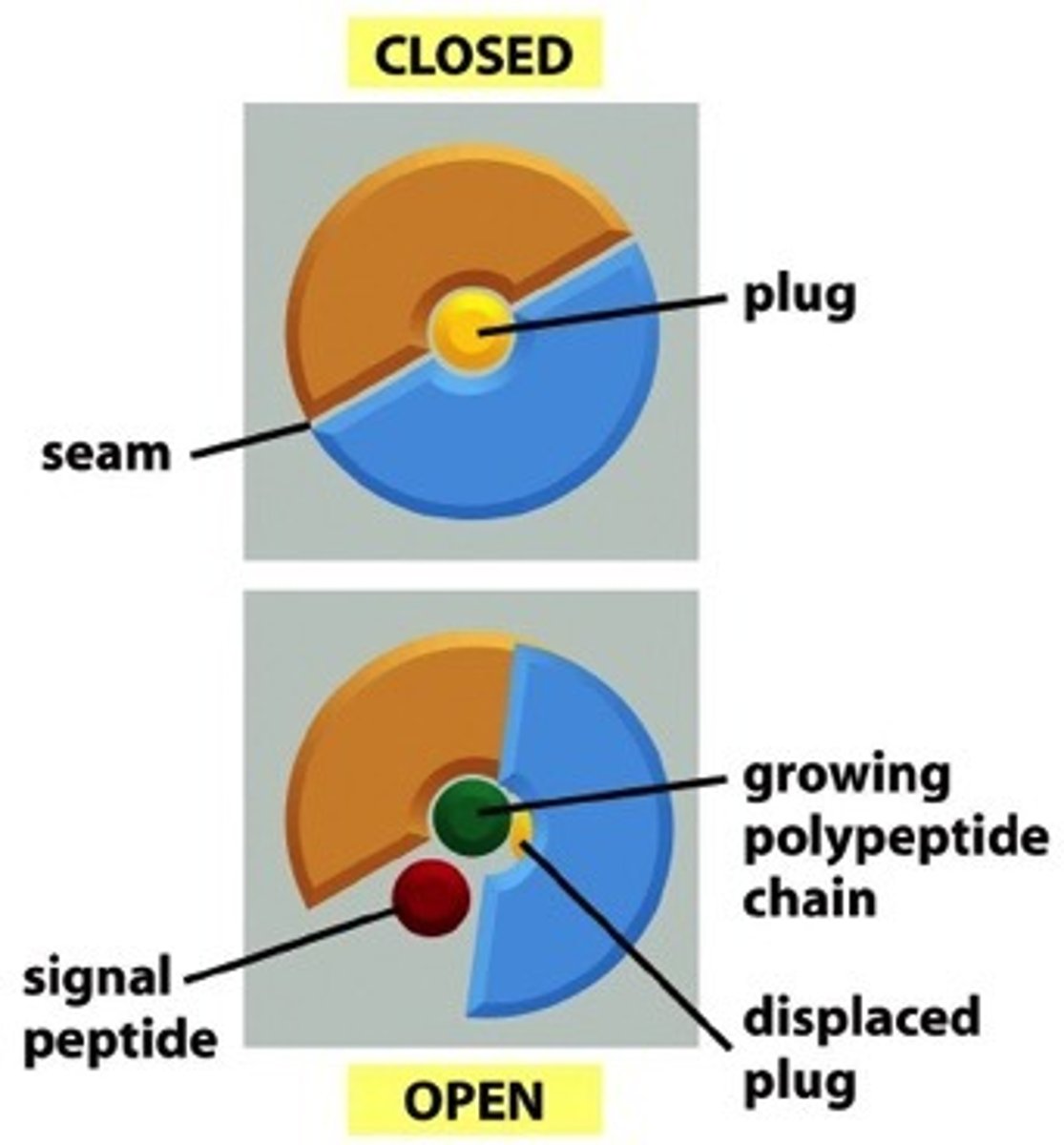
The translocator is gated in two directions, explain the significance of this.
List steps involved in translocation of soluble protein to ER lumen (include start transfer signal).
1. ER signal sequence binds to a specific binding site in the pore, serves as a start-transfer signal that opens the pore
2. Signal sequence remains bound to Sec61 complex during translocation
3. Signal sequence also in contact with lateral seam
4. Signal peptidase cleaves signal sequence, mature protein released into lumen
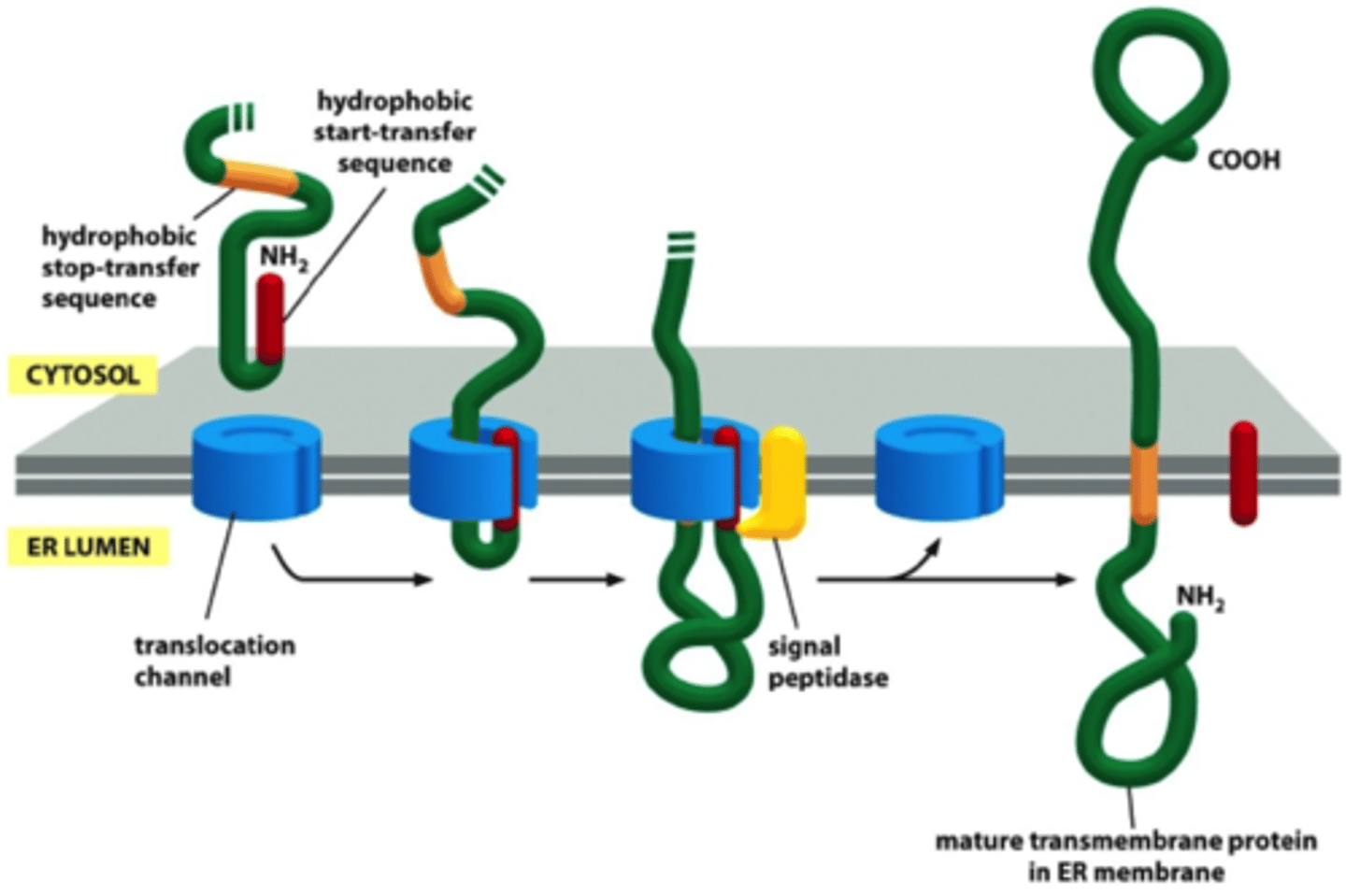
How are single pass (include both N terminal signal and internal signal sequence examples) proteins inserted into ER membrane?
-N-terminal signal sequence opens translocator; additional hydrophobic segment (stop transfer sequence) interacts with binding site within the pore; translocator opens laterally, and stop transfer signal anchors protein in the membrane in bilayer (N terminus is extracellular in lumen because extracellular space is TE to lumen; C-terminus in cytosol)
-Internal signal sequence; + charged AA cluster upstream of start transfer sequence; C-terminus passed across membrane
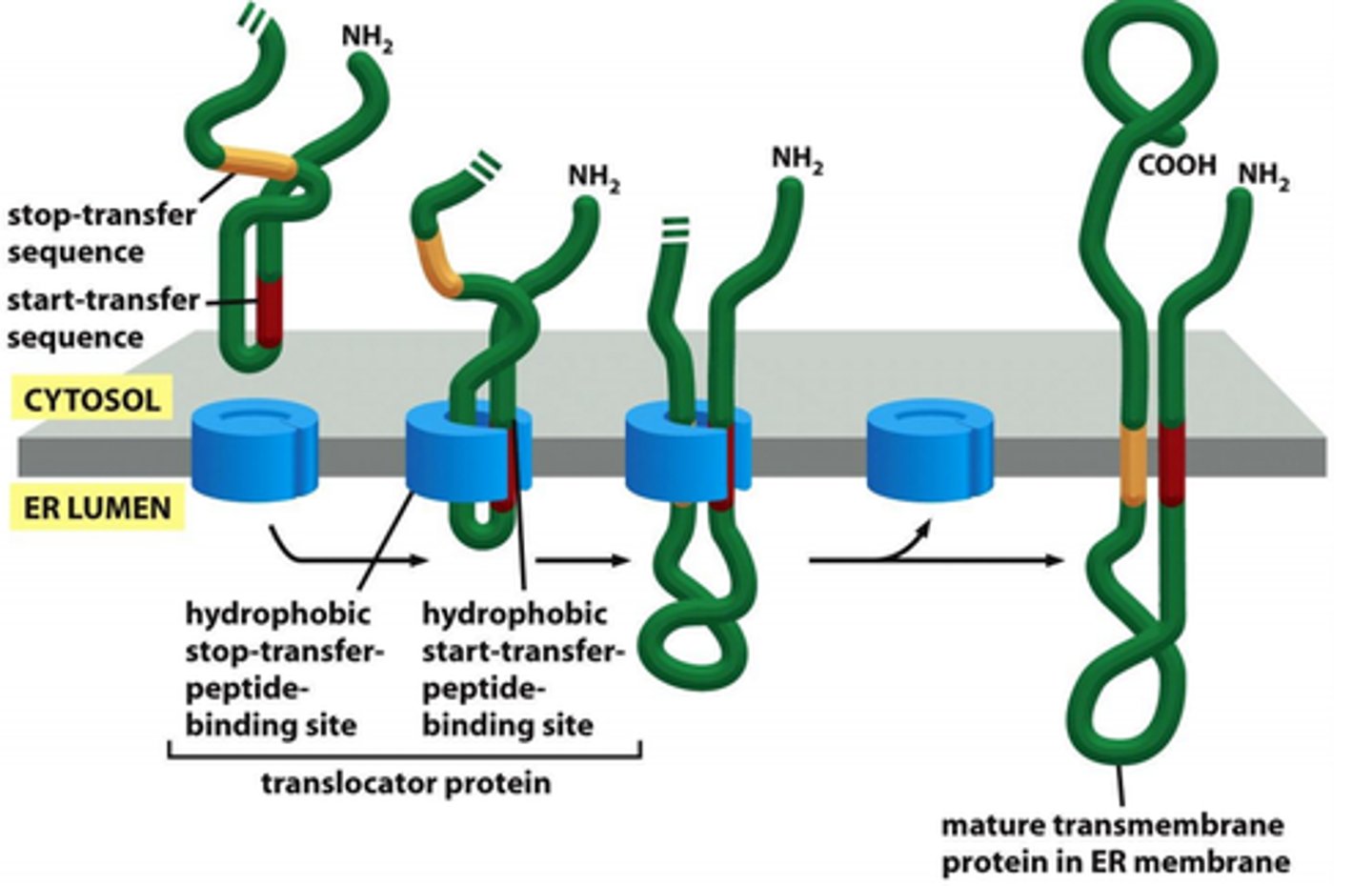
How are multipass proteins inserted into ER membrane?
Polypeptide chain passes back and forth repeatedly across the lipid bilayer as hydrophobic alpha helices; number and position of start and stop transfer sequences determine side of N- and C-termini
What is an example of ER resident protein and function?
Proteins that normally reside in ER lumen and contain ER retention signal
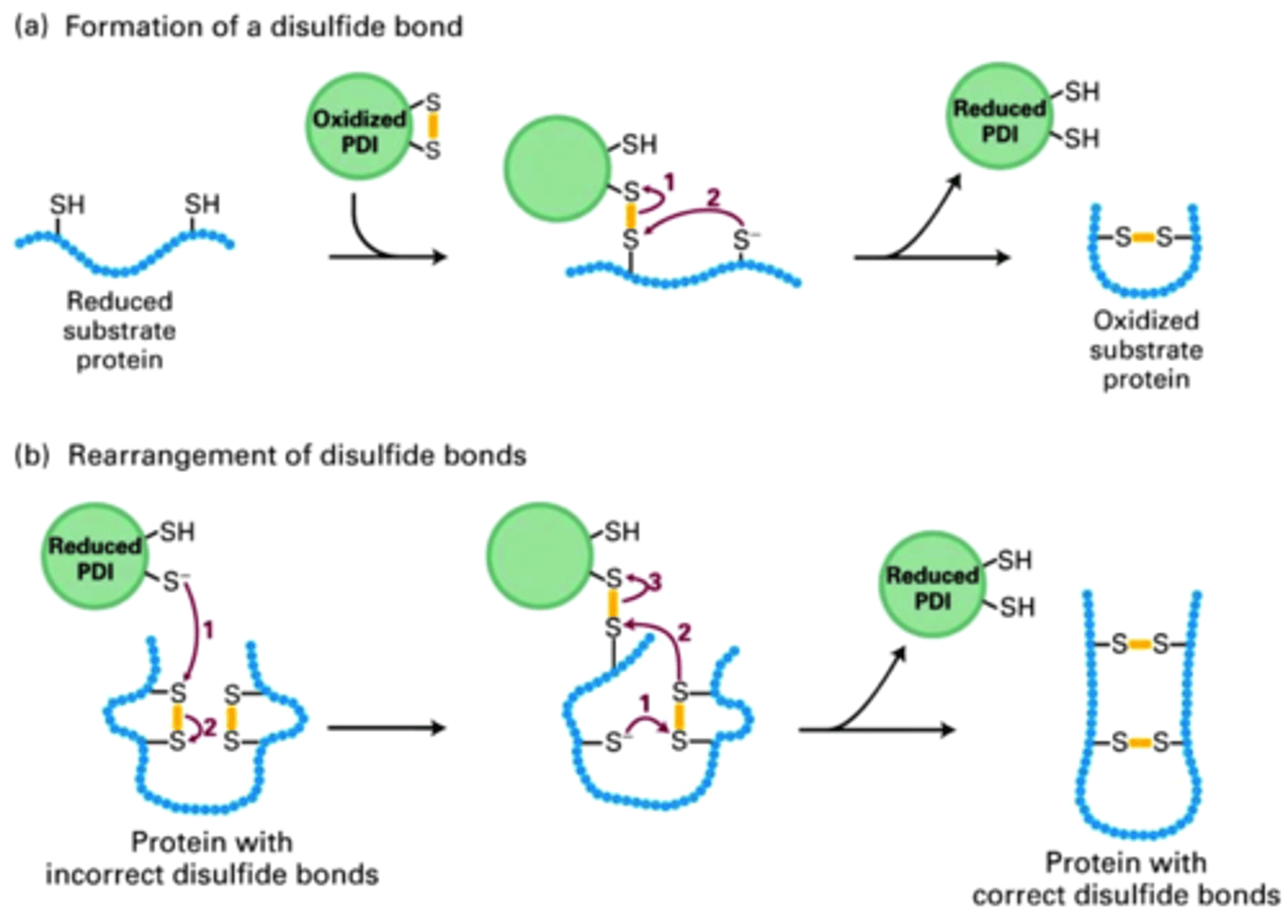
Ex. PDI (protein disulfide isomerase) catalyzes oxidation of -SH groups on cysteines to form disulfide bonds
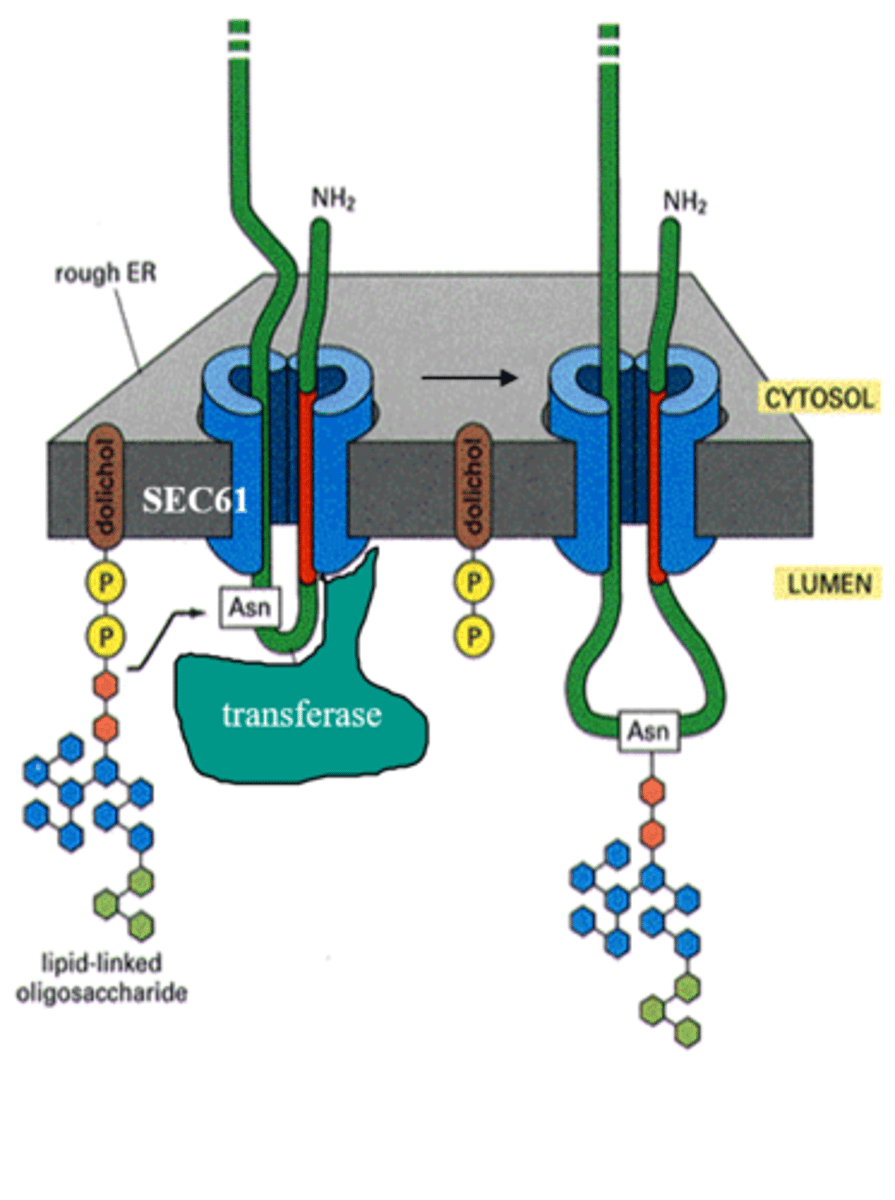
Describe the protein glycosylation reaction (oligosaccharyl transferase), N-linked protein glycosylation. What is the purpose of dolichol?
Protein glycosylation catalyzed by oligosaccharyl transferase, which mediates the transfer of oligosaccharide (small sugar) to the Asn residue on the protein (N-linked protein glycosylation)
-Dolichol holds onto sugar
What is the role of N-linked glycosylation in ER protein folding (know example of calnexin), enzymes
involved in glucose trimming and addition? Figure 12-49 (text)
Calnexin - chaperone protein that binds carbohydrates; glucosidase removes glucose but glocosyl transferase recognizes exposed hydrophobic patches and adds glucose back to prompt another cycle with Calnexin
Describe the mechanism of retrotranslocation.
Proteins that fail to achieve properly folded structure; N-linked oligosaccharides are timers; mannosidase trims mannose, recognized by ER lectins (slower proteins at risk)
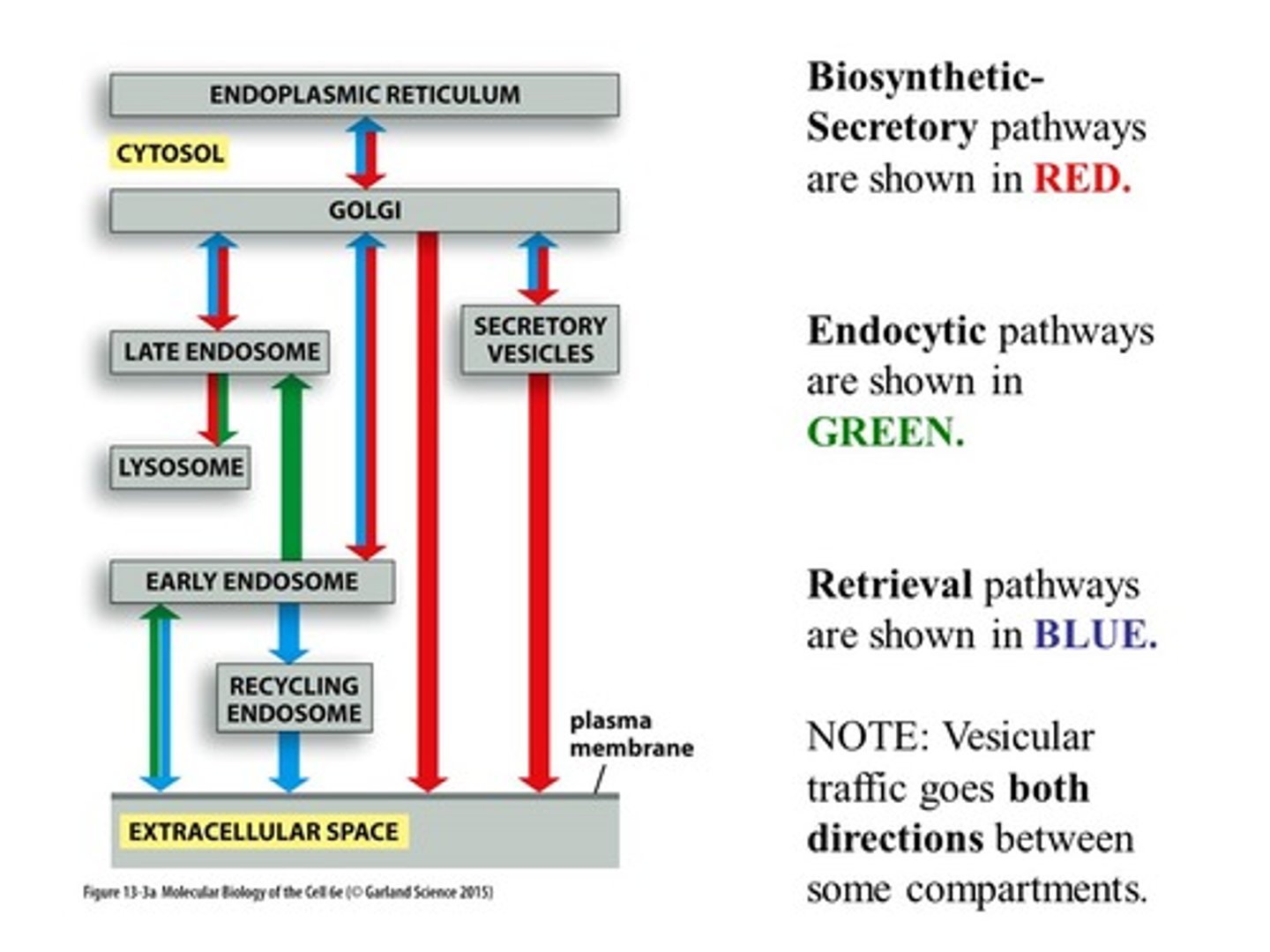
List and describe 3 pathways of vesicular transport.
1. Secretory pathway - from ER to Golgi and cell surface, with side route to lysosomes
2. Endocytic pathway - inward from plasma membrane
3. Retrieval pathways - balance flow of membrane between compartments (opposite direction; recycling)
Describe the structure of the Golgi Apparatus (ie cis, medial, and trans face).
Vesicles enter cis face and exit trans face; medial cisterna are flat pancakes in between
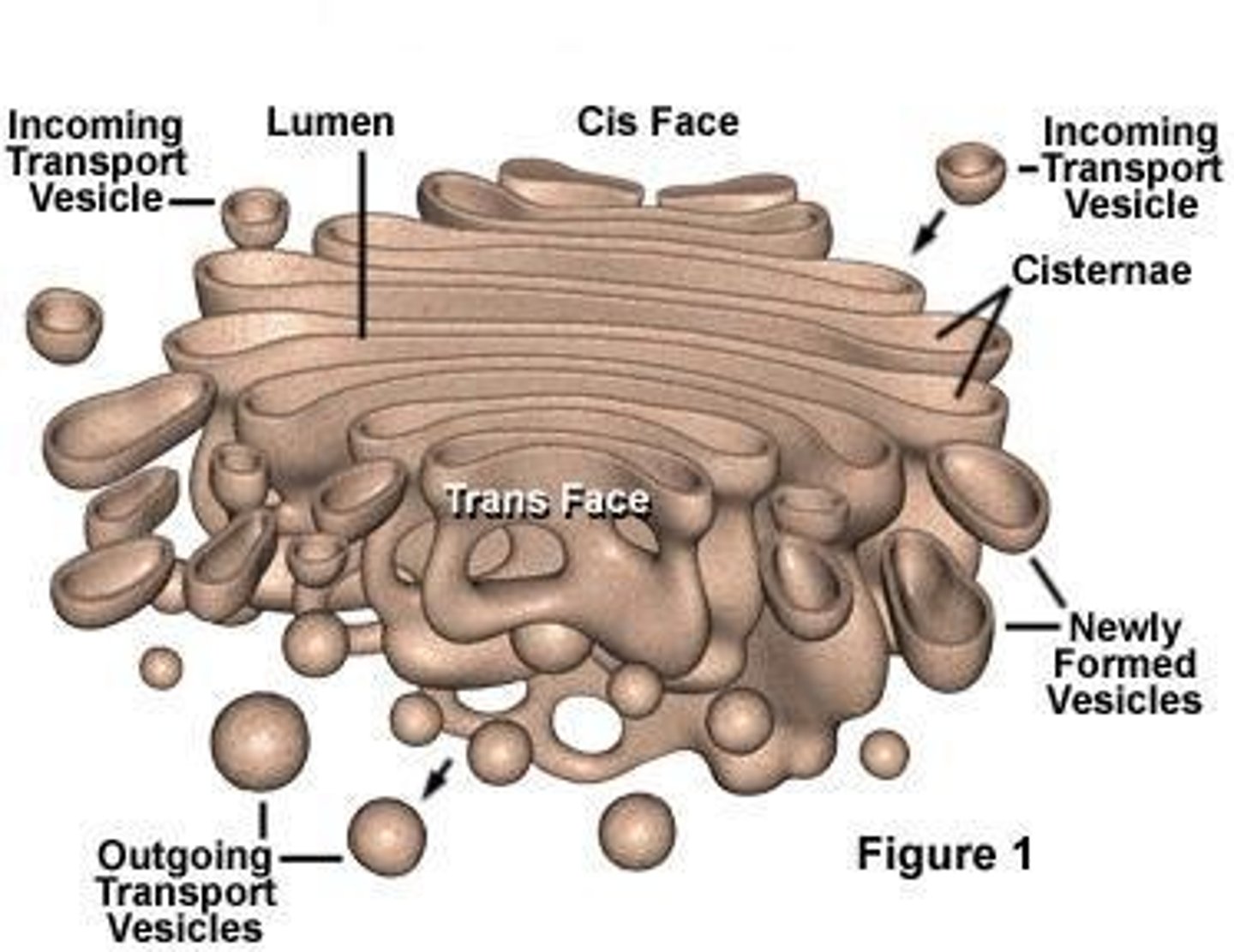
What are the main functions of the Golgi Apparatus?
Transporting, modifying, and packaging proteins and lipids into vesicles for delivery to targeted destinations; glycoproteins formed
What are the 3 types of coated vesicles (clathrin coat, COPI, and COPII)? What pathways are each found in? Key players in coat assembly and vesicle budding
1. Clathrin - endocytic; from PM and between endosomal and Golgi compartments
2. COPI - retrieval/recycling; Golgi to ER
3. COPII - secretory; ER to Golgi
What are the clathrin triskelions and dynamin?
Triskelions assemble into basketlike framework to form coated pits (buds) on cytosolic surface of membranes; dynamin pulls membranes closer together
What do adaptor proteins (inner coat) do in clathrin coated vesicles?
Form inner layer of the coat between clathrin cage and membrane; bind coat to membrane (bind to PIP) and trap cargo receptors (select for specific set of proteins) and package them into vesicles
Describe components of inner and outer coat layer.
Inner coat: allows specificity (concentrates specific membrane proteins in patch, selects membrane molecules for transport
Outer coat: shapes the vesicle (coat assembles into basketlike lattice, deforms the membrane patch and shapes the vesicle)
What is the role of coat recruitment GTPases (ARF and Sar)?
ARF: assemble COPI and clathrin coats at Golgi
Sar1: assemble COPII coats at ER membrane
Describe the detailed role of Sar in both coat assembly and disassembly in COPII coated vesicles.
Assembly:
1. Inactive SAR1-GDP binds to a Sar1-GEF in ER, now SAR binds to GTP; GTP triggered conf. change in Sar1, exposes amphiphilic helix, which inserts into ER membrane (initiates membrane bending)
2. GTP-Sar1 binds to 2 adaptor proteins (Sec 23/24) which form inner coat
3. Complex of 2 additional coat proteins form the outer shell (assemble into cages like clathrin)
Disassembly:
GDP-bound Sar by GAP
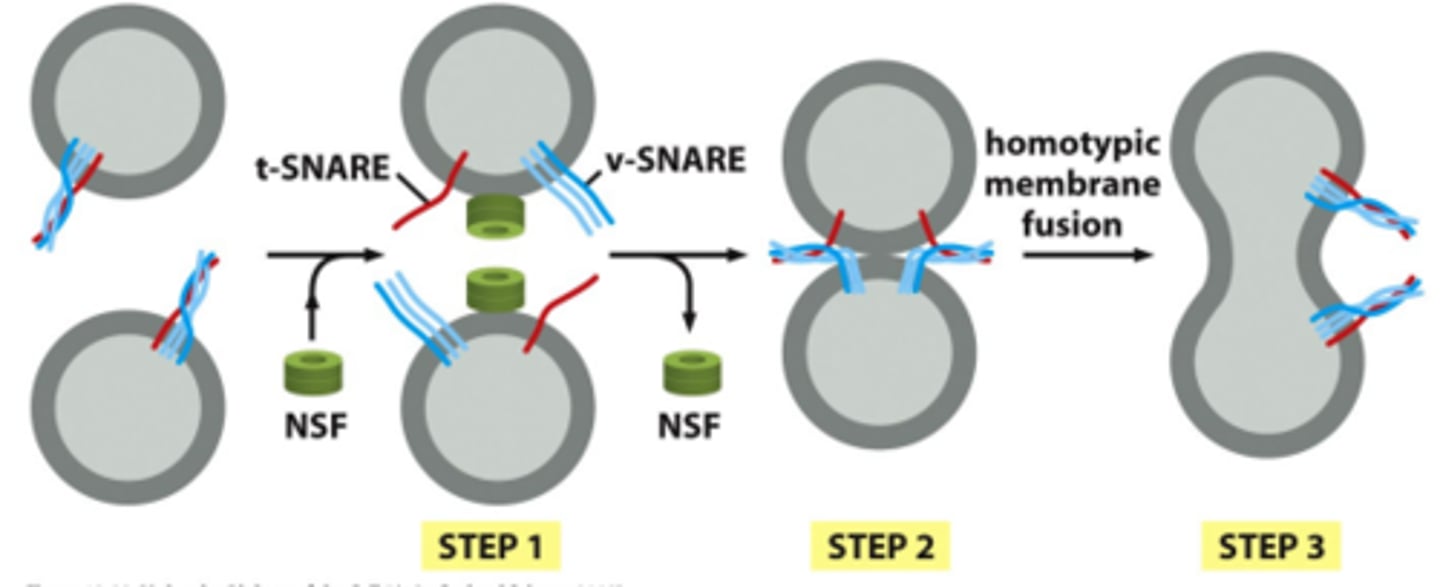
What is the process of membrane fusion? Include the function of Rab and Rab effectors, SNAREs, and NSF.
Rab proteins - surface markers that help guide vesicle to membrane; direction
Rab effector - tethering protein; helps reel it in
SNAREs - t-snares and v-snares interact; overcome hydration barrier to mediate fusion by using energy released from helices wrapped around each other
NSF - disassembles snare complex
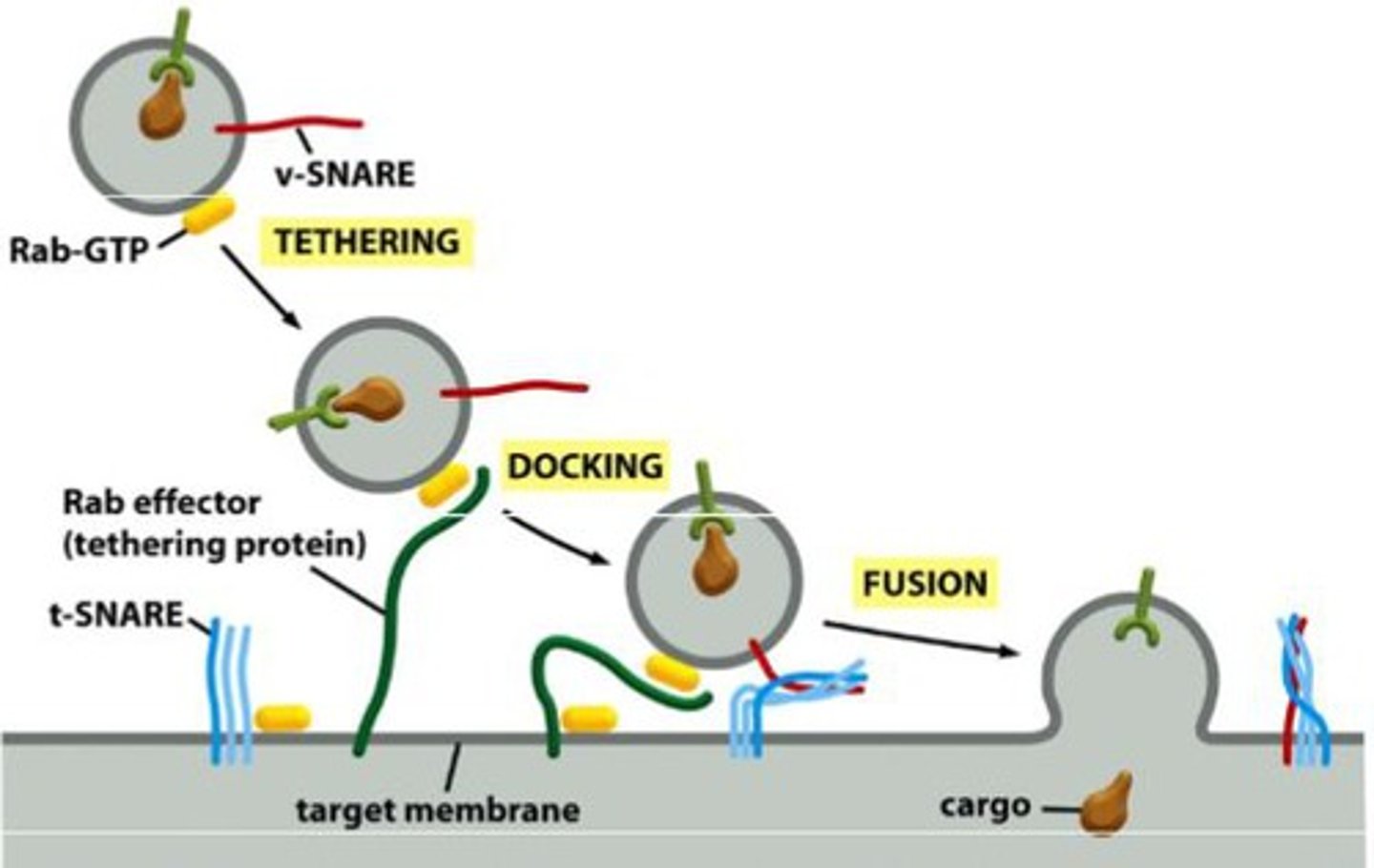
Describe Rab as a GTPase.
-GDP-Rab is inactive and bound to another protein, GDP (Rab-GDP dissociation inhibitor), protein is soluble
-GTP-Rab is active and associated with membrane of organelle or transport vesicle
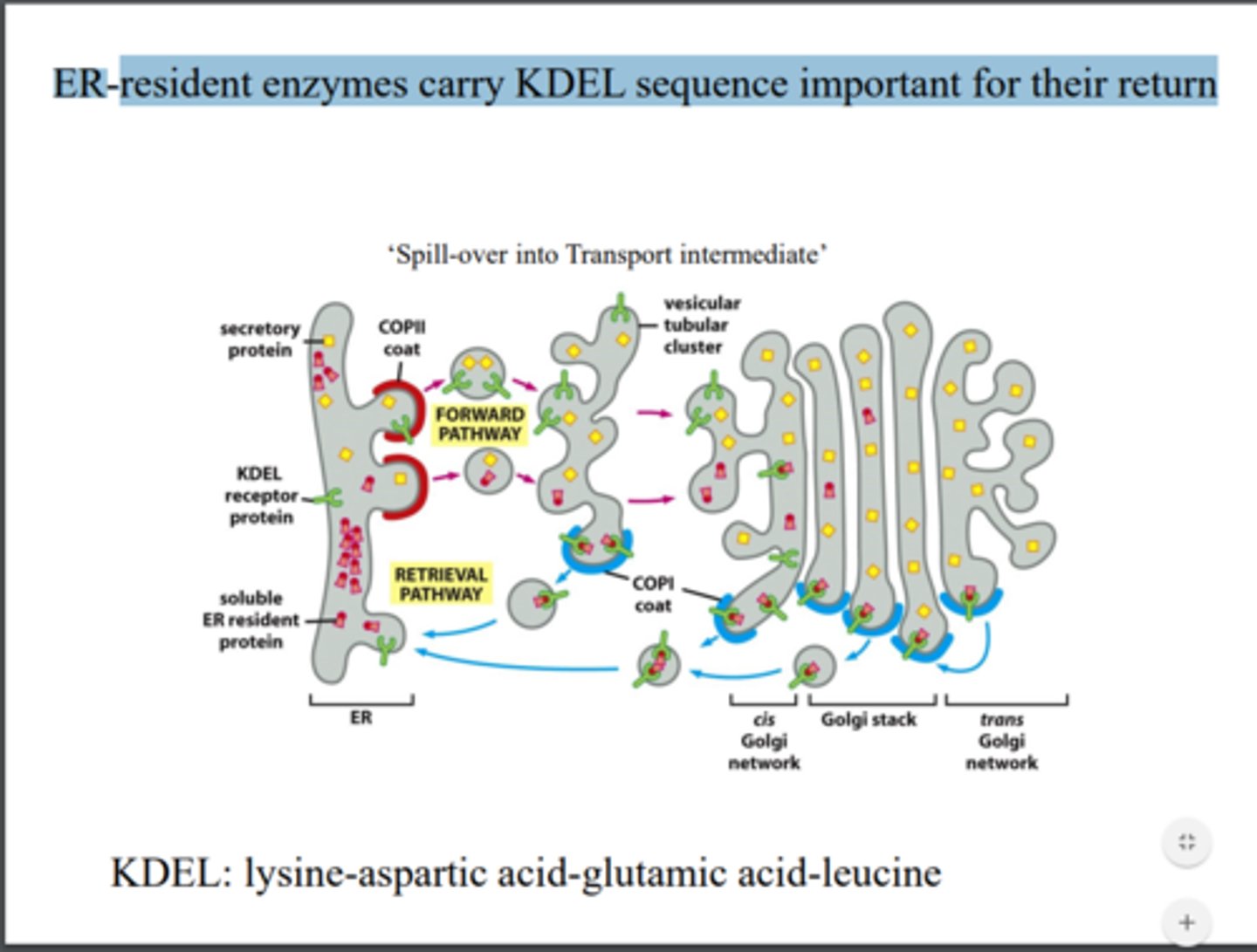
What are exit signals? Do resident ER proteins have exit signals?
No but they have retrieval mechanisms (COPI coated vesicles) like KDEL sequence
What is homotypic fusion and when is this observed?
Fusion of the same vesicle membranes
Retrieval pathway: Describe different affinities of KDEL sequence and KDEL receptor in Golgi vs. ER.
Higher affinity of KDEL in Golgi to capture them and bring them back to ER (microenvironment in Golgi is more acidic, so stronger association in Golgi and greater disassociation in ER)
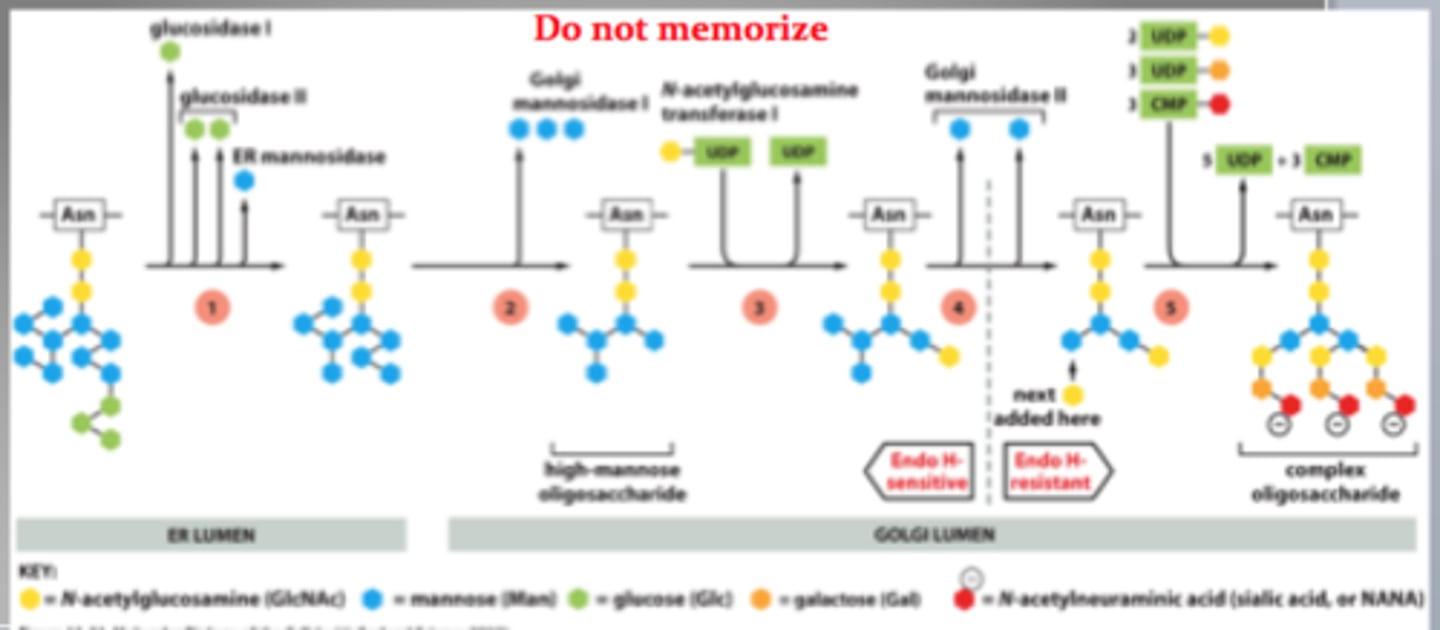
Oligosaccharide processing in Golgi. Why are proteins glycosylated?
Occurs in organized sequence in Golgi stack with each cisternae containing a characteristic mix of processing enzymes
-Promotes protein folding (quality control)
-More resistant to digestion by proteases (protection)
-Cell adhesion (selectins)
-Immune function
-Regulatory role
What is the purpose of Endo H?
Bacterial toxin that cleaves off sugar between 2 GlcNac sugars, if specific mannose is present
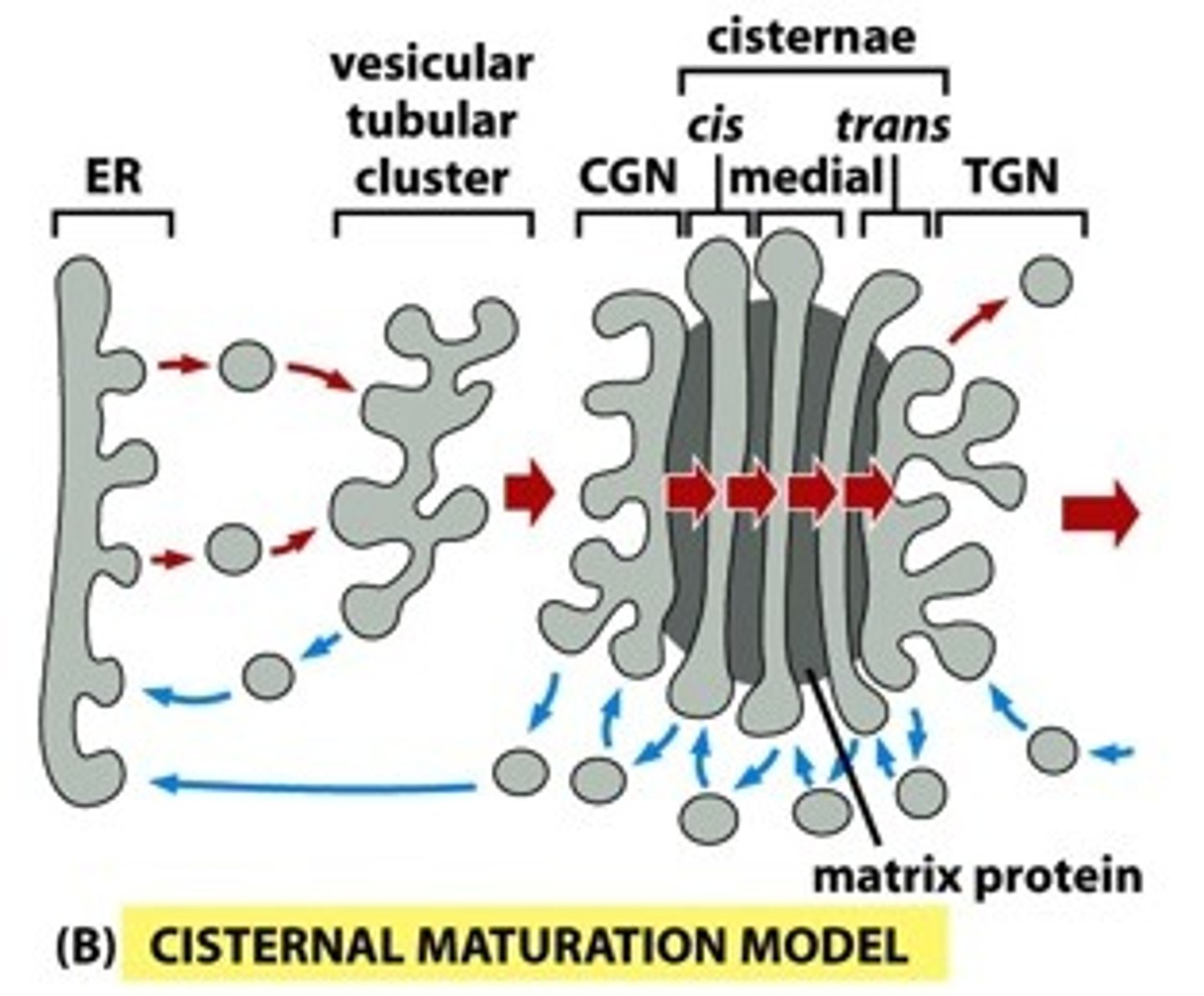
Describe the cisternal maturation and vesicle transport model of transport through the Golgi Apparatus. Compare and contrast forward movement of proteins across golgi stack.
Cisternal maturation: compartments change/evolve; cis -> medial -> trans); newly formed cis cisterna need the right enzymes, so there is retrograde transport from medial to cis
Vesicle transport: each cisterna is stationary and the cargo itself moves forward (retrograde transport just to return enzymes)
What is the function of the lysosome? How is acidic environment maintained? What is the role of acid hydrolases?
Intracellular digestion; V-type pumps acidify lumen; hydrolyses break down macromolecules such as nucleic acids, proteins, and polysaccharides
Why are membrane proteins glycosylated?
Protective function; prevent hydrolyses from leaking out
Why do we call lysosomes heterogeneous?
They vary dramatically in size and morphology
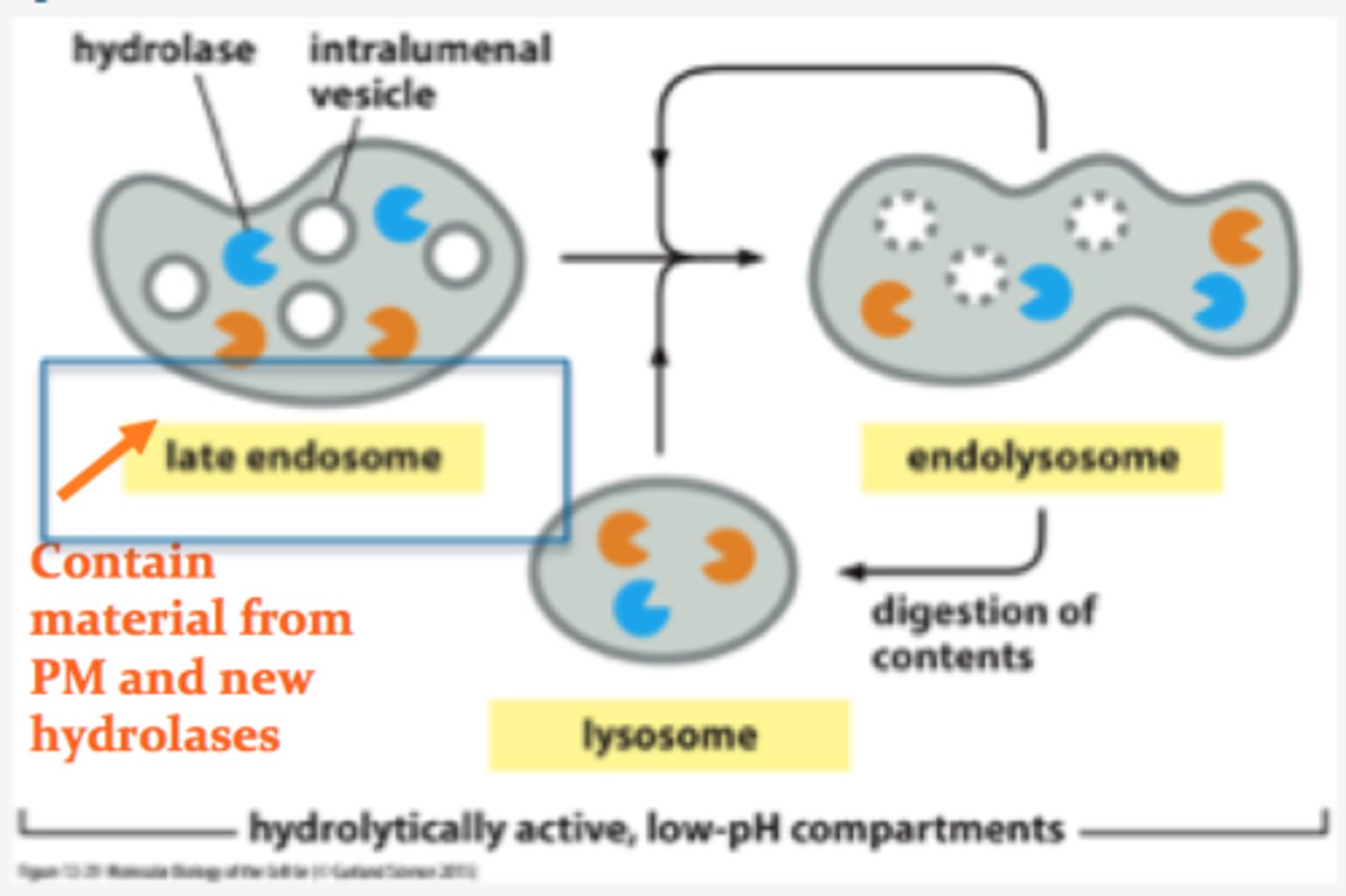
How are endolysosomes formed? When do they mature into lysosomes?
-Late endoscope and lysosome fuse to form endolysosome
--Endolysosomes mature into lysosomes once hydrolases complete digestion
What are the four pathways to degradation in lysosomes (endocytosis, phagocytosis, macropinocytosis, and autophagy)?
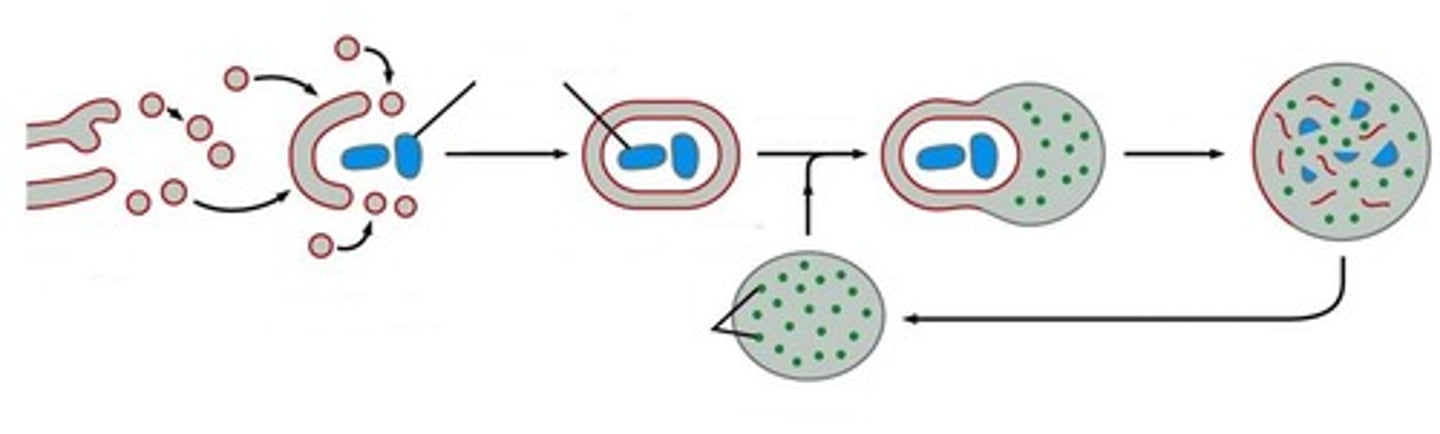
Describe the process of autophagy (as outlined in Figure 13. 43). Why is autophagy important?
1. Starts with a signaling pathway, which starts nucleation
2. Vesicles fuse into a membrane around autophagosome
3. Autophagosome fuses with lysosomes with acid hydrolases that digest its content
-Important in growth and development (helps restructure differentiating cells), can digest large objects that the proteasome cannot handle
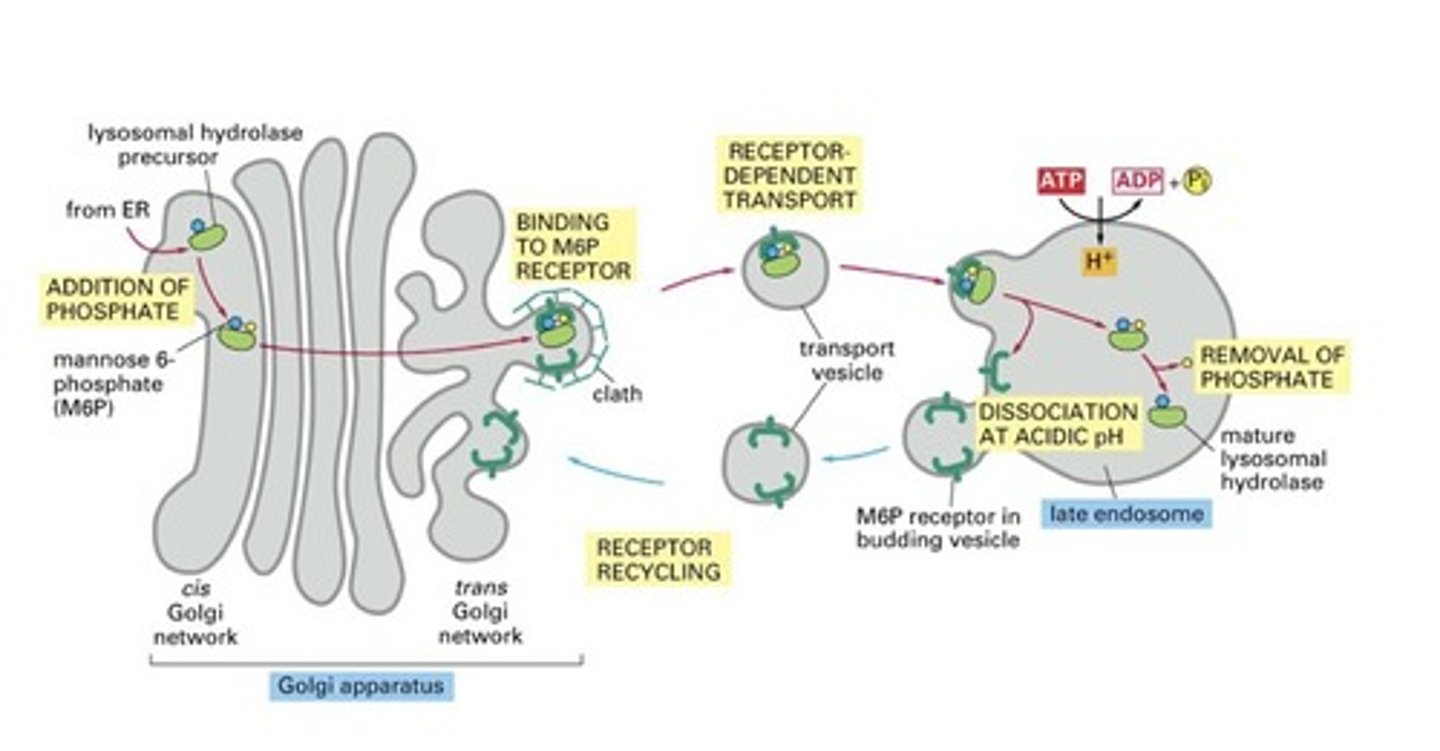
How are lysosomal hydrolases recognized and selected in the Trans Golgi? What happens in the early endosome?
-Mannose 6 phosphate (M6P); glycosylation important for sorting; adding M6P in Golgi tags this hydrolase
-In Golgi, M6P is modified and then recognized by M6P receptor, which bind to the lysosomal hydrolases on the lumenal side of the membrane and to adaptor proteins in assembling clathrin coats on the cytosolic side
-Acidic pH of endosome causes hydrolyses to dissociate from M6P receptor
Describe mutation associated with I-cell disease.
Most hydrolases are missing from lysosomes due to defect in GlcNAc phosphotransferase (no M6P tag, so M6P receptors fail to sort and target hydrolases lacking tag)
-Hydrolases secreted (found in blood)
What do most endocytic vesicles fuse with, near the cell periphery?
Endocytic vesicles fuse with receiving compartment (early endosome), where internalized cargo is sorted
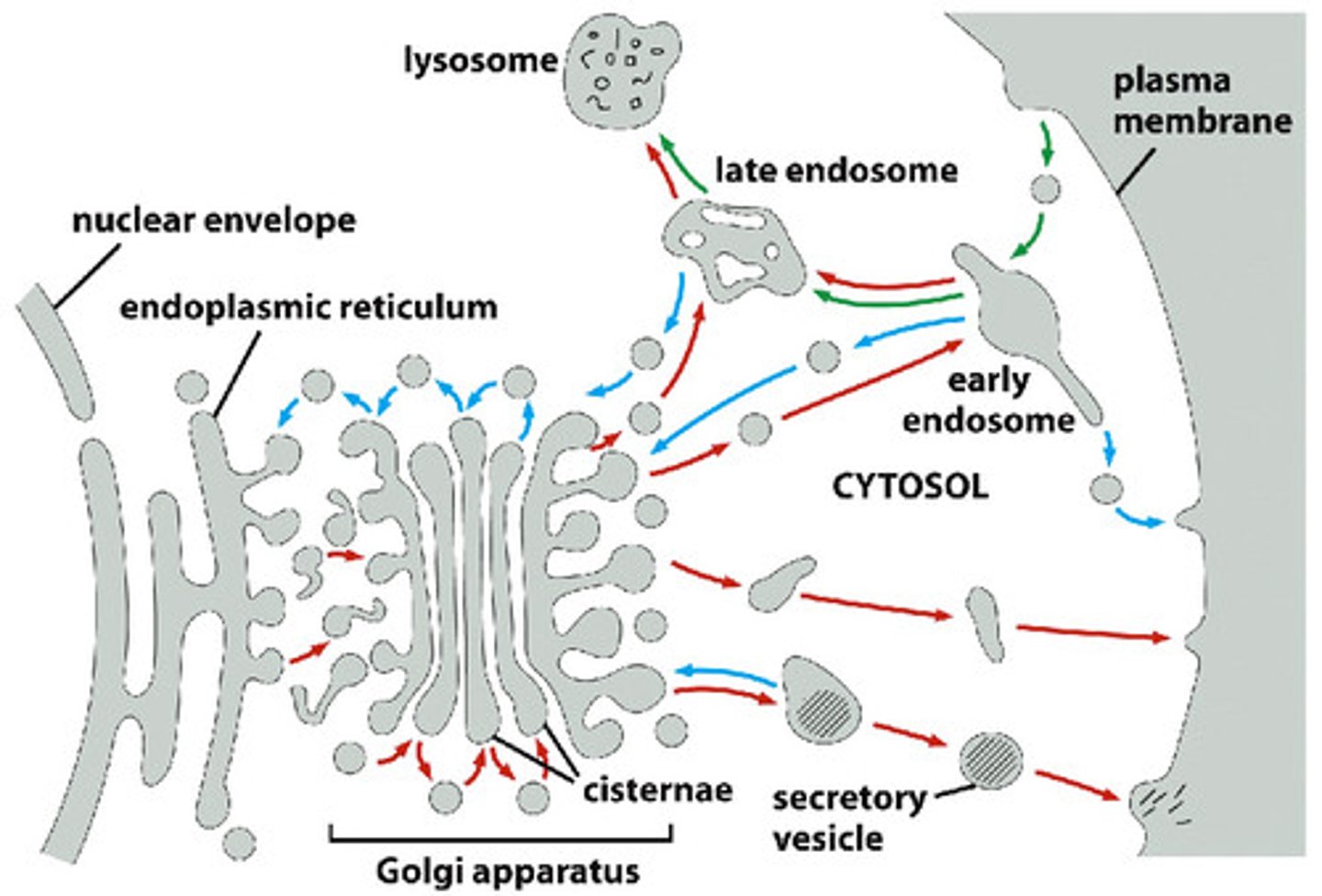
Where do early endosomes come from? What happens to early endosomes?
-Early endosomes come from fusion of endocytic vesicles with receiving compartment (sorted)
--Early endosomes go to PM (recycling endosome) or designated for degradation (late endosome)
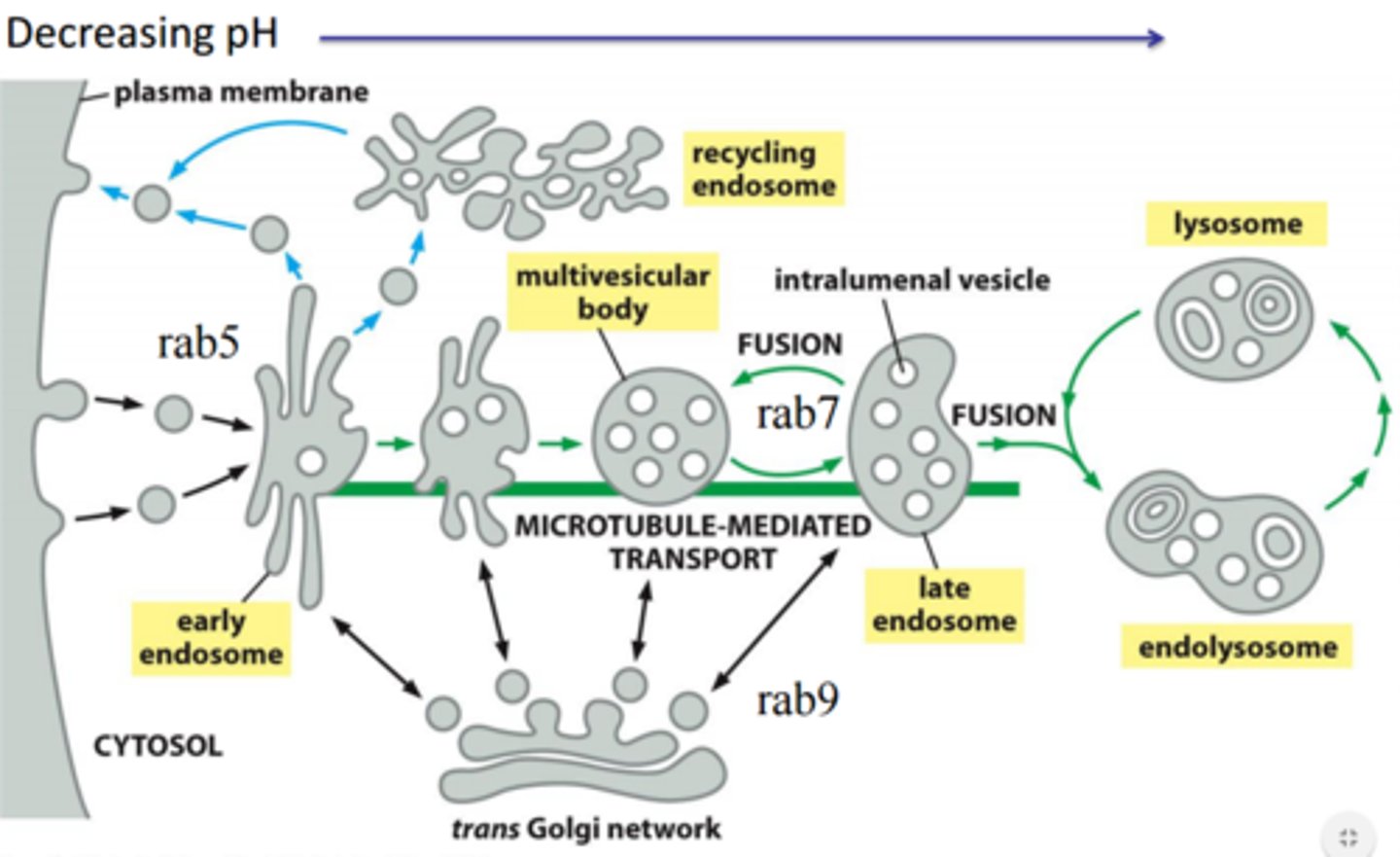
Describe endosome maturation (specific changes that take place), and destiny of cargo within the late endosome.
-Lose tubular projections
-More vesicles
-Location change further into cell interior
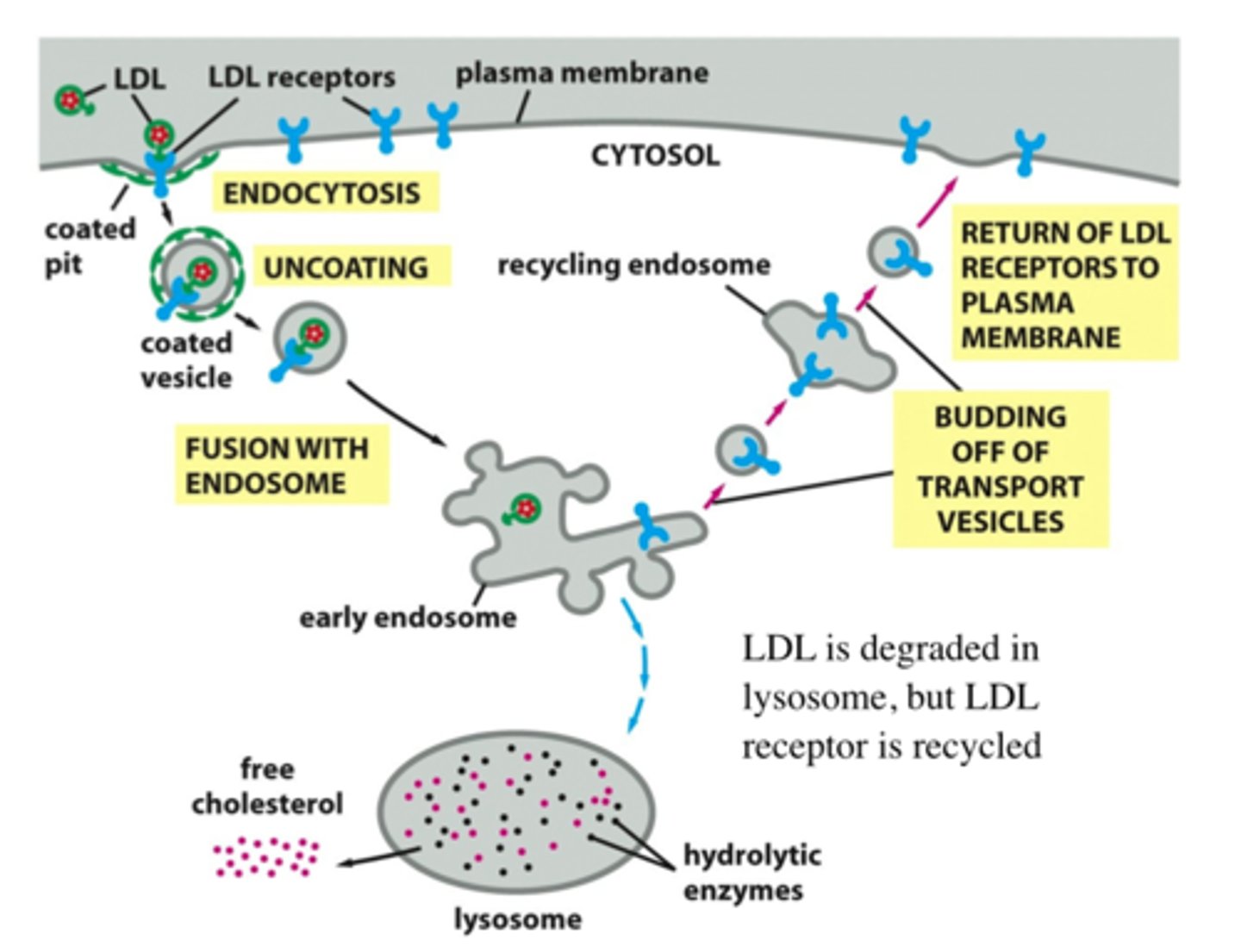
List steps of receptor-mediated endocytosis of LDL (detailed steps).
LDL captured by receptors, clathrin coat assembled, uncoated, fusion of vesicle with early endosome (LDL receptor returns to plasma membrane), LDL inside early endosome --> late endosome + lysosome, cholesterol is freed and LDL is destroyed
Define exocytosis.
Fusion of vesicles with the plasma membrane
What is the constitutive secretory pathway? What kinds of cells use the regulated secretory pathway?
"Default" pathway; in unpolarized cells, it delivers proteins with no special features to the cell surface
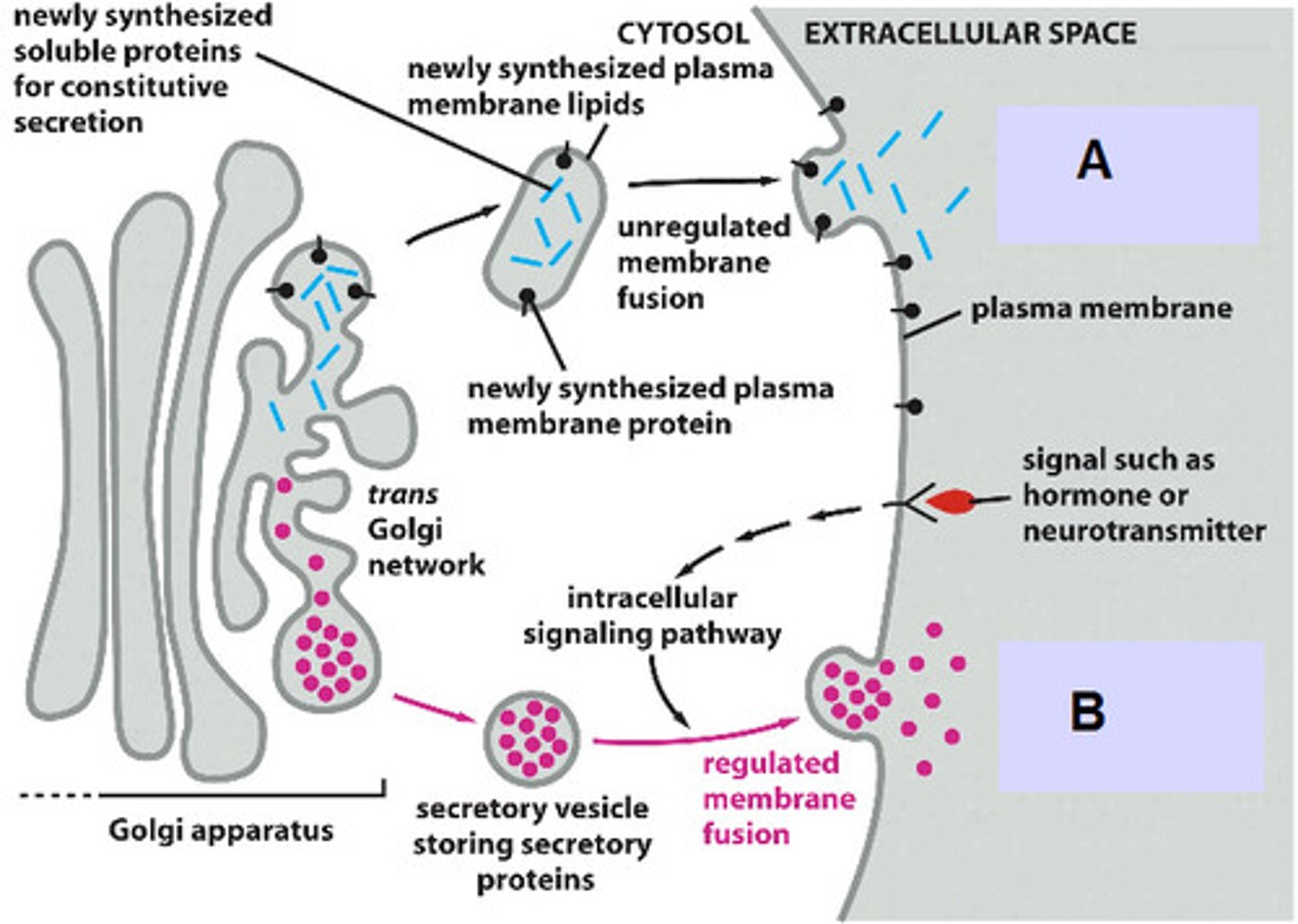
List 3 pathways of protein sorting in trans Golgi network.
1. Signal-mediated diversion to lysosomes (via endosomes)
2. Signal-mediated diversion to secretory vesicles (for regulated secretion)
3. Constitutive secretory pathway (not regulated)
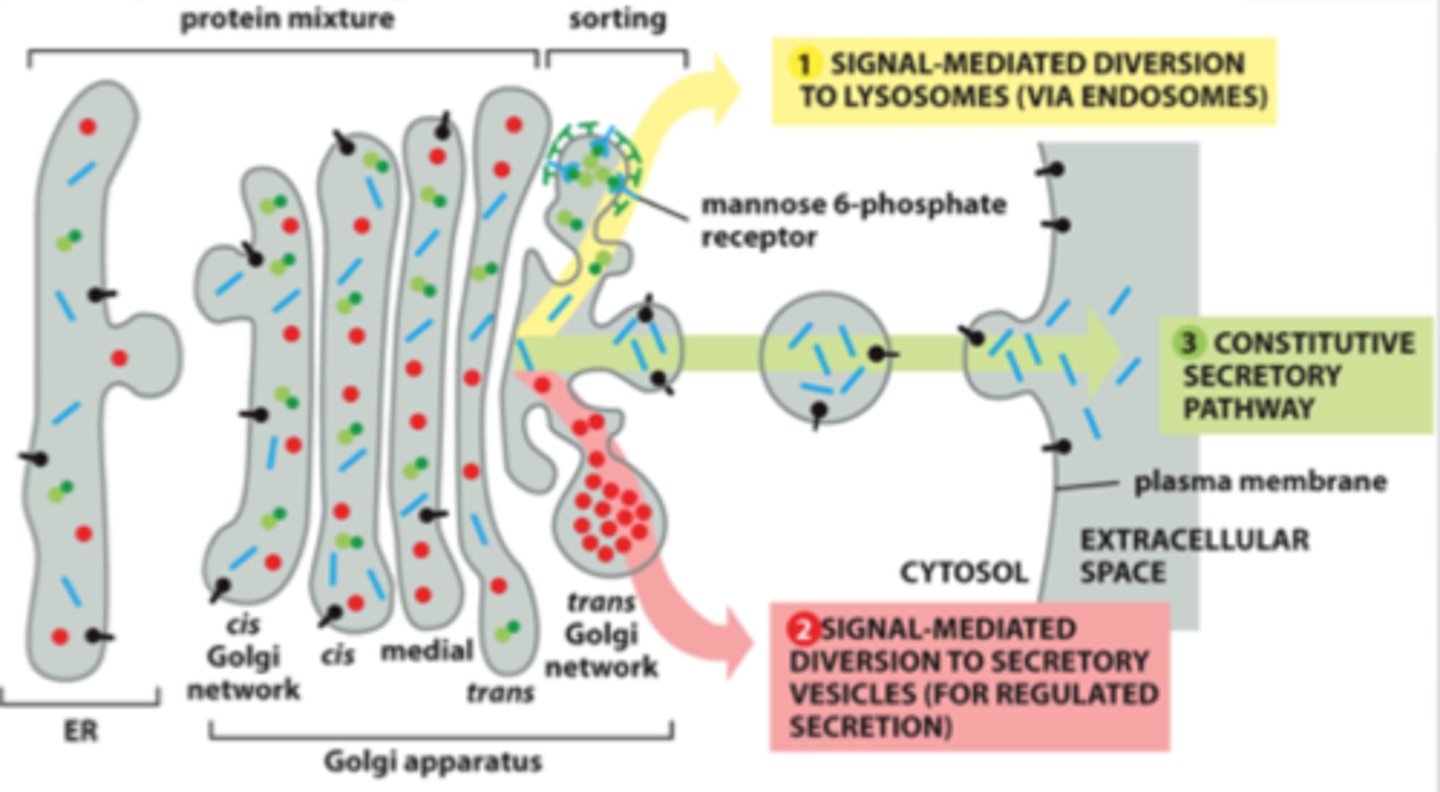
In regulated secretion, when do secretory vesicles release their contents?
Proteins are concentrated and stored until an extracellular signal stimulates their secretion (ex. neurotransmitters/hormones)
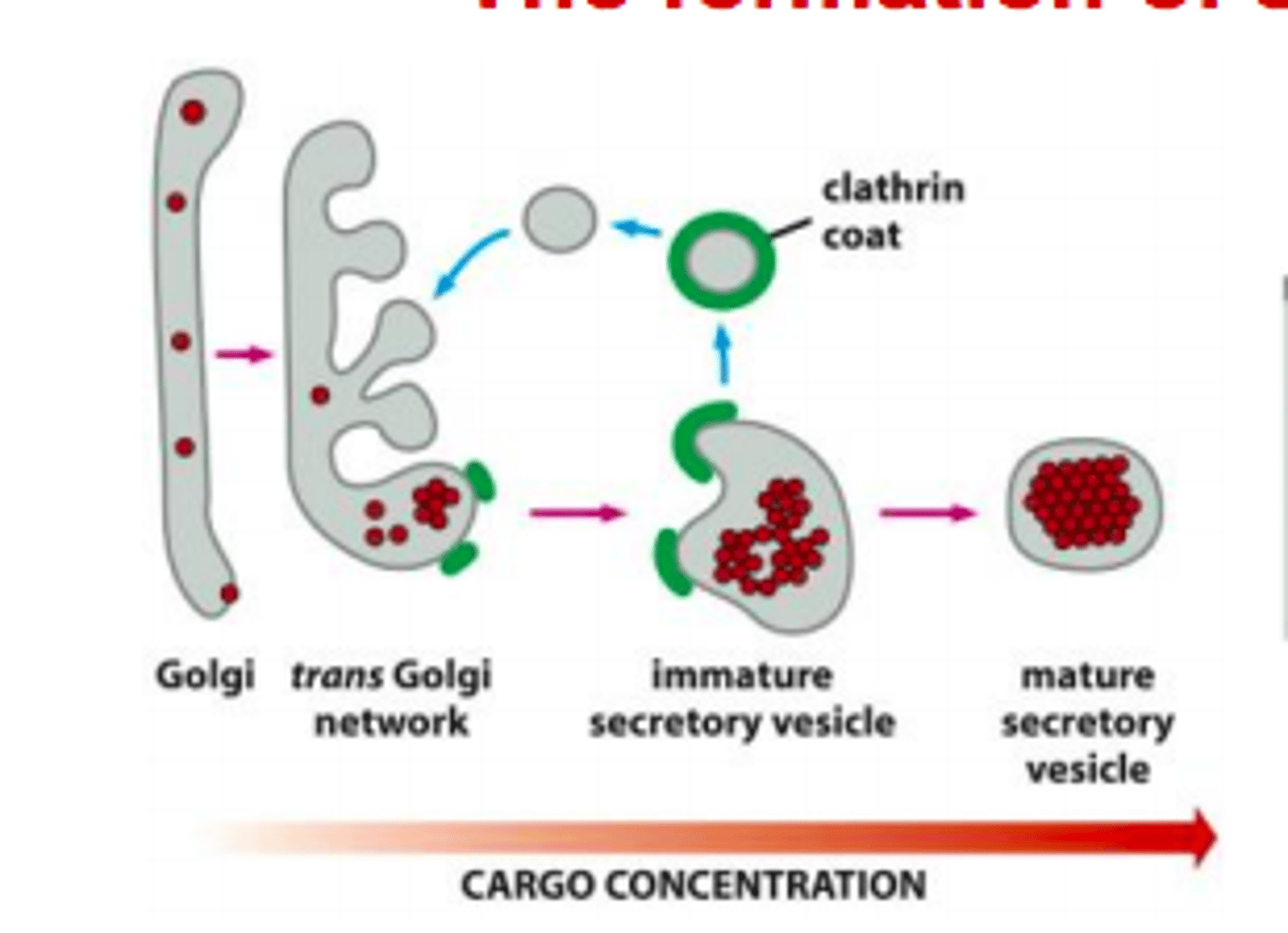
How do secretory protein become segregated and highly concentrated in secretory vesicles? Describe formation of secretory vesicle.
1. Clathrin coated vesicles retrieve membrane and luminal (soluble) content back to Golgi
2. Secretory proteins aggregate in acidic environment
3. Vesicles fuse
Why is proteolytic processing so common in secretory pathway?
-Some peptides are too short in their mature forms to be co-translationally transported into the ER lumen or to include the necessary signal for packaging into secretory vesicles
-Prevents protein from being active too early (digestive enzymes)
What do synaptic vesicles (specialized secretory vesicles) store?
Neurotransmitters
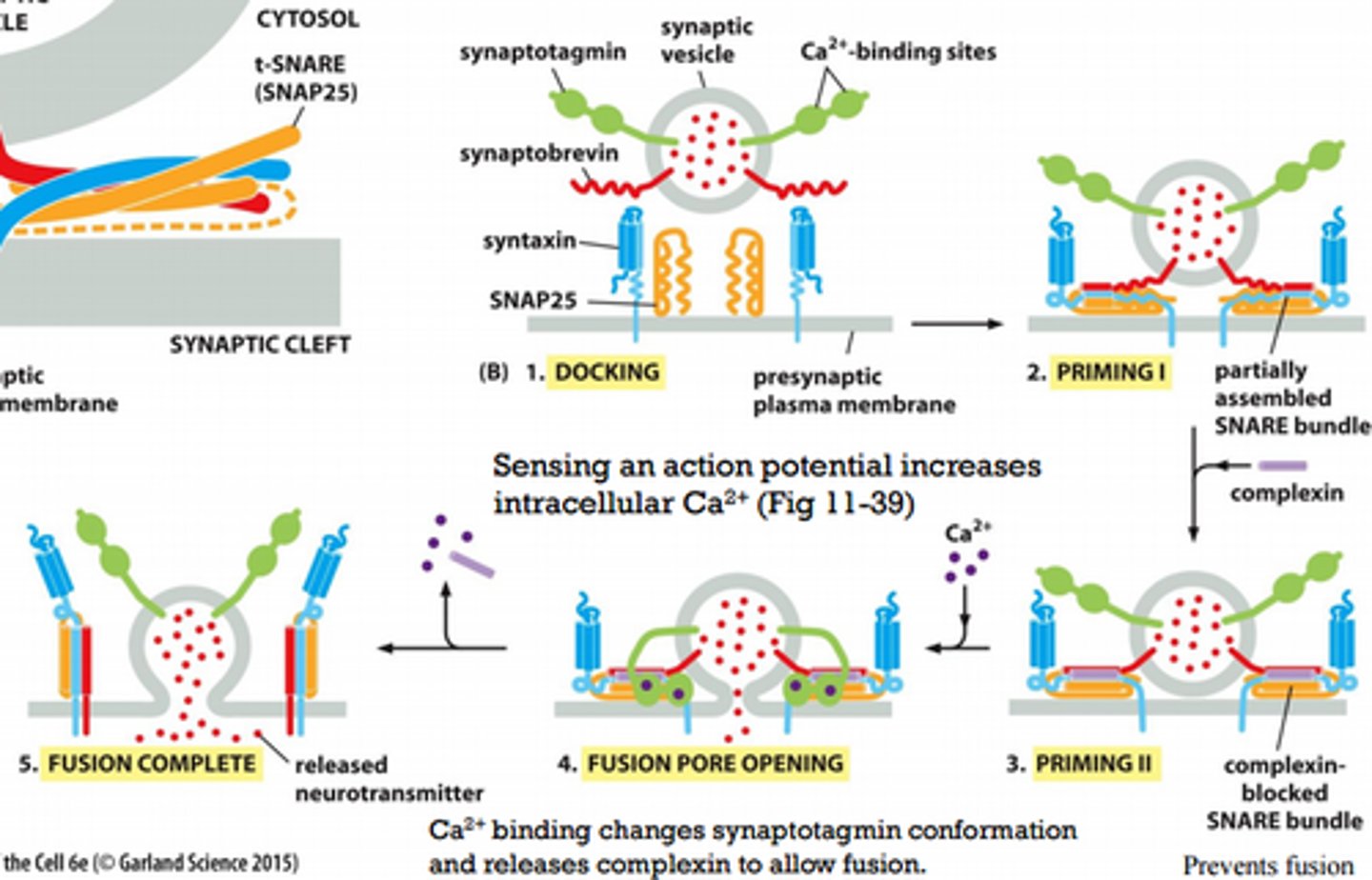
Describe steps involved in exocytosis of synaptic vesicles (detailed, Figure 13.67).
1. Synaptic vesicles dock at the membrane
2. SNARE bundle (1 v-snare, 3 t-snares), helices not fully wound partially assembles, resulting in a "primed vesicle" that is already drawn close to the membrane
3. SNARE bundle assembles further but complexin (brake pedal) prevents fusion
4. Upon arrival of an action potential, Ca2+ enters the cell and binds to synaptotagmin, which releases complexin and opens a fusion pore
5. Further rearrangements complete the fusion reaction and release the fusion machinery, which now can be reused
Describe mechanism of botulinum toxin.
Disassembles SNARE complex due to light chain activity (prevents presynaptic vesicle from fusing)
What is the cause of Niemann Pick Type C?
Mutation of NPC1 or NPC2 gene causes cholesterol to accumulate in the lysosome and not be used for membrane insertion
What are the symptoms, diagnosis, and treatment of Niemann Pick Type C?
Symptoms: lose intellectual function, clumsiness, spleen/liver enlargement, sudden loss of muscle strength; fatal, neurodegenerative disease (loss of myelin sheath)
Treatment: cyclodextrin (cholesterol lowering drug) or histone deacetylase inhibitors (increase gene expression of NPC1 protein), gene therapy
What is the connection between NPC and Ebola?
NPC1 cholesterol transporter is essential for Ebola virus infection