Unit 7 Matter Concepts 1 & 2
1/26
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
27 Terms
Matter
anything that takes up space and has mass
Substance
all particles in matter are identical (fixed composition)
Mixture
two or more substances physically combined (variable composition)
Element
simplest form of matter from which more complex substances are made (listed on the periodic table)
Compound
two or more elements chemically combine in a fixed proportion
Homogeneous Mixture
components are evenly distributed out on a microscopic level
Heterogeneous Mixture
substances in the mixture are not evenly spread out
What is the difference between a compound and a mixture?
a compound is when the two or more elements are chemically combined in a fixed proportion while a mixture is when the substances are physically combined
draw a flow chart to represent how matter can be classified
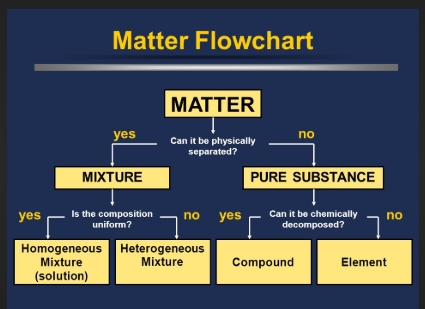
Classify each example below as the type of matter it is (element, compound, homogeneous mixture or heterogeneous mixture)
table salt - compound
milk - heterogeneous mixture
oxygen - element
lemonade - homogeneous mixture
water - compound
carbon dioxide - compound
vegetable soup - heterogeneous mixture
peanut butter - heterogeneous mixture
melting point
temperature at which a solid becomes a liquid
Boiling Point
temperature at which a liquid becomes a gas
Viscosity
a fluid’s resistance to flow
Solubility
a measure of how well a solute dissolves in a solvent
Solute
what is being dissolved (ex. lemonade power)
Solvent
what is doing the dissolving (ex. water)
Solution
the result (ex. lemonade)
density
measure of compactness or consistency of a material
combustibility
how easily a substance will set on fire
ex. burning charcoal
reactivity
how easily a substance reacts, usually with an acid or base
oxidation
how easily a substance reacts with oxygen
chemical reaction
process by which the atoms of one or more substances are rearranged to form different substances
law of conservation of matter
matter is neither created or destroyed during a chemical change. It only changes forms.
Physical properties
properties that can be observed directly or measured without changing the chemical identity of the substance
ex. mass, volume, shape, size, color, luster, hardness, etc.
Chemical properties
properties that can only be observed or measured by changing the chemical identity of a substance
ex. toxicity, flammability, combustibility, radioactivity, etc.
Explain the difference between physical and chemical changes
chemical changes the identity of the substance, physical do not
List evidence that a chemical change, and thus a chemical reaction has occurred
release of light, temperature change, odor change, sudden color change, gas given off