CHEM 1.5 - KINETICS
1/11
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
12 Terms
activation energy Ea
activation energy is the minimum amount of energy that particles must have in order to react
collision theory
for a reaction to occur, particles must collide with enough energy (at least the activation energy) and in the correct orientation
why do most collisions not lead to a reaction?
because they do not have enough energy or do not collide in the correct orientation
rate of reaction
rate of reaction is the change in concentration of a reactant or product per unit time

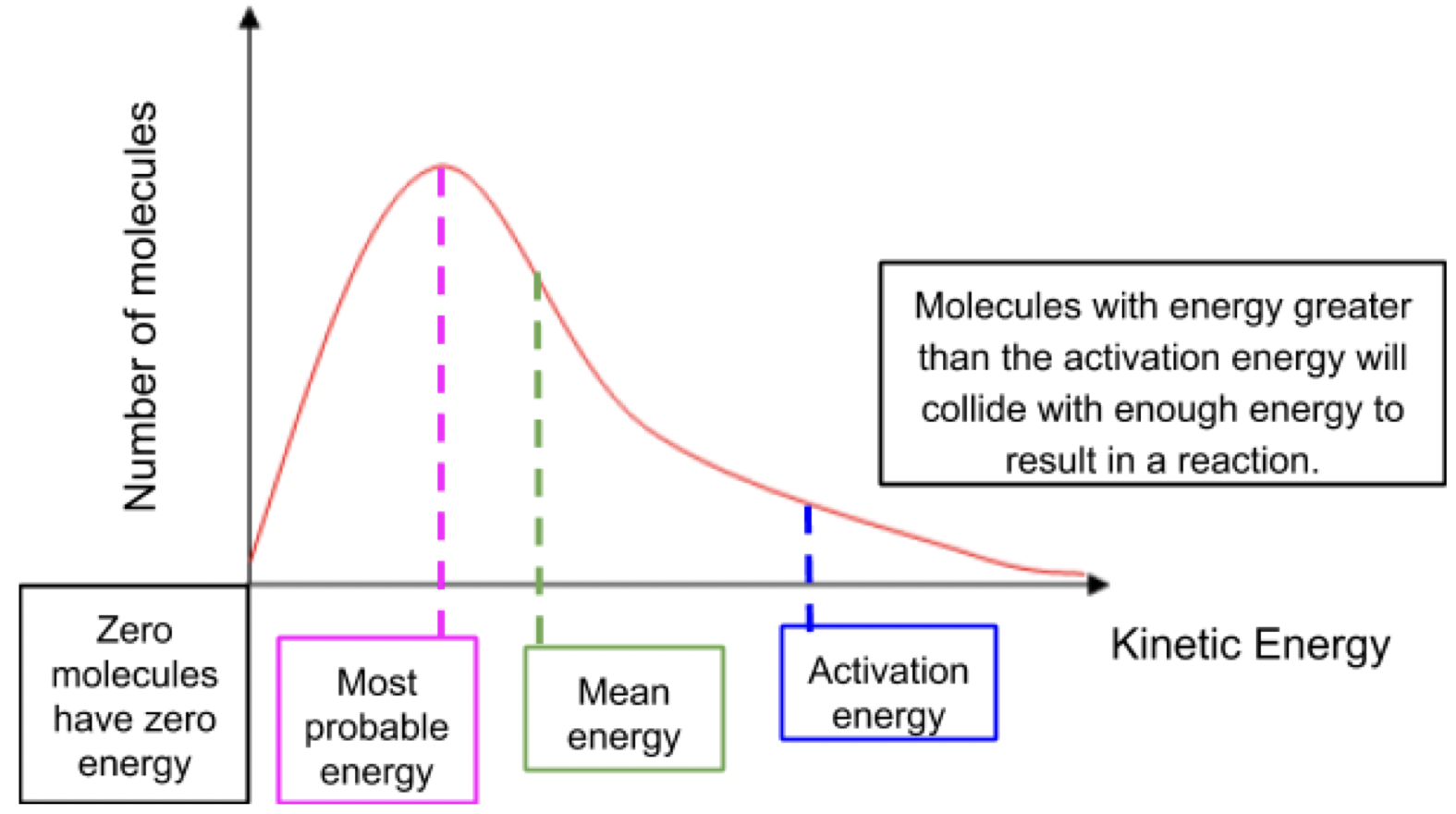
maxwell boltzmann graph
the most probable energy is the energy that the largest number of particles in the sample have
area underneath the curve is the no of particles
maxwell boltzmann graph - higher/lower temp
the new curve should only overlap the old one once
higher temp= curve moves to the right and peak becomes shorter. this is because more particles should have higher energy than the Ea whilst the area underneath curve remains the same
lower temp= curve moves to the left and peak becomes longer. this is because less particles should have more energy than the Ea whilst the area underneath curve remains the same
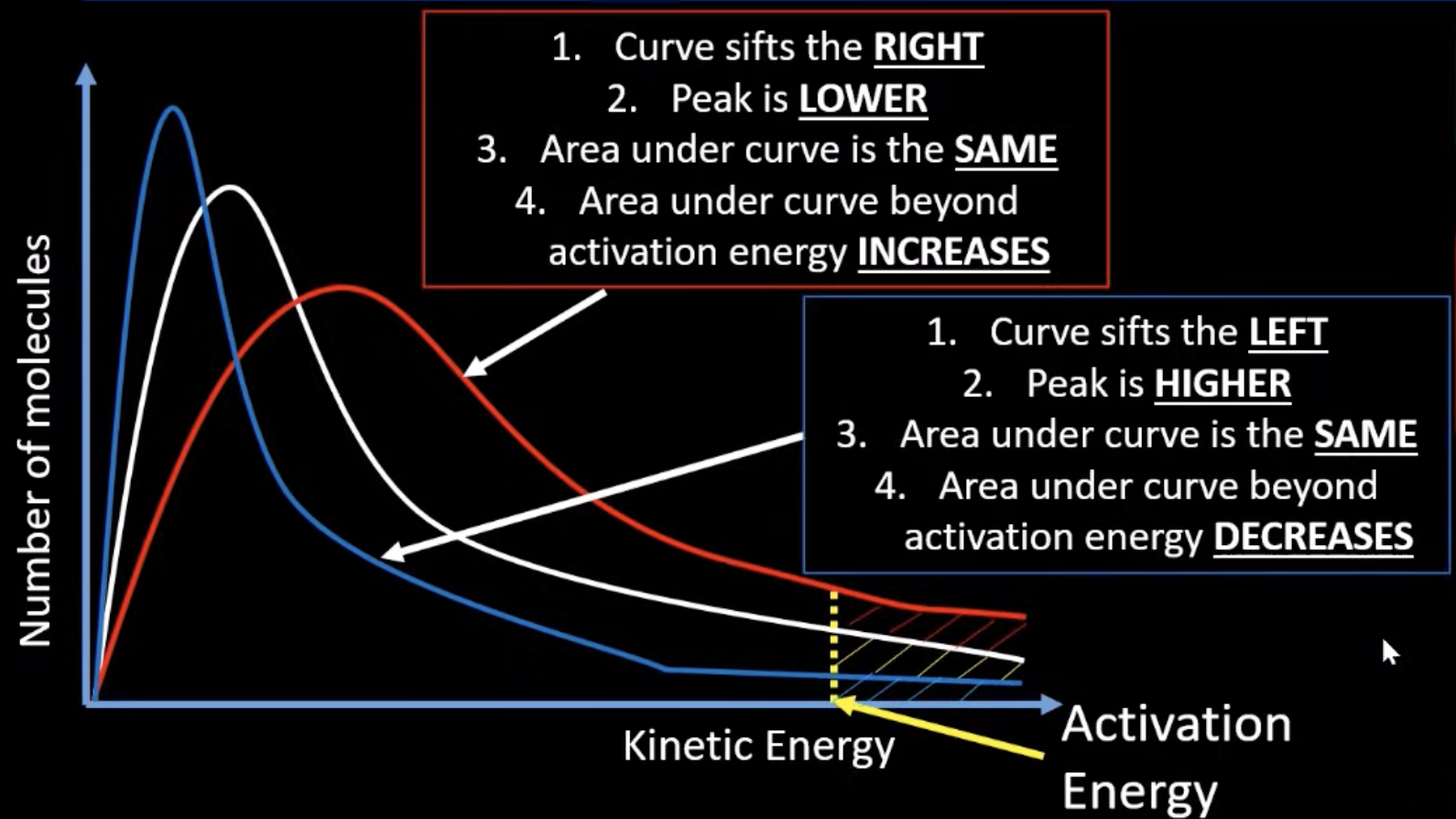
why a small increase in temp can lead to a large increase in rate
because the curve shifts to the right meaning overall a larger amount of molecules have energy greater than or equal to the Ea
effect of concentration on rate of reaction
increased conc = increased rate
particles are closer together and collide more often. more frequent collisions and higher chance of reaction
effect of pressure on rate of reaction
increased pressure = increased rate
particles are closer together and collide more often. more frequent collisions and higher chance of reaction
effect of catalyst on rate of reaction
activation energy moves to the left on the Maxwell–Boltzmann graph, so more particles have energy equal to or greater than the activation energy
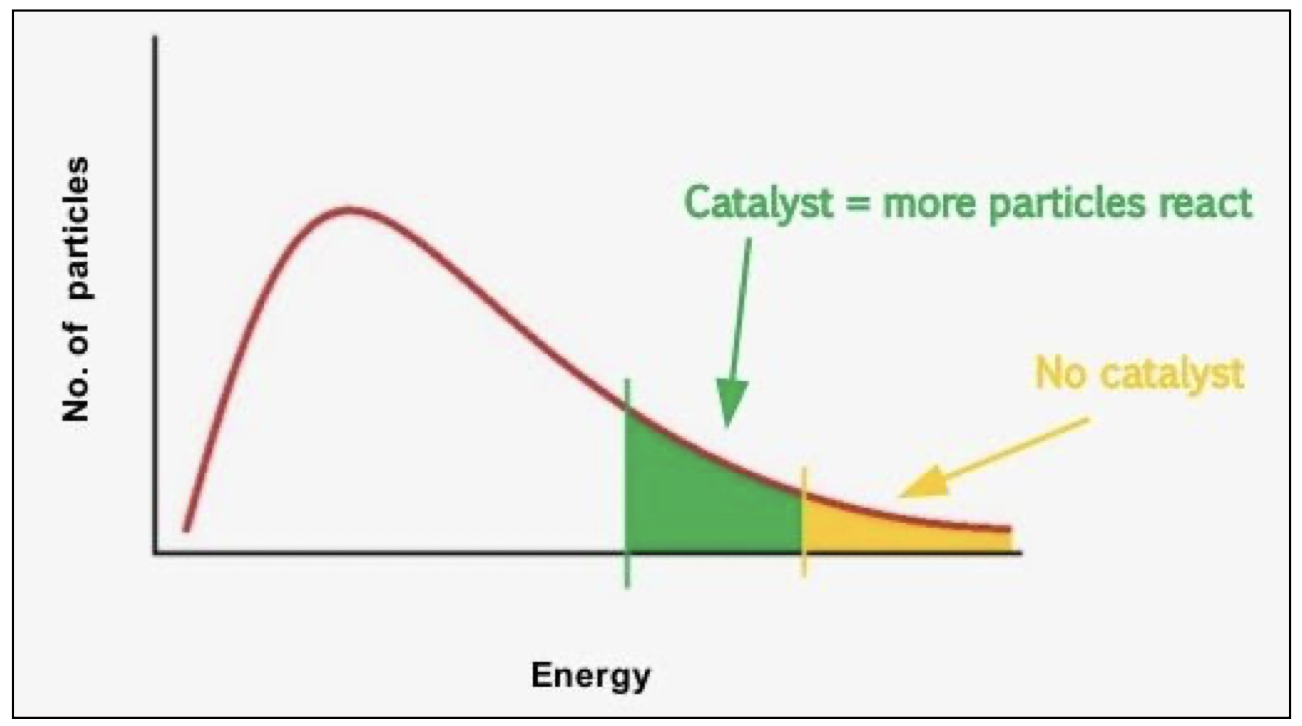
catalyst
a catalyst increases the rate of a reaction by providing an alternative pathway with a lower activation energy. it is chemically unchanged at the end of the reaction
what effects Maxwell Boltzmann distribution graphs
ONLY TEMPERATURE !
pressure, concentration etc do not change curve of the graph
they only affect rate of reaction but energy of molecules stays the same