AP Chemistry Unit 9
1/73
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
74 Terms
What is entropy are represented using what?
S
What is Entropy?
The measure of energy dispersal, disorder or randomness, and a number of possible arrangements(microstates).
More possible arrangements mean what specifically?
Higher possibility entropy explains why processes happen in one direction
What do systems tend towards?
Higher entropy
What is a system?
the specific portion of the universe—such as a reaction mixture, beaker, or set of atoms—that is currently being studied or observed
What’s included in organized molecules?
Organized molecules, low entropy, and only one arrangement
What’s included in disorganized molecules?
High entropy, more arrangements, and greater possibility
What are microstates?
A specific arrangement of particles or energy in a system
In an ordered system, where the molecules are organized, are there few or lots of micro-states?
Few micro-states
In an dis-ordered system, where the molecules are un-organized, are there few or lots of micro states?
Lots of microstates
What does the Second Law of Thermodynamics state?
That the entropy of the universe will always increase
Natural processes tend toward’s what disorder?
A greater disorder
As time goes on, what happens to energy?
It spreads out
What do systems tend to move toward?
More probable states
What are examples of entropy spreading out?
Ice melting, Gas expanding, and perfumes spreading through a room
What does Entropy include?
Disorder and energy distribution
As energy naturally spreads out, what happens to entropy?
It increases
What is a spontaneous process?
it’s a process that occurs without continuous outside energy input
Does a spontaneous process mean that it’s fast, True or False?
False because it only measures the energy input
Examples of spontaneous processes
Gas expansions
What is entropy in physical processes?
the measure of a system's molecular disorder, randomness, or energy dispersal
When the volume increases, what does it mean about the space and arrangements of molecules?
There’s more space and more arrangements
When the temperature increases, what happens to Kinetic energy, collisions, energy distributions, and phase changes
Kinetic energy, collisions, energy distributions increase and the phase changes towards gases
What phase do you need to calculate entropy?
Gases
What is the definition of Entropy Change(s)?
the measure of the difference in a system's molecular disorder or randomness between its final state (Sf) and initial state (Si)
What is the entropy change equation of a reaction?
Delta S degree reaction = sum of S degree products - sum of S degrees of reactants
When Delta S is positive, what happens to entropy?
It increase
When Delta S is negative, what happens to entropy?
Entropy decreases
What is the equation to Delta S degrees universe?
Delta S(system) - Delta S(surroundings)
What does it mean when Delta G is less than 0?
It’s spontaneous(exergonic)
What does it mean when Delta G is greater than 0?
It’s non-spontaneous(endergonic)
What does it mean when Delta G is 0?
It’s at equillibrium
What does Delta H in a Gibbs Free energy equation?
Enthalpy change
What does Delta S in a Gibbs Free energy equation?
Entropy Change
What is the Gibbs Free energy equation?
Delta G = Delta H - T times Delta S
What does T mean in a Gibbs Free energy equation?
Temperature in Kelvin
What is a spontaneous reaction?
A reaction that occurs without continuous external energy input
Another way of saying a spontaneous reaction is?
Thermodynamically favorable
A thermodynamically favorable reaction can occur spontaneously without?
continuous external energy input, indicated by a negative Gibbs free energy change
What are the units for Entropy?
J/K or J x K^-1
What are the units for Enthalpy?
J
The relationship for Entropy change at equilibrium is?
enthalpy change/temperature
Delta H represents
Enthalpy change
Energy changes affects?
Energy dispersal and microstates
Entropy of the system is used by scientists to calculate what?
The entropy of the universe
When Delta H is less than 0 what does it represent?
Exothermic, releasing heat to its surroundings
When delta H is more than 0, what does it represent?
Endothermic, absorbing heat to it’s surroundings
Delta G degrees is less than 0 represents what?
A thermodynamically favorable and the entropy of the universe increase
What are the steps for dissolution?
!.) breaking down solute interactions then 2.) rearranging solvent molecules and then 3.) forming solute-solvent molecules
What is Thermodynamics based on?
Spontaneity
What is Delta G degrees a measure of?
the thermodynamic spontaneity of a chemical reaction under standard conditions
When the problem is kinetically favorable(faster rate), what does it mean about the activation energy?
It’s lower
When the problem is kinetically unfavorable(faster rate), what does it mean about the activation energy?
It’s higher
A given system at a fixed volume and energy reaches thermodynamic equilibrium when what happens?
It’s total entropy is maximized
Equilibrium and Free energy equation
Delta G degrees = -RT ln(K equillibrium)
The K rate constant equation?
e^(- Delta G degrees/RT)
If K is greater than 1, what does it mean for the forward reaction’s thermodynamically favorability and which is favored products or reactants?
It’s thermodynamically favorable and the products is favored
If K is less than 1, what does it mean for the forward reaction’s thermodynamically favorability and which is favored products or reactants?
It’s thermodynamically unfavorable and the reactant is favored
If Delta G is positive, than what does it mean for K
It’s small
If Delta G is negative, than what does it mean for K
Large
If Delta G is 0, than what does it mean for K equillibrium constant
It’ll be 1
When activation energy is higher what does it mean about the rate at which it reaches it's reaction?
The reaction rate will be slower
When activation energy is lower what does it mean about the rate at which it reaches it's reaction
The reaction rate will be faster
At high temperatures, will it be thermodynamically favorable and why
It’ll be thermodynamically favorable because a high temperature in the Gibbs free energy equation will Delta G negative
What is a Coupled Reaction?
the pairing of a thermodynamically favorable (exergonic, Delta G < 0) reaction with a thermodynamically unfavorable (endergonic, Delta G > 0) one
In a Coupled Reaction, which reaction drives the other?
The favorable reaction drives the non-favorable reaction
When the favorable reaction drives the unfavorable one, what does it then combine to?
a combined spontaneous reaction and has a negative delta G
In a Coupled Reaction, how does a thermodynamically favored reaction drive the unfavorable thermodynamic reaction?
By production so much of one of the unfavored reaction reagents that it shifts the unfavored reactions equilibrium, therefore driving it toward it’s products
Changing the reaction conditions changes what?
the free energy’s relationship to equilibrium
What does the Le Chartelie’s principle state?
if a dynamic equilibrium is disturbed by changing conditions (concentration, pressure, or temperature), the system shifts its equilibrium position to counteract that change and restore balance
The equation for total Delta G?
Delta G favored - Delta G un-favored
When Delta G is negative, what is the signs for Delta S and Delta H
Delta S is positive while Delta H is negative
To find Delta S of a specific molecule, what do you do?
Sum of Delta S products - the sum of Delta S reactants
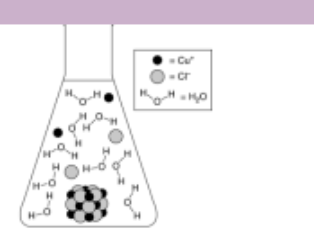
When you see a graph in a problem that has a percipatate with many molecules, what does it mean about the enthalpy
It’s high