BIO 4127 - 5 Insulin and Glucagon
1/112
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
113 Terms
Why is it important to tightly control glucose levels in the body?
- glucose is key energy source for cellular function
- brain cannot synthesize or store glucose (too much or too little can be rapidly lethal)
How does the pancreas develop?
it develops as a glandular outgrowth of the intestine
2 parts of the pancreas
endocrine and exocrine
Endocrine pancreas
produce peptide hormones which are secreted into the blood
Cells of the endocrine pancreas (4)
Alpha cells
Beta cells
Delta cells
PP cells
Alpha cells secrete..?
glucagon
Beta cells secrete..?
insulin
Delta cells secrete...?
somatostatin
PP cells secrete...?
pancreatic polypeptide
Exocrine pancreas
digestive enzymes secreted into pancreatic duct leading to small intestine
Enzymes secreted by the exocrine pancreas
amylase (glycogen breakdown)
lipase (triglyceride breakdown)
trypsin/chymotrypsin and carboxypeptidase (protein breakdown)
The endocrine pancreas is made up of many?
islets of Langerhans
What is an Islet of Langerhans
the functional unit of the endocrine pancreas. Contains alpha, beta, delta, and PP cells.
Acinar cells
enzyme-secreting cells of the exocrine pancreas
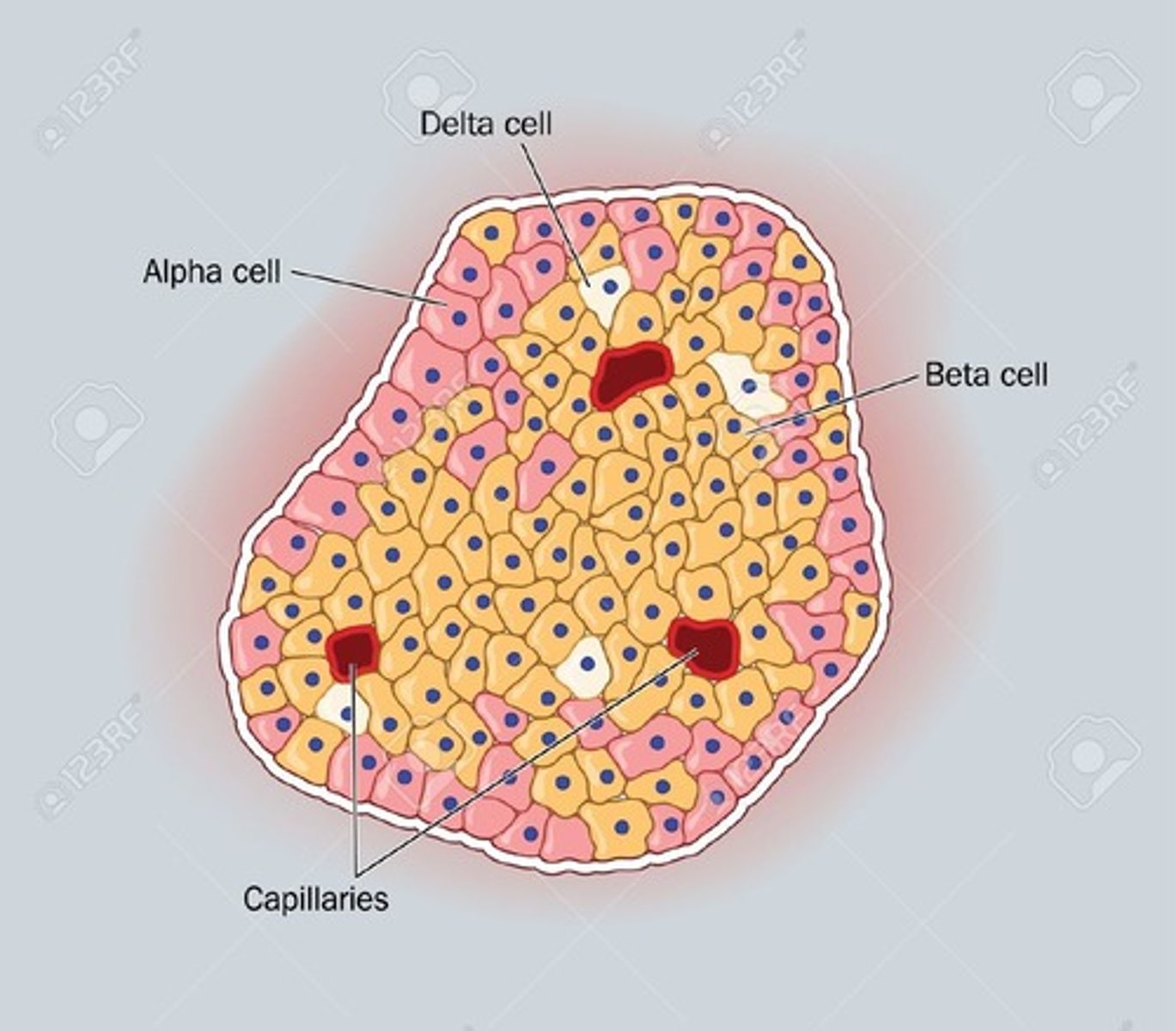
Islet of Langerhans organization
Insulin secreting beta cells are in the middle of the islet and the other types of cells are on the periphery
Appearance of endocrine pancreas cells along the vertebrate evolutionary tree
over 400 million years ago; these cells are ancient
Pancreatic hormones (4)
- Insulin
- Glucagon
- Somatostatin (SST, SRIF)
- Pancreatic polypeptide
Insulin
- rapidly secreted from beta-cells when plasma glucose increases
- acts on many cells (e.g., liver, muscle, adipose)
Insulin function
Increases glucose uptake and conversion of glucose to glycogen
Glucagon
29 amino acids, single chain peptide that stimulates hepatic glucose synthesis and release
Glucagon function
- Stimulates hepatic glucose synthesis and release
- stimulates conversion of amino acids to glucose, and stimulates lipolysis
Somatostatin (SST, SRIF)
prohormone (28 a.a.) but active peptide is 14 a.a. (1 cysteine bond)
- paracrine mode of action
Somatostatin function
- inhibits secretion of insulin and glucagon from pancreas
- inhibits gastric function (inhibits acid secretion, H2CO3 -, and pancreatic enzyme secretion)
Pancreatic polypeptide
- 37 a.a. in human, 36 a.a. in rat
- single peptide
- Not well studied
Pancreatic polypeptide function
decreases liver glycogen and pancreatic secretion
Oral glucose tolerance test (OGTT)
Given a certain amount of glucose to ingest, then blood samples are taken periodically to assess your clearance of glucose (activity of your beta-cells)
Normal blood sugar level before a meal (fasting)
4-7mmol/L
As blood glucose levels increase, what happens to glucagon?
glucagon secretion is shut off
As blood glucose levels increase, what happens to insulin?
insulin secretion increases
Human insulin gene codes for...?
Pre-proinsulin with signal peptides that tells the cell where the polypeptide should go
Insulin biosynthesis is controlled by which molecule?
glucose, so that insulin is only made when needed
Pre-proinsulin to insulin conversion
Proteases remove the signal peptide and fold the remaining polypeptide
Proinsulin to insulin conversion
cleavage of the c-chain
insulin C-chain
Portion of the proinsulin peptide that links the A and B-chain. It is cleaved to form insulin
C-chain cleavage site type
Dibasic cleavage site
How are the A and B insulin chains linked?
by two cysteine bridges
What class of proteases cleave proinsulin to form insulin?
Prohormone convertases
Insulin synthesis steps
- Preproinsulin is secreted into the endoplasmic reticulum
- Post-translational processing cleaves the N-terminal signal sequence and forms the disulfide bridges
- Proinsulin passes through the Golgi and is packaged into vesicles
- PC enzymes within the acidic vesicle cleave proinsulin at two positions
- Secretory vesicle now contains active insulin and free C-chain
Insulin secretion is dependent on which ion?
Calcium
Insulin across evolution
is conserved from protochordates like amphioxus to fish to mammals
Insulin A and B chain conserved residues
A-chain - cysteines at pos. 6, 7, 11, and 20 (considerable variation at 8, 9, 10)
B-chain - cysteines conserved at 7, 19 (variation around position 30)
Fish Pancreas
endocrine and exocrine parts are separate unlike mammals
Endocrine fish pancreas is called the...?
Brockmann body
- location where amino acids will signal insulin secretion and synthesis
Insulin receptor type
tyrosine kinase
- Tetramer
- Two identical α subunits linked via a disulfide bond
- Two beta subunits
Tyrosine kinase domain
Enzyme-like domain that is activated upon insulin binding to receptor. -
What happens to IR when insulin binds IR
IR autophosphorylation: adds a phosphate to tyrosine molecule
What IR subunit does insulin bind?
IR alpha subunits
Insulin receptor activation cascade
phosphorylation cascade
Phosphorylation
the addition of a phosphate group (PO4) to a molecule
Insulin action (insulin triggering IR) steps
1. Mature IR
2. Insulin binds to the alpha subunit of the receptor
3. Autophosphorylation of beta subunits leads to a conformational change in the IR
4. Kinase activity of the IR increases
5. Phosphorylation of intracellular proteins initiates the signal transduction cascade for insulin action
How are the two types of insulin receptors derived?
alternative splicing of mRNA from the IR gene
Types of Insulin receptors
Isoform A and B
IRa binds...?
insulin and insulin-like growth factor 2
IRb binds..?
IRS (insulin receptor substrate) preferentially and insulin
Insulin receptor isoforms affinity
Affinity of IRa for insulin is 2-fold higher (in vivo)
- Preferentially IRa is the target for insulin
- IRb couples more efficiently to signalling substrates IRS-1 (in vivo)
How do cells express insulin receptor isoform?
A cell may express both or either isoforms of insulin
Most insulin signals are produced or modulated through...?
tyrosine phosphorylations
Insulin Receptor Substrate Proteins
IRS1 and IRS2
- crucial cytoplasmic adaptor proteins that transduce signals from insulin and IGF-1 receptors to downstream effectors, regulating growth, metabolism, survival, and differentiation
- They act as docking stations, linking receptor tyrosine kinases to pathways like PI3K, essential for glucose homeostasis
IRS1 function
controls body growth and peripheral insulin action
IRS2 function
regulates brain growth, body weight control, glucose homeostasis, involved in female fertility
What downstream kinase does insulin activate?
PI3 kinase
After PI3 kinase is activated, what other kinases are recruited and where?
- 3-phosphoinositide-dependent protein kinase-1 (PDK1)
- Protein Kinase B (also known as AKT, several forms)
To the inner membrane
PDK1 is activated by steps of phosphorylations, and then PKB is activated
PKB phosphorylates serines in multiple other proteins
What proteins do PKB phosphorylate downstream? (3)
- BAD (BCL2-associated agonist of cell death)
- FOXO-1 (forkhead box O1)
- Glycogen synthase kinase-3 beta
What amino acid does PKB phosphorylate?
serine
Insulin phosphorylation cascade steps
IR tyrosine kinase → PI3 kinase →PDK1 → PKB → BAD + FOXO-1 + Glycogen synthase-3 beta
BAD
Cell survival protein; positively regulates apoptosis
- Member of the BCL-2 family known to be regulators of programmed cell death.
FOXO-1
Transcription factor that promotes β-cell function
- decrease in FOXO-1 inhibits gluconeogenesis and adipocyte differentiation
Glycogen synthase kinase-3 beta
Regulates growth and glycogen synthesis
Excess glucose in blood is stored as...?
glycogen
2 enzymes needed for glucose-glycogen interconversion
- Glycogen synthase
- Glycogen phosphatase
Glycogen synthase
Synthesizes glycogen from glucose
- inactive when phosphorylated
- active when dephosphorylated
Insulin action on glycogen synthase
Insulin de-phosphorylates (activates) glycogen synthase because insulin promotes glucose conversion
Glycogen phosphatase
Removes phosphate groups from glycogen
Insulin effects (phosphorylation)
- stimulate phosphatase
- inhibit phosphorylation
- stimulate glucose transporter (on alpha and beta cells)
Insulin/Glucagon effects (ATP to ADP)
- Insulin inhibits cAMP dependent kinase stopping ATP conversion to ADP
- Glucagon activates adenylate cyclase and stimulates cAMP dependent kinase (promotes ADP → ATP)
Glucagon effects (molecular)
- adenylate cyclase/ cAMP increases
- activation of a cAMP-dependent kinase
- phosphorylation = inactivation of glycogen synthase
What does metabolism of glucose do in beta-cells?
produce various metabolites that will specifically augment insulin secretion
- increase ATP/ADP ratio
- increase intracellular pH
- phospholipid metabolism
Metabolic signals effect on insulin synthesis
- increase insulin transcription,
- Decrease mRNA insulin degradation,
- Increase mRNA translation
Insulin Secretion steps**
- Rise in blood glucose levels
- Uptake of glucose by the GLUT2 transporter of beta-cell
- Glycolytic phosphorylation of glucose causes a rise in the ATP:ADP ratio
- This rise inactivates the potassium channel that depolarizes the membrane (charge in beta cell becomes increasingly positive)
- The calcium channel to open up allowing calcium ions to flow inward
- The rise in levels of calcium leads to the exocytotic release of insulin from their storage granule
IDDM (type 1 Diabetes)
- lack of insulin (beta cells destroyed)
- treat with tissue grafts, add insulin, or stem cell therapies
Why is stem cell therapy for T1D difficult to implement?
The islet of Langerhans is the functional unit (beta cells function best with the other types of cells around) so another therapy is islet of langerhans transplantation (Edmonton protocol)
NIDDM (type 2 Diabetes)
Beta cell insensitivity - many reasons
- insulin autoantibodies, mutant insulin, insulin receptor malfunction, lack of glucose transporters, mutant glucokinase or other enzymes
- treat with drugs to increase insulin sensitivity exercise, diet
T2D risk can increase due to...?
Poor in utero nutrition, obesity, lack of exercise
How can enlarged adipocytes contribute to obesity and diabetes?
In some obese people, enlarged fat cells have reduced IR, so the cells are less likely to respond to insulin
How does the body compensate for diabetes?
Increased insulin secretion, however normal/responsive cells respond to high levels of insulin with receptor down regulation
- Result: hyperglycemia
Nutritional factor controlling an endocrine cell example
Blood glucose levels regulating insulin secretion
Signalling regulation of islets of langerhans
autocrine, paracrine, endocrine, and nutritional signals
Another action insulin has on IR
IR internalization
What is IR internalization?
When insulin binds IR it also promotes the downregulation of IR and some leave the beta-cell surface.
- With too much insulin secretion, IR internalization occurs at such a high level that the cell cannot respond to insulin and glucose is not cleared from the blood.
Primary regulator of insulin secretion
glucose
Other secondary potentiators and inhibitors of insulin secretion origins
- From alpha cell
- From delta cell
- parasympathetic/sympathetic innervation of pancreatic inlet
Pancreastatin effect on insulin release
inhibits insulin release; autocrine (from beta-cell)
Glucagon effect on insulin release
promotes insulin release; paracrine (from alpha cell)
Peptide YY effect on insulin release
inhibits insulin release; paracine (from alpha cell)
Galanin and NPY effect on insulin release
inhibits insulin release; neurocrine (from sympathetic nerves)
ACh, VIP, gastrin releasing peptide effect on insulin release
promotes insulin release; neurocrine (from parasympathetic nerves)
CCK effect on insulin release
promotes insulin release; neuroncrine
Epinephrine effect on insulin release
Can inhibit and promote insulin release; endocrine (from blood)
Gamma aminobutyric acid (GABA) and insulin secretion
GABA inhibits insulin secretion by autocrine feedback
- GABA may have some protective effects on β-cells
What enzyme converts glutamic to GABA?
glutamic acid decarboxylase (GAD)