Radioactivity
1/17
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
18 Terms
What is the function of the control rods in a nuclear reactor
They absorb excess neutrons
What is the function of the moderator in a nuclear reactor
It slows down neutrons
What is ionising radiation
Removing electrons from an atom
What is the random process
Not knowing when nuclei will decay
What is released by nuclear fission
Two daughter nuclei, neutrons and kinetic energy
What is the role of the shielding in the nuclear reactor
To absorb the ionising radiation to prevent it escaping as it is dangerous
What is nuclear fission
The splitting of the nucleus of an atom which releases energy
What is nuclear fusion
Fusing together a nucleus
What is a chain reaction
If neutrons produced by one fission strike other U-235 nuclei that leads to further reactions
Why is high pressure and temperature needed for fusion
It needs around 10,000,000 degrees Celsius as it doesn’t work for lower temperatures and pressure due to electrostatic repulsion of protons
What are the two ways to detect radioactivity
Geiger- Muller tube and photographic film
What is a half-life
Is the time taken for half of the radioisotopes in a sample to decay
What is background radiation
It is radiation that is all around us
What are sources of background radiation
Food
Radon gas
Cosmic rays
Gamma rays
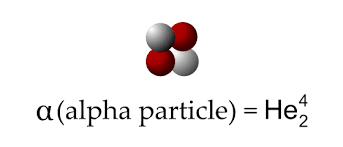
Alpha particle (2 protons, 2 neutrons)
What is it range, what it is blocked by, what is it ionising ability, what it is charge and mass
Range in air- 5cm
Blocked by a sheet of paper
High ionising ability
Charge +2
Mass 4

Beta particle (electron)
What is it range, what it is blocked by, what is it ionising ability, what it is charge and mass
Range in air- 1m
Blocked by 2-3mm of aluminium
Medium ionising ability
Charge -1
Mass 0.0005
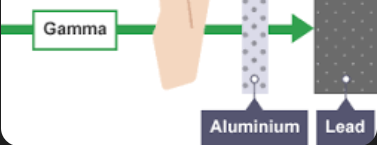
Gamma (EM wave)
What is it range, what it is blocked by, what is it ionising ability, what it is charge and mass
Range in air- Long
Blocked by 2-3cm of lead
Low ionising ability
Charge and mass- 0
What is an isotope
Isotope is an atom with different number of neutrons and same number of protons