IB biology : Topic A.1.1 - water
1/21
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
22 Terms
bond inside a water molecule
polar covalent bond
breaking polar bond
mechanical force is needed
weak, intermolecular force
how to represent a covalent polar bond between water molecules
( add a little delta )
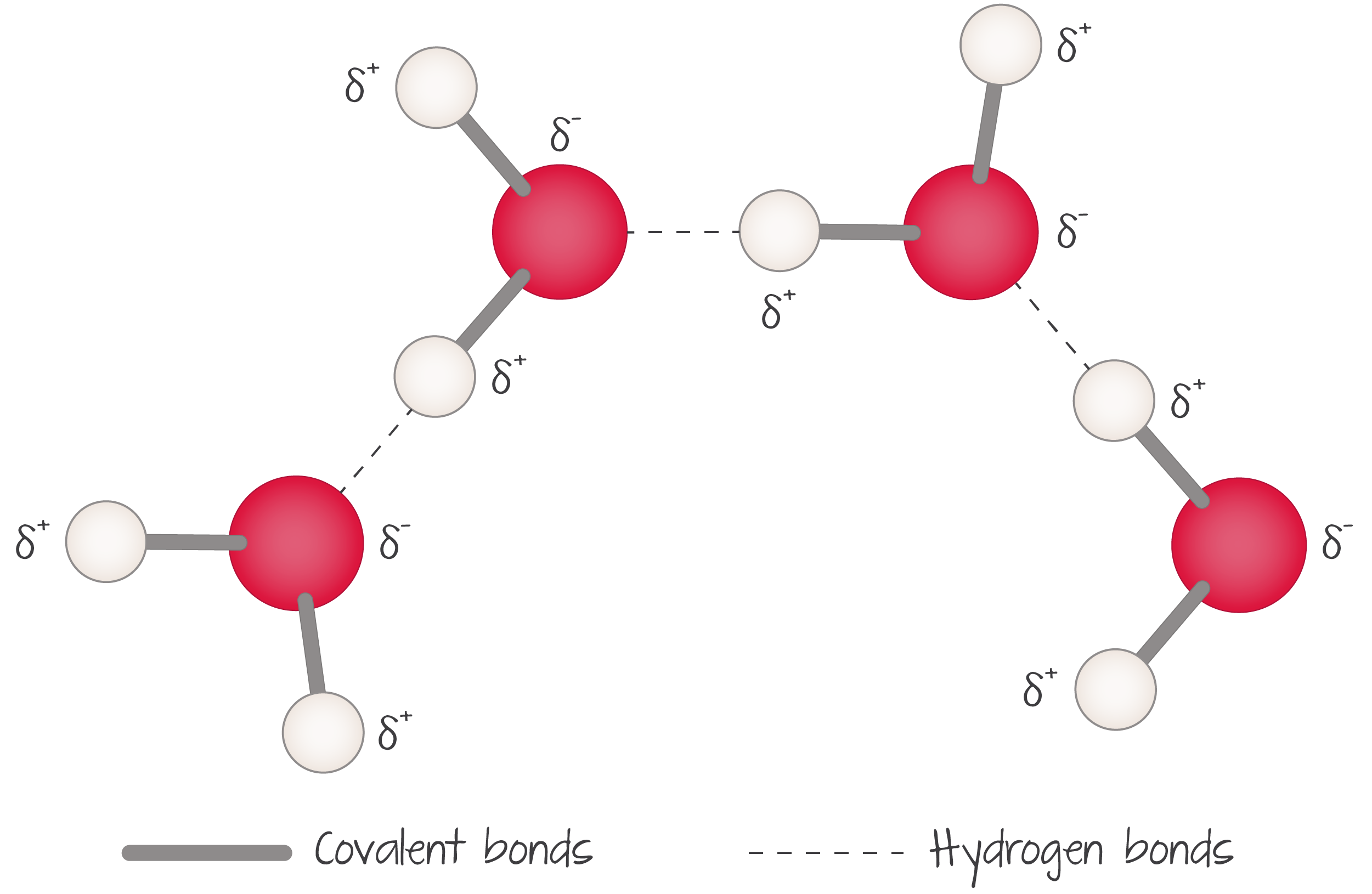
cohesion
process in which “like” polar molecules are attracted to each other’s positive and negative parts.
cohesion consequences
resists increases of surface area; by gravity or foreign objects
water uses in life
transport
Medium in which chemical reactions of the metabolism take place
solvent
adhesion
water attraction to polar/ charged materials
capillary action
The ability of a liquid to move through narrow tubes that results from cohesion, adhesion and surface tension.
Caused by adhesion of water molecules to surrounding materials.
happens when adhesion is greater than cohesion
is how water moves in the xylem
hydrophilic
molecules that are attracted to water, usually polar molecules
hydrophobic molecules
molecules why are not attracted chemically to water. Usually non polar molecules are like that
Aquaporins
Transmembrane proteins which are anchored in place on the plasma membrane by they hydrophobic exterior and hydrophilic interior. It allows for efficient transport of water in and out of the cell
Rules for dissolving
Like dissolves like; polar molecules dissolve polar molecules, and non polar molecules dissolve non polar molecules
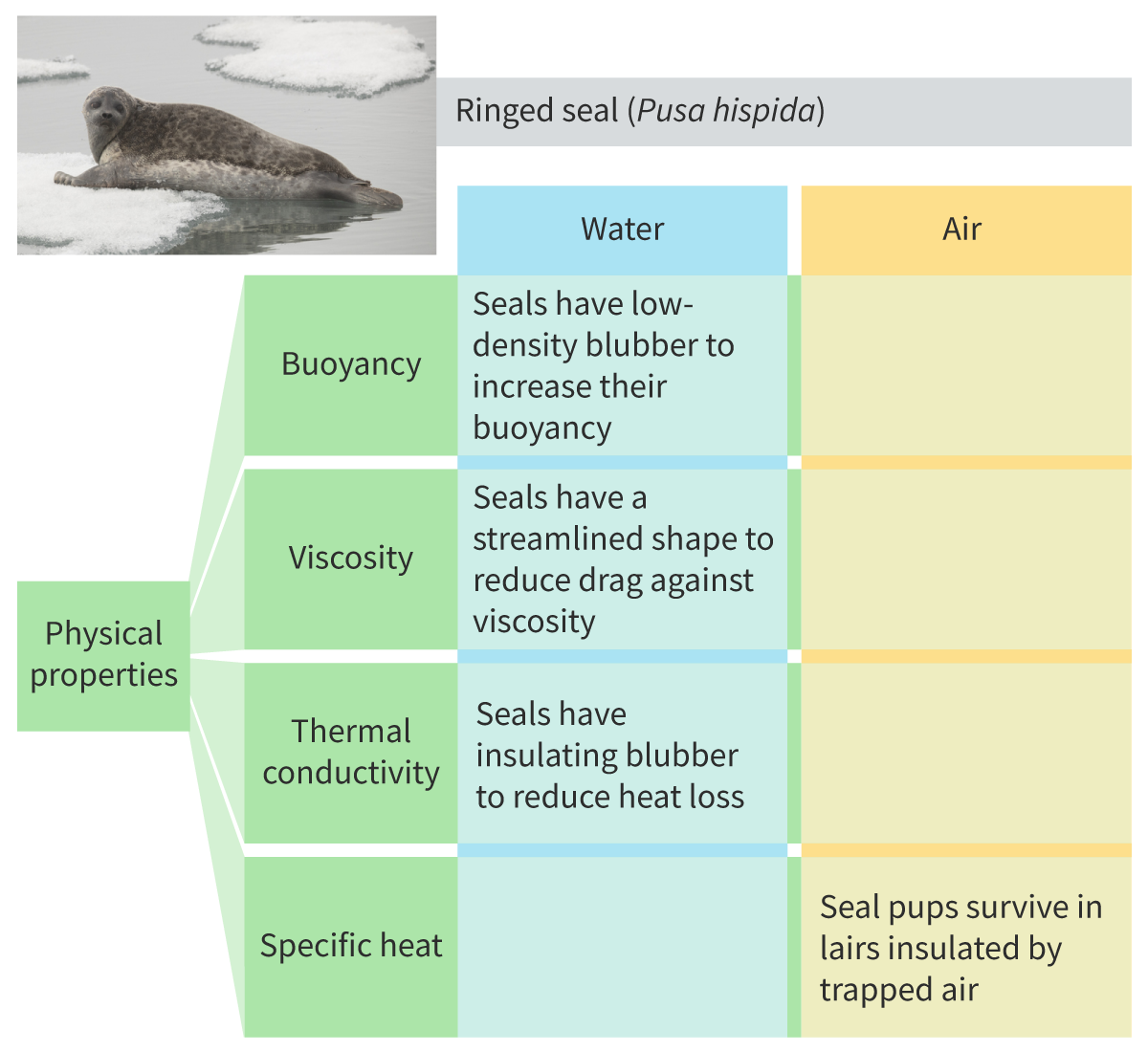
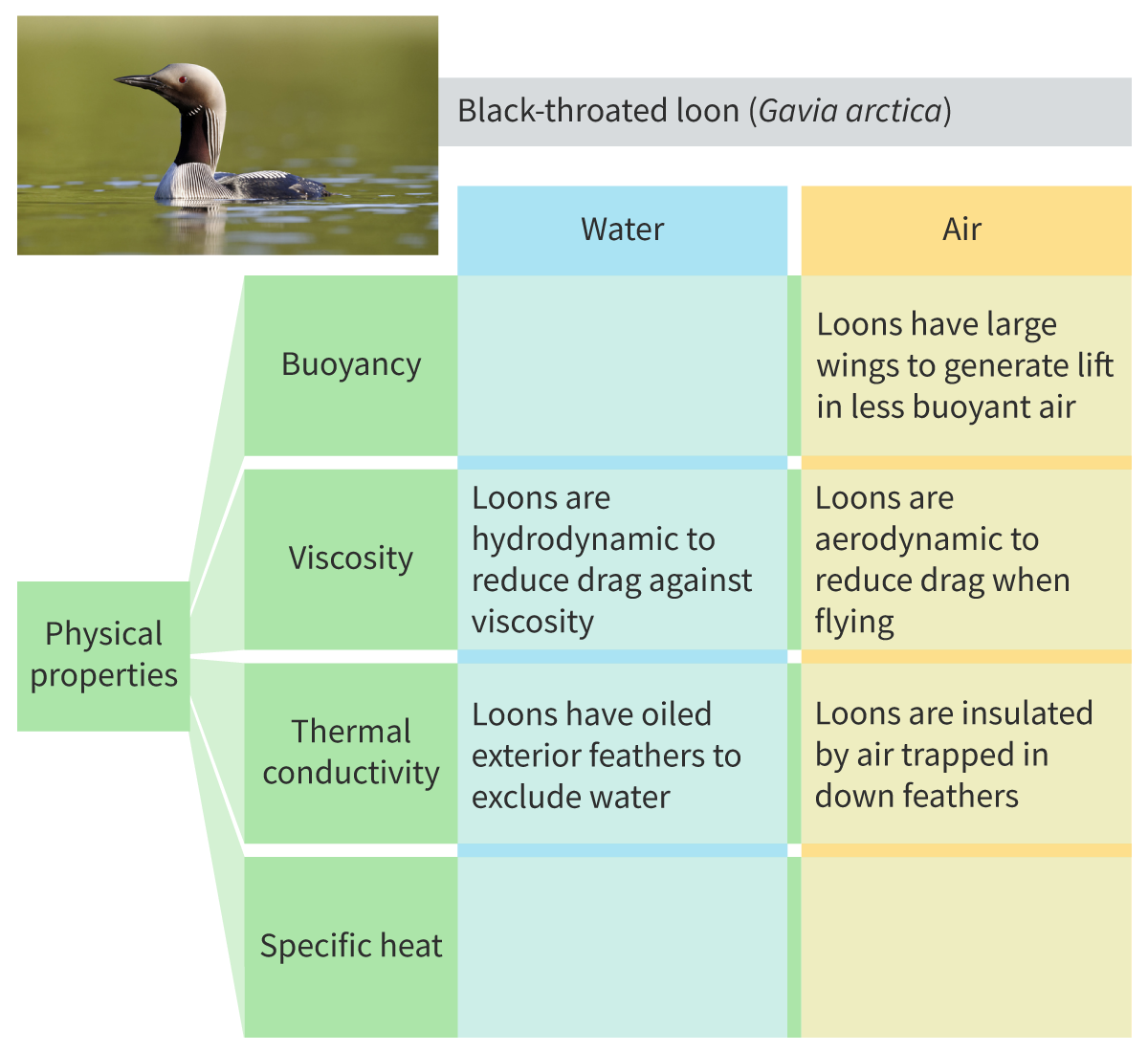
Physical properties of water
High buoyancy
Low Viscosity
High Thermal conductivity
High Specific heat capacity
Buoyancy property
Counteracts gravity because of high density. Colder water is denser, so it sinks. Ice is less dense than water
Viscosity
How much energy is needed to change the shape of the liquid. Resistance to flow
Thermal conductivity
Ability of a substance to transfer heat when there is a temp difference. High thermal conductivity compared to other liquids ( which aren’t liquid metals ) : air has a much lower thermal conductivity than water
Specific heat capacity
How much energy is needed to raise the temperature of a substance. Water has a high one, higher than hydrogen, so a lot of energy is required to increase it’s temperature
Case study of seals
case study of black-throated loon ( bird)
goldilock zone
area around a sun which allows for water to be liquid, thus for life to form
it is also called the “habitable” zone
other planets in such a zone might have the conditions for extraterrestrial life to emerge
origin of water on earth
collisions with water rich asteroids/ comets 4-5 billion years ago brought water on earth
why is water on earth
temperate temperature: water doesnt evaporate nor it freezes, allowing it to be the ideal medium of life
gravitational forces are strong enough to keep the water on earth’s surface