NS 3310 Lecture 11
1/92
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
93 Terms
Functional Features of Proteins: Proteins mediate almost every process that takes place in a cell, exhibiting an almost endless diversity of functions. What are 7 examples?
Enzymes
Hormones
Structural proteins
Immunoproteins
Transport proteins
Receptor proteins
Motor proteins
Our cells produce proteins with strikingly different ___and _____ by combining a common set of 20 amino acids.
properties ; functions
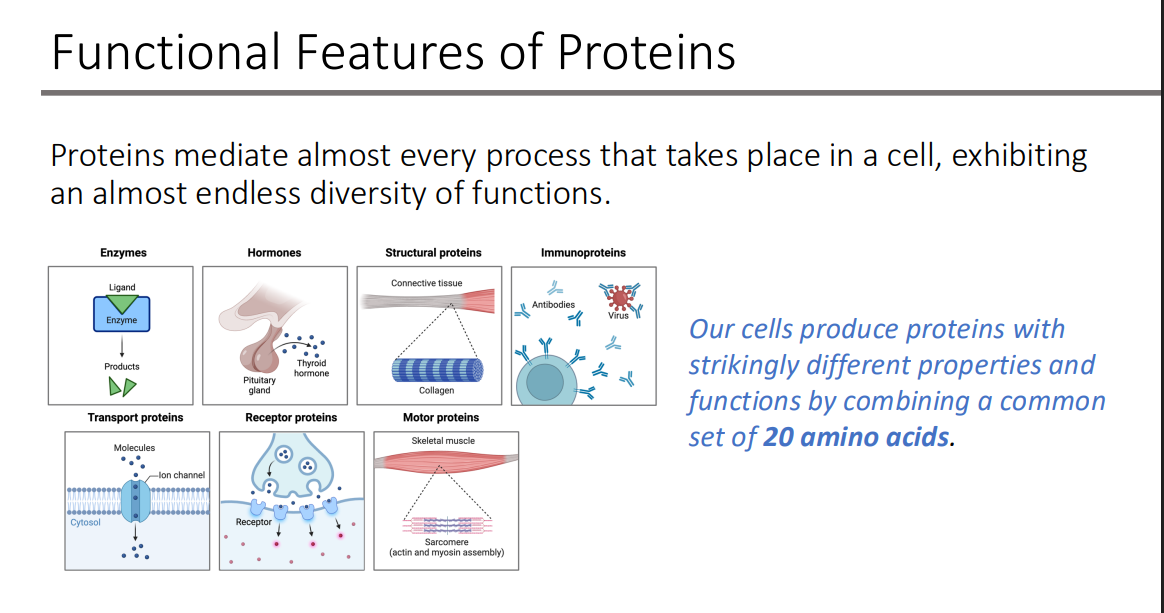
Amino Acids are the building blocks of proteins: Amino acids (AA) contain what three things that are all attached to the α-carbon?
Carboxyl group
Amino (or imino)
Side chain (R group)
Only which -isomers can be used to build up proteins
L-isomers
What determines if it is an amino or imino?
location of the double bond
Except for __, all proteogenic AA have a chiral center.
Gly
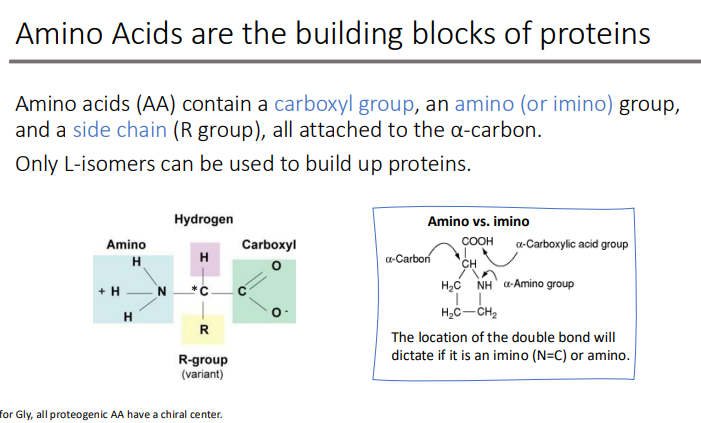
The _____ of an amino acid determines its unique chemical properties, such as what three things? which will influence the aa role in protein structure and function
side chain
Polarity
Charge
Size
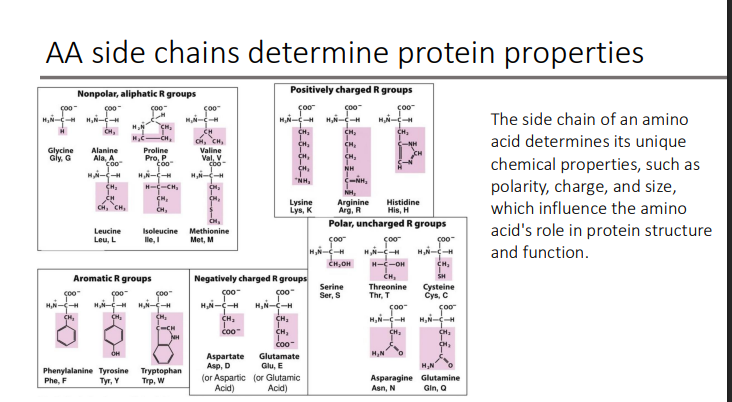
Proteins form a structure in a hierarchical manner from so-called primary, secondary, tertiary, and quaternary structures. What is the primary structure? What does it direct?
polypeptide sequence
which directs the folding of the protein into its three-dimensional structure.
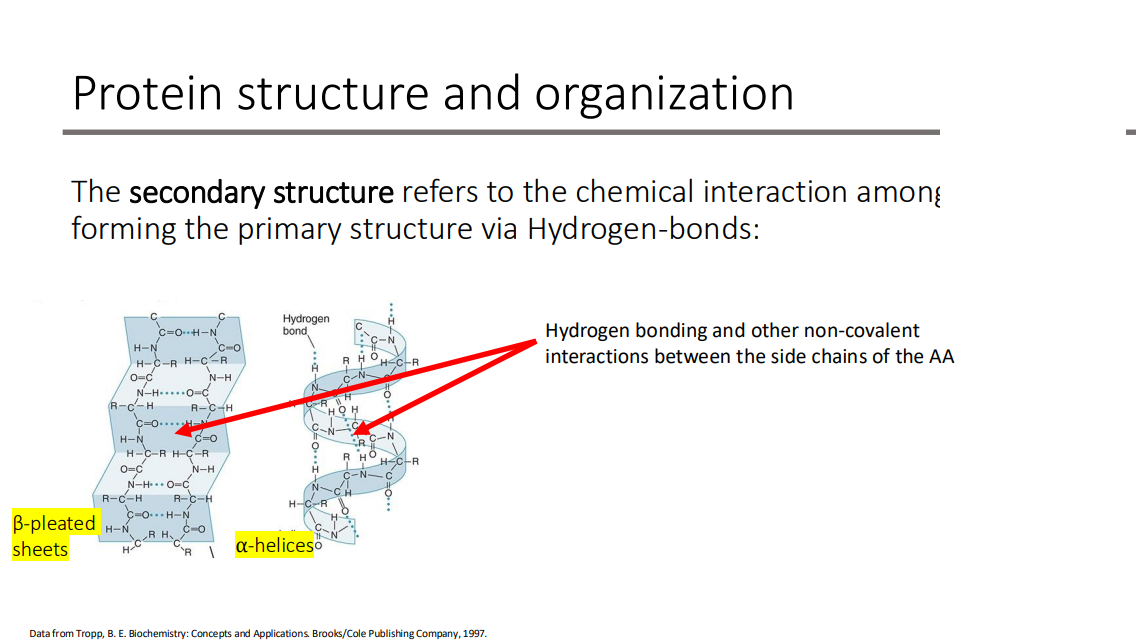
The secondary structure refers to the chemical interaction among the AA forming?
the primary structure via Hydrogen-bonds
Hydrogen bonding and other non-covalent interactions between the side chains of the AA
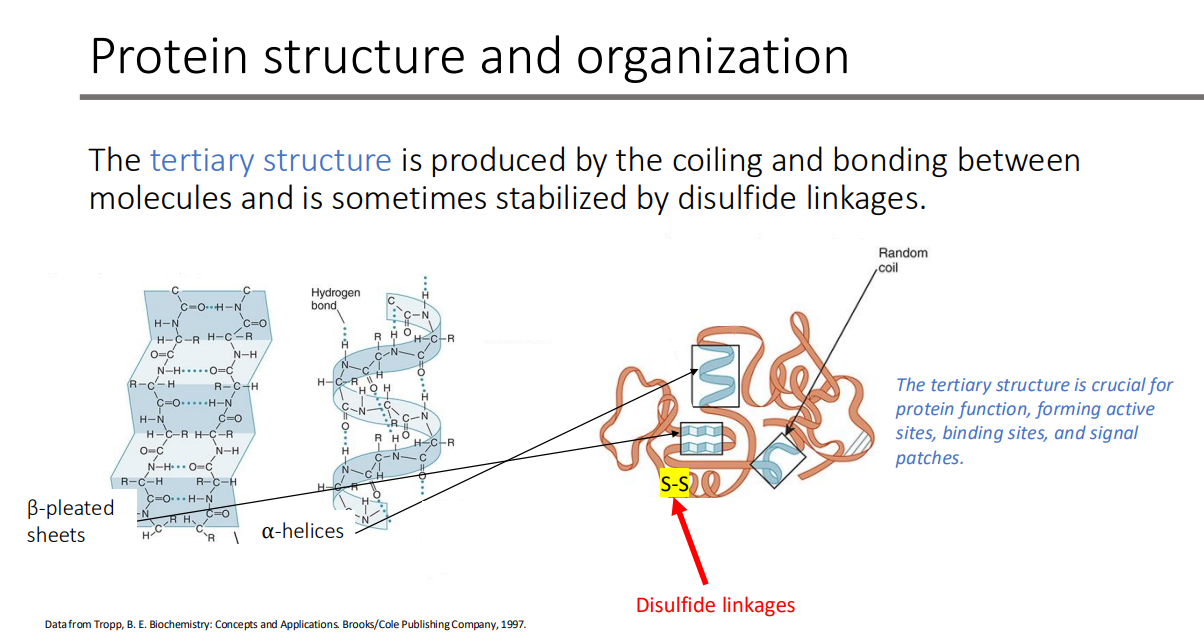
The tertiary structure is produced by the ___and ____ between molecules and is sometimes stabilized by ____ linkages.
coiling; bonding; disulfide
The tertiary structure is crucial for what 4 things?
Protein function
Forming active sites
Binding sites
Signal patches
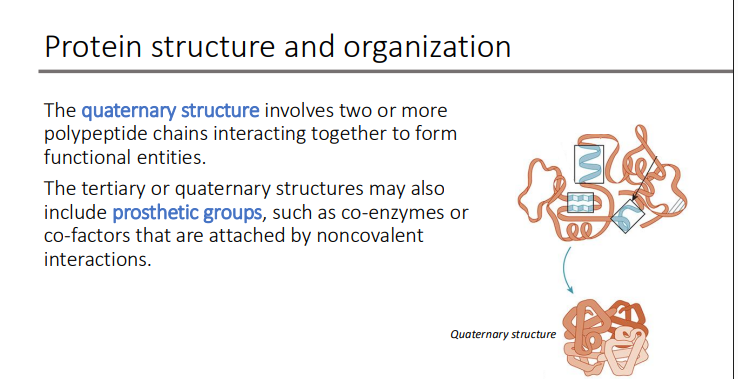
The quaternary structure involves two or more?
polypeptide chains interacting together to form functional entities
The tertiary or quaternary structures may also include?
prosthetic groups
such as co-enzymes or co-factors that are attached by noncovalent interaction
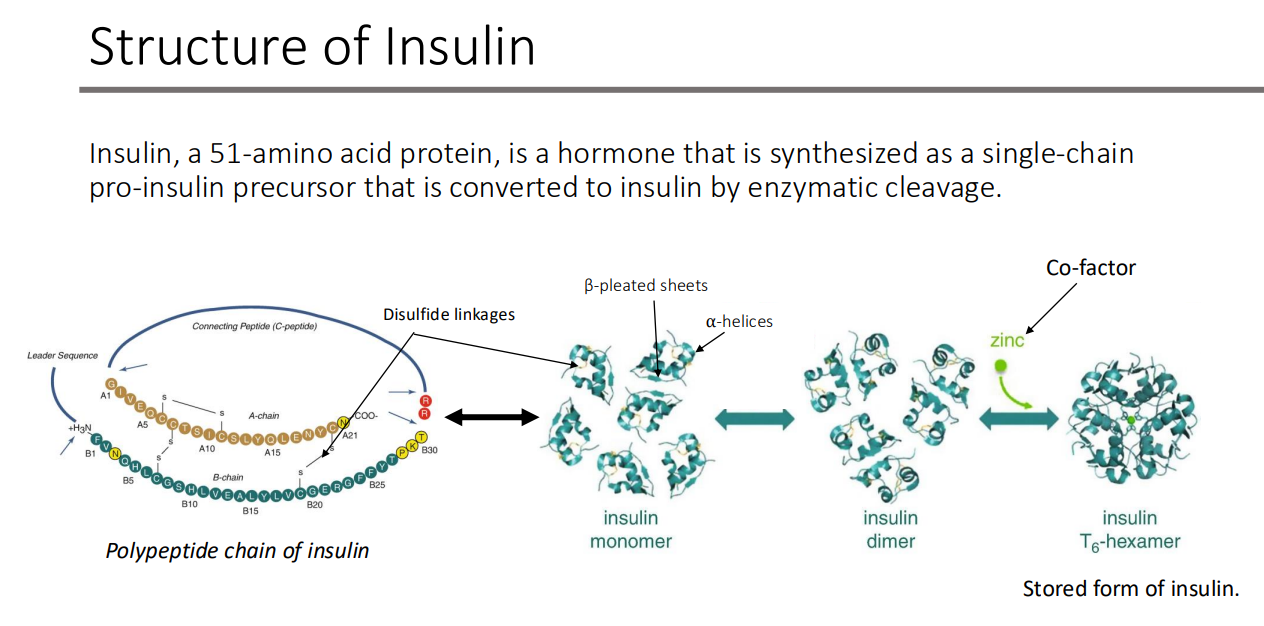
Structure of Insulin: Insulin, a 51-amino acid protein, is a hormone that is synthesized as a single-chain?
pro-insulin precursor that is converted to insulin by enzymatic cleavage
zinc is a co-factor!
Humans need an adequate supply of dietary proteins to obtain what two things?
amino nitrogen and essential amino acids
Humans can only produce 11 out of the 20 proteinogenic AA, making dietary protein essential for cellular function and health.
Unlike CHO and lipids, the body doesn't store protein. Where does excess AA go?
they are catabolized, and nitrogen is excreted as urea.

What are the 11 essential amino acids?
PVT TIM HALL
Phenylalanine
Valine
Threonine
Tryptophan
Isoleucine
Methionine
Histidine
Arginine*
Leucine
Lysine
*Arginine is non-essential for adults but essential for growth in children and during certain pathological or physiological conditions

The RDA for protein is based on analyses of available nitrogen balance studies: In studies with healthy adults with presumably good nutritional status, it is generally assumed that the protein requirement is achieved when an individual is in?
zero nitrogen balance.
Nitrogen balance is the difference between
nitrogen intake and the amount excreted in urine, feces, skin and miscellaneous losses
What is a positive nitrogen balance?
(Nin > Nout) occurs when the body retains nitrogen to build up tissues
What is negative nitrogen balance?
(Nin < Nout) occurs when the amount of nitrogen loss is greater than the amount retained, which is an indicator of protein catabolism

A Protein RDA for Adults is Set at?
0.8g/kg/d
RDAs for protein are given on a per weight basis, and on a per day basis
RDAs are based on the body’s needs for? plus the need for?
maintenance plus the need for net protein synthesis during growth, pregnancy, and lactation.

What is the AMDR of protein?
10-35%
The lower ends of these ranges were set to ensure adequate protein intake, whereas the upper ends were set to balance the AMDRs for CHO and lipids and are well above typical intakes of protein

Sources of Dietary Protein Include Both Animal and Plant Foods
Fats/Sweets → ~0
Milk + Milk products → ~8 g/serving
Vegetables → ~2g/serving
Meat/Fish/Poultry → ~7 g/ounce
Fruits → 0
Starches→ ~3 g/serving; 7 g/serving of beans (1/2 cup)

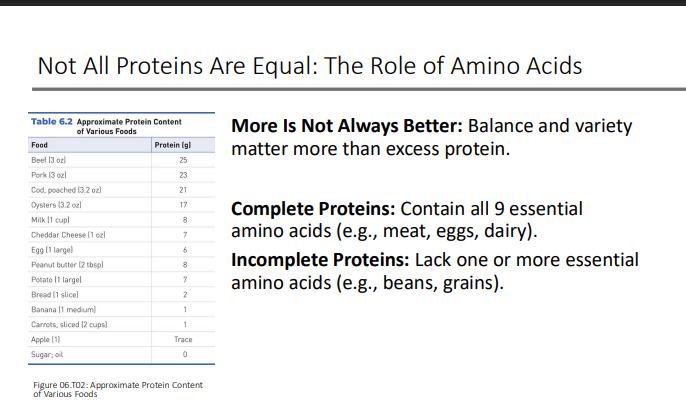
Not All Proteins Are Equal: The Role of Amino Acids: More Is Not Always Better: what matters more than excess protein?
balance and variety
What are complete and incomplete proteins?
Complete Proteins: Contain all 9 essential amino acids (e.g., meat, eggs, dairy).
Incomplete Proteins: Lack one or more essential amino acids (e.g., beans, grains).
Which level of protein structure involves the interaction of two or more polypeptide chains to form a functional protein complex?
Quaternary structure

Select TRUE statements about protein food sources and requirements.
C. ii, iii, vi
What are the 2 semi-essential Proteinogenic Amino Acids (which are derived from essential AA)?
Tyrosine → from phenylalanine
Cysteine → from methionine
What are the 9 nonessential AA?
Gly, Ser, Glu (glutamic acid), Gln, Ala, Asn, Asp (aspartic acid), Arg, Pro.
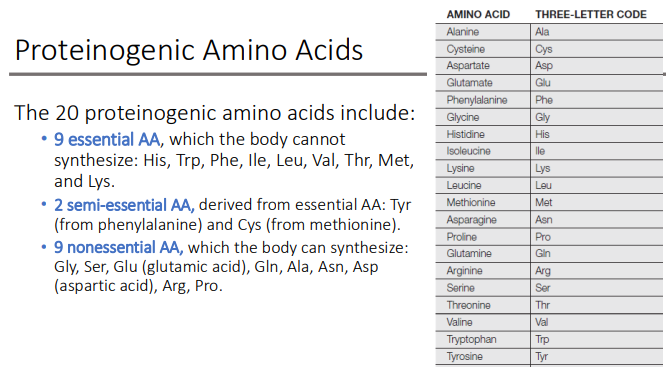
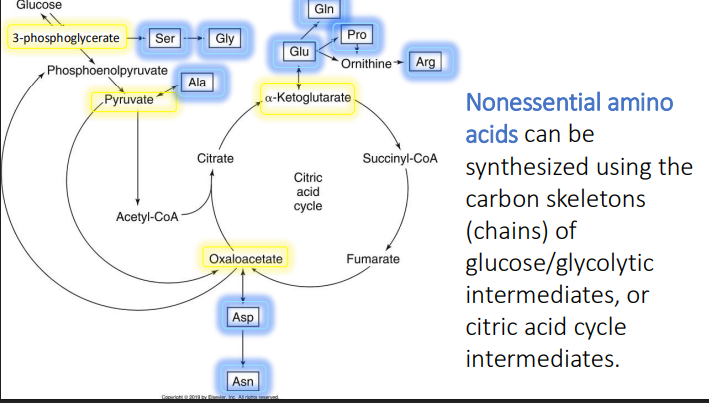
Nonessential amino acids can be synthesized using the?
carbon skeletons (chains) of glucose/glycolytic intermediates, or citric acid cycle intermediates.
What two AA can 3-phosphoglycerate make?
Ser
Gly
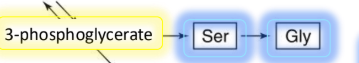
What AA can pyruvate make?
Ala

What 4 AA can a-ketoglutarate make?
Glu (directly)
Gln
Pro
Arg
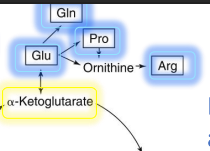
What 2 AA can oxaloacetate make?
Asp
Asn
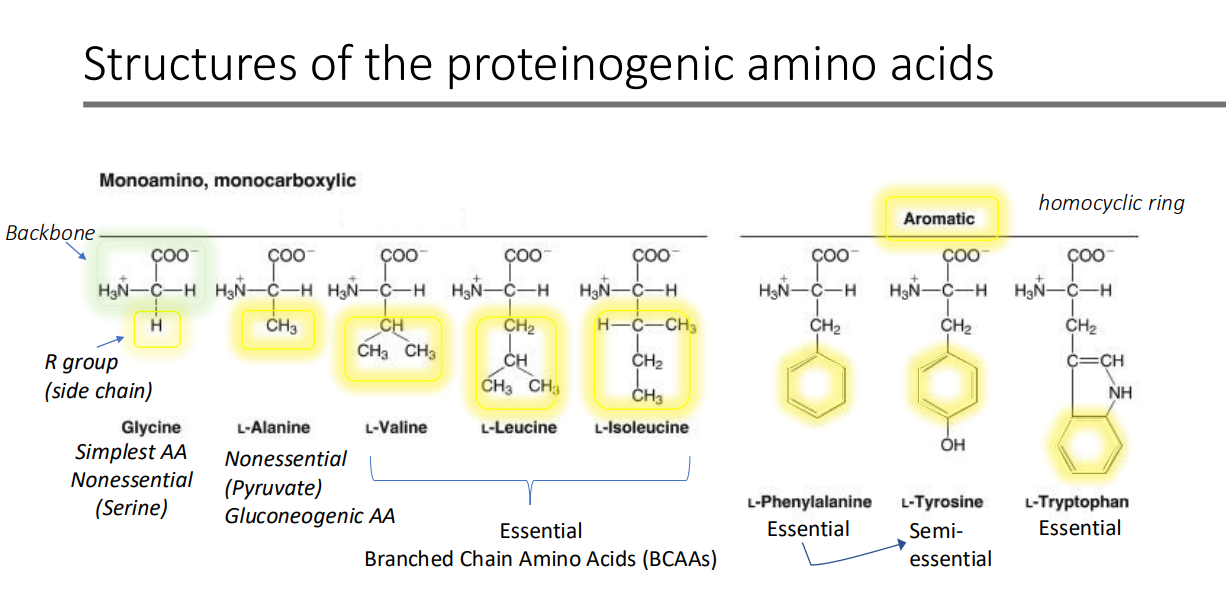
Structures of the proteinogenic amino acids
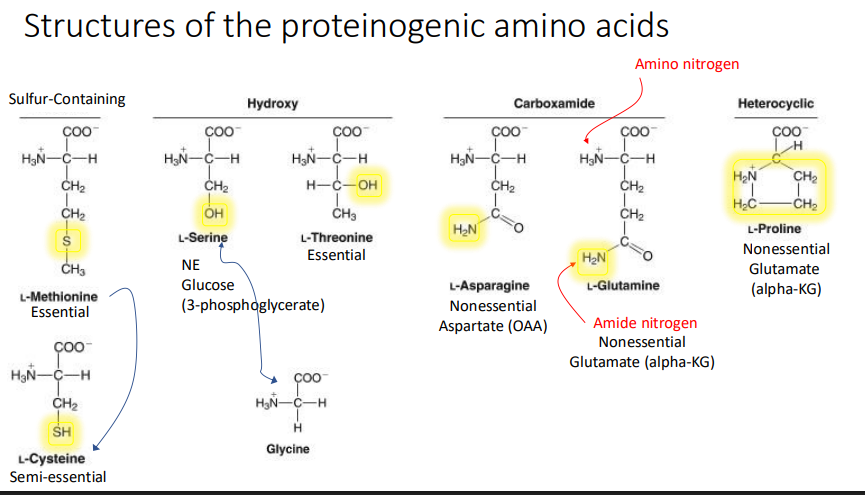
Structures of the proteinogenic amino acids
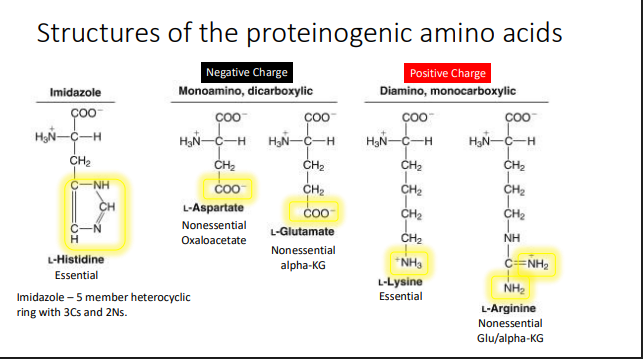
Structures of the proteinogenic amino acids
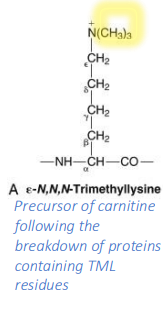
Some AA residues in proteins undergo modification after they have been incorporated into the protein (post-translational modification). Describe E-N,N, N Trimethyllysine, a post-trans mod of lysine
precursor of carnitine following the breakdown of proteins containing TML residues
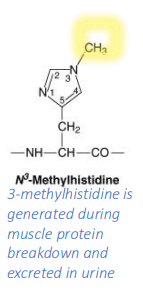
Describe 3-methylhistidine, a post-trans mod of histidine
generated during muscle protein breakdown and excreted in urine
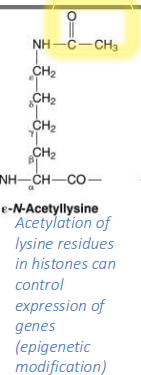
Describe E-N-Acetyllysine, a post-trans mod of lysine. Acetylation of lysine residues in histones can control?
expression of genes (epigenetic modification)
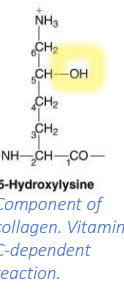
Describe 5-hydroxylysine, a post-trans mod of lysine.
Component of collagen. Vitamin C-dependent reaction.
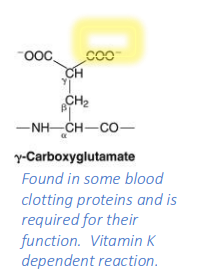
Describe y-carboxyglutamate, a post-trans mod of glutamate.
Found in some blood clotting proteins and is required for their function
Vitamin K dependent reaction.
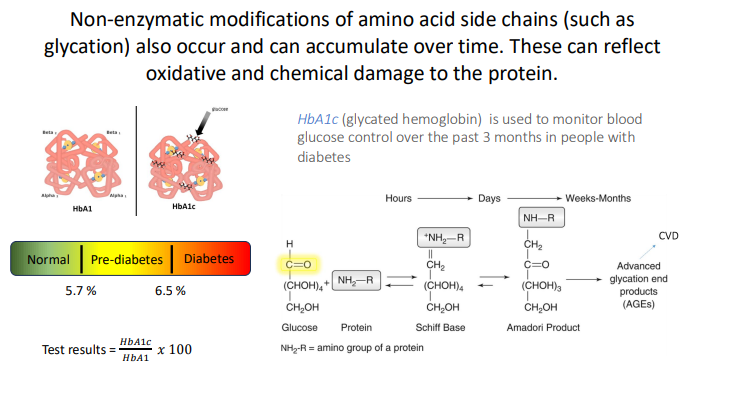
Non-enzymatic modifications of amino acid side chains (such as glycation) also occur and can accumulate over time. These can reflect what type of damage to the protein?
oxidative and chemical
HbA1c (glycated hemoglobin) is used to monitor?
blood glucose control over the past 3 months in people with diabetes
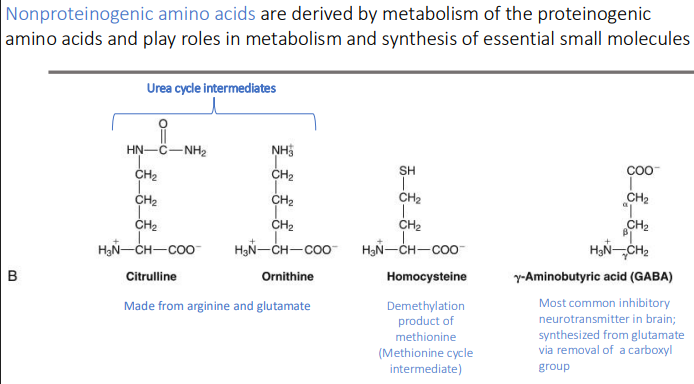
Nonproteinogenic amino acids are derived by? They play roles in?
metabolism of the proteinogenic amino acids
play roles in metabolism and synthesis of essential small molecules
What are the two urea cycle intermediates? What 2 AA are they made from?
Citrulline
Ornithine
Made from arginine & glutamate
Homocysteine is a nonproteinogenic amino acids. It is a demethylation product of?
methionine (Methionine cycle intermediate)
y-Aminobutyric acid (GABA) is most common? How is it synthesized?
Most common inhibitory neurotransmitter in brain
glutamate via removal of a carboxyl group
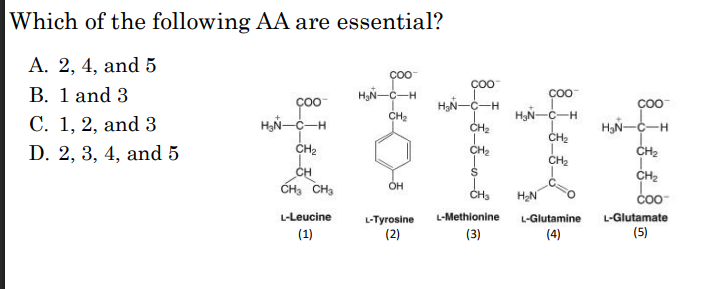
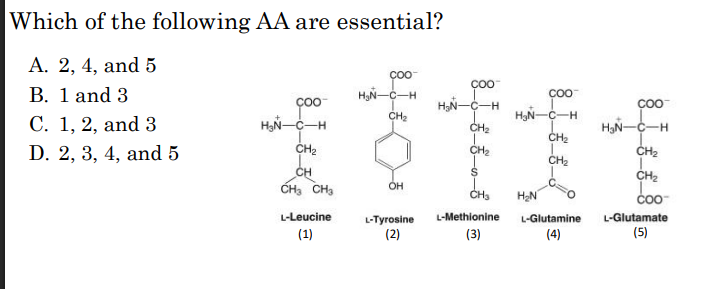
Which of the following AA are essential?
1 and 3
Which of the following AA can be synthesized from OAA?
A. Ala
B. Arg
C. Pro
D. Asp
D. Asp
Overview of protein digestion and absorption
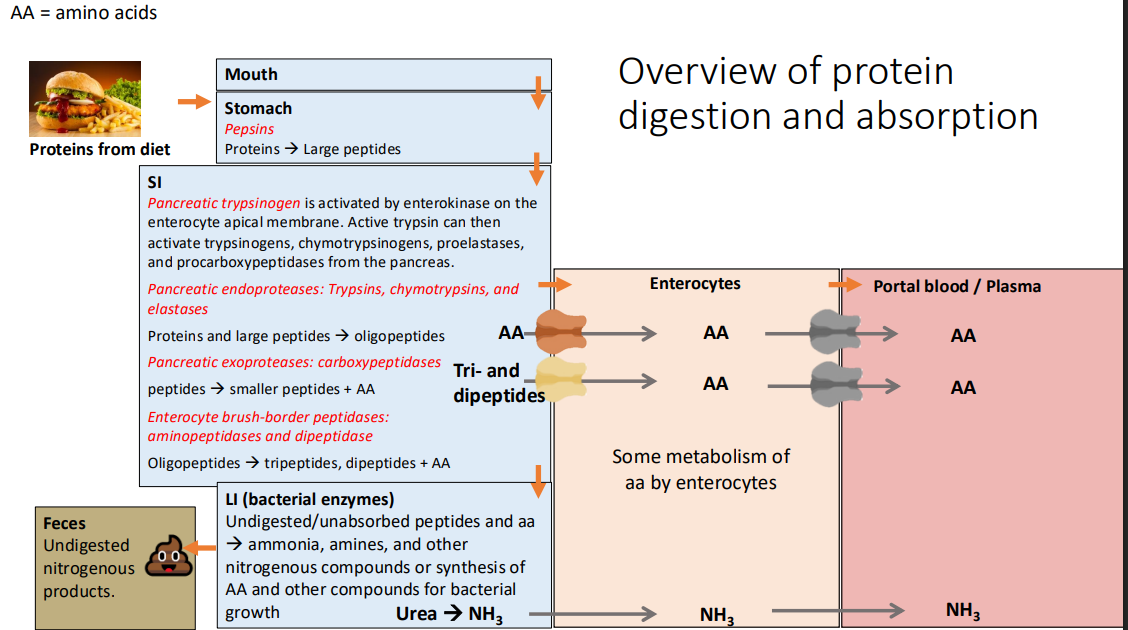
From blood capillaries into circulation: The digestion and absorption process ultimately supply?
portal blood with free amino acids
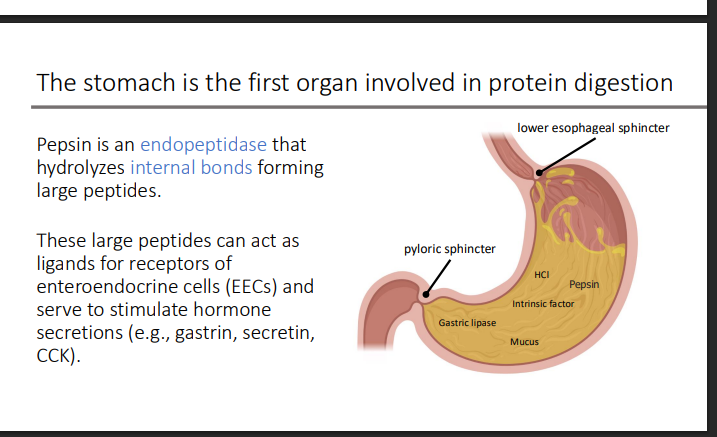
What organ is first involved in protein digestion?
the stomach
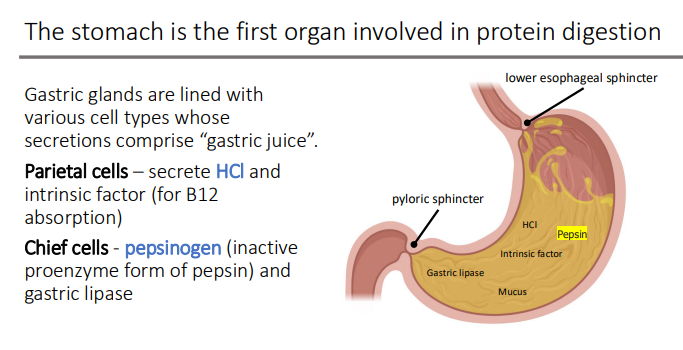
Protein Digestion Begins in the Stomach: Proteins are denatured by gastric acid (HCl), unwinding the protein to expose its?
peptide bonds, providing a greater surface area for pepsin
HCl also stimulates the conversion of ____ to ____
pepsinogen; pepsin (optimal pH <3.5)
Pepsin catalyzes the hydrolysis of?
peptide bonds
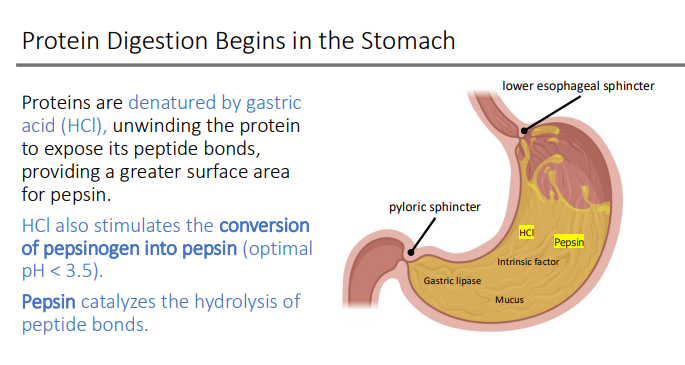
Pepsin is an ____ that hydrolyzes internal bonds forming what types of peptides?
endopeptidase; large peptides
These large peptides can act as ____ for receptors of enteroendocrine cells (EECs) and serve to stimulate?
ligands
hormone secretions (e.g., gastrin, secretin, CCK).
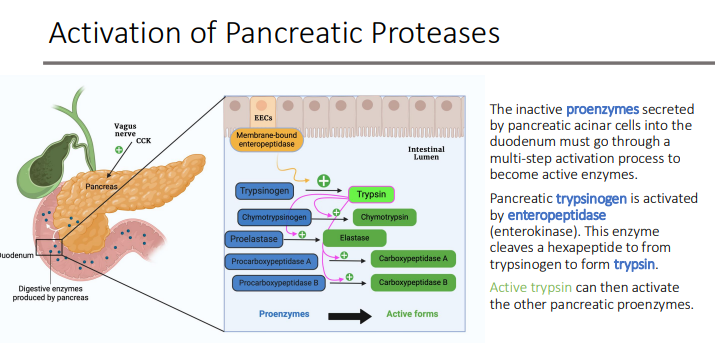
Activation of Pancreatic Protease: The inactive proenzymes secreted by pancreatic acinar cells into the duodenum must go through a multi-step activation process to become active enzymes. Pancreatic trypsinogen is activated by? What does it do?
enteropeptidase (enterokinase).
This enzyme cleaves a hexapeptide from trypsinogen to form trypsin.
Active trypsin can then activate?
the other pancreatic proenzymes
The bulk of protein digestion happens in the? by the action of?
lumen of the SI by the action of pancreatic proteases
What are three endopeptidases that cleave internal peptide bonds?
Trypsin’s
Chymotrypsin
Elastases
What are Carboxypeptidases A and B? What do they do?
exopeptidases
cleave free amino acids, one at a time from the C-terminus
Together the pancreatic proteases digest protein to?
oligopeptides (2-20 amino acids), small peptides (2-8 residues), and free amino acids.

Brush border peptidases are required for the breakdown of ?
larger peptides
Aminopeptidases are brush border exopeptidases that?
cleave amino acids one at a time from the amino end of the amino acid
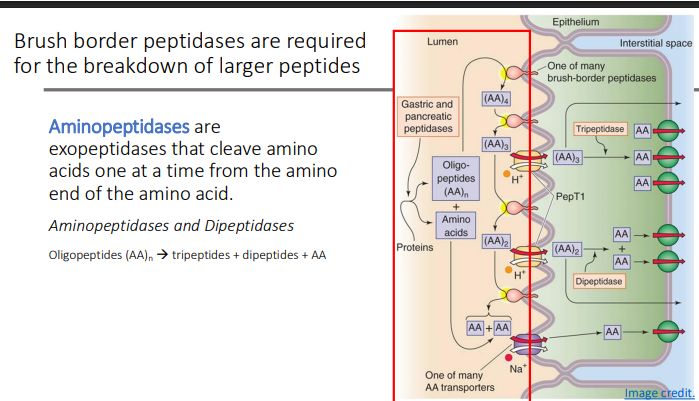
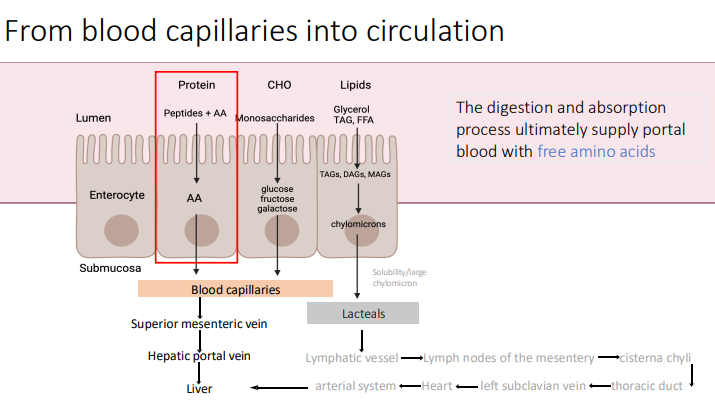
Tripeptides, dipeptides and free AA are taken up by?
enterocytes
Tripeptides and dipeptides undergo final hydrolysis to amino acids by?
cytosolic aminopeptidases.
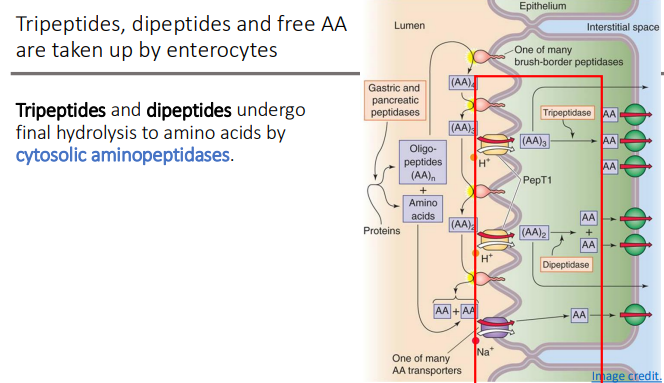
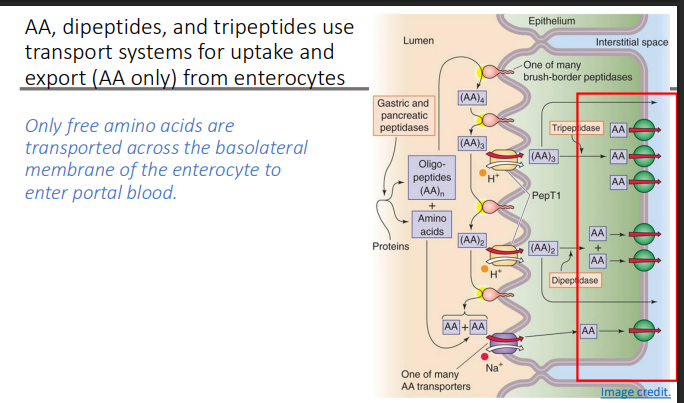
AA, dipeptides, and tripeptides use ____ for uptake and export (AA only) from enterocytes
transport systems
Only _____ are transported across the basolateral membrane of the enterocyte to enter portal blood
free amino acids
Separate carriers are present in the microvilli membrane to transport four groups of AA. What are the neutral AA?
Aromatic – tyrosine, tryptophan, histidine, and phenylalanine
Aliphatic - alanine, serine, threonine, valine, leucine, isoleucine, and glycine
Other – methionine, glutamine, asparagine, and cysteine
Separate carriers are present in the microvilli membrane to transport four groups of AA. What are the 3 basic AA?
Lysine
Arginine
Ornithine
Separate carriers are present in the microvilli membrane to transport four groups of AA. What are the 2 Dicarboxylic AA?
Glutamic and aspartic acids
Separate carriers are present in the microvilli membrane to transport four groups of AA. What are the 2 Imino AA?
Proline and hydroxyproline. Glycine may also use this carrier

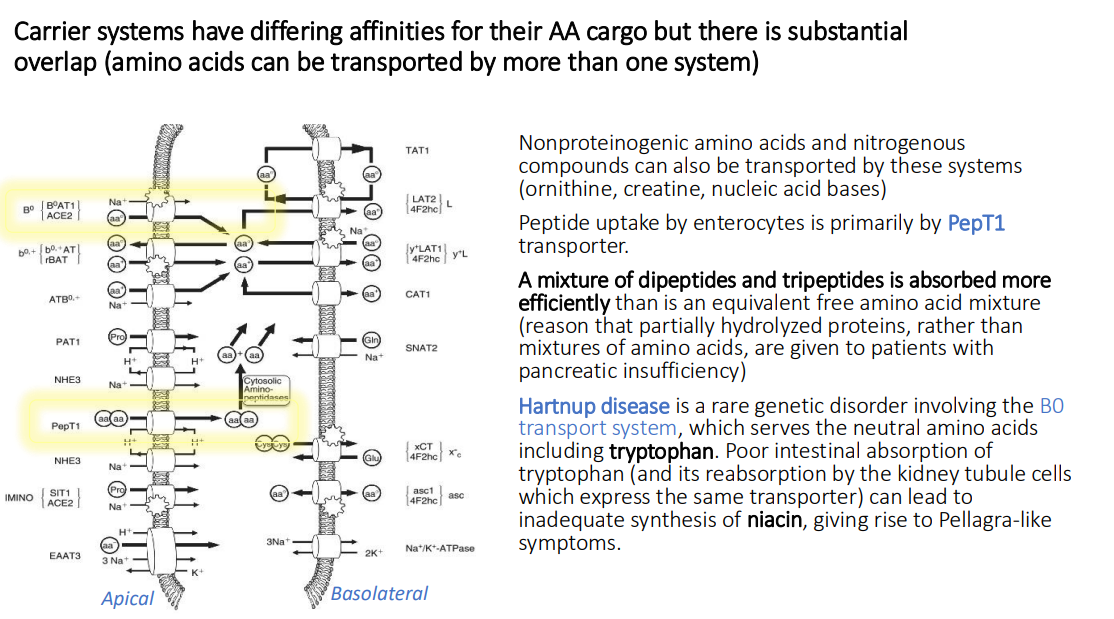
Carrier systems have differing affinities for their AA cargo but there is substantial overlap (amino acids can be transported by more than one system): What two things can also be transported by these systems?
Nonproteinogenic amino acids and nitrogenous compounds
(ornithine, creatine, nucleic acid bases)
Peptide uptake by enterocytes is primarily by which transporter?
PepT1
A mixture of ___ and _____ is absorbed more efficiently than is an equivalent free amino acid mixture
dipeptides; tripeptides
(reason that partially hydrolyzed proteins, rather than mixtures of amino acids, are given to patients with pancreatic insufficiency)
What is Hartnup disease?
a rare genetic disorder involving the B0 transport system, which serves the neutral amino acids including tryptophan
Poor intestinal absorption of tryptophan (and its reabsorption by the kidney tubule cells which express the same transporter) can lead to?
inadequate synthesis of niacin, giving rise to Pellagra-like symptoms
Most AA carrier systems are coupled with what for absorption?
sodium for absorption
Most AA carrier systems are coupled with sodium for absorption: The Na+ K + pump is maintained by an? whereby the concentration of Na+ is ___ outside the mucosal cell.
ATPase enzyme; greater
Most AA carrier systems are coupled with sodium for absorption: Both the AA and Na+ bind to the
membrane carrier and the complex transverses the membrane (Na+ goes down its concentration gradient)
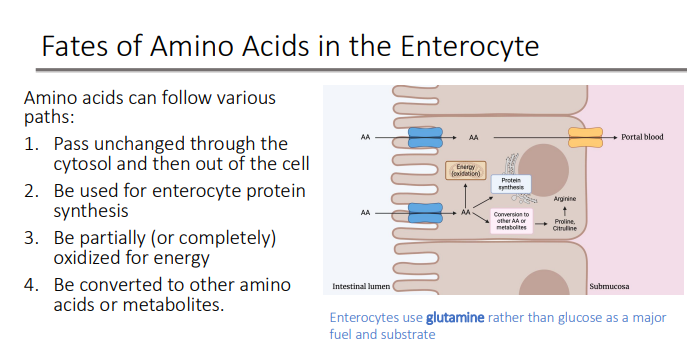
What are the 4 fates of AA in the enterocytes?
1. Pass unchanged through the cytosol and then out of the cell
2. Be used for enterocyte protein synthesis
3. Be partially (or completely) oxidized for energy
4. Be converted to other amino acids or metabolite
What do Enterocytes use instead of glucose as a major fuel and substrate?
glutamine
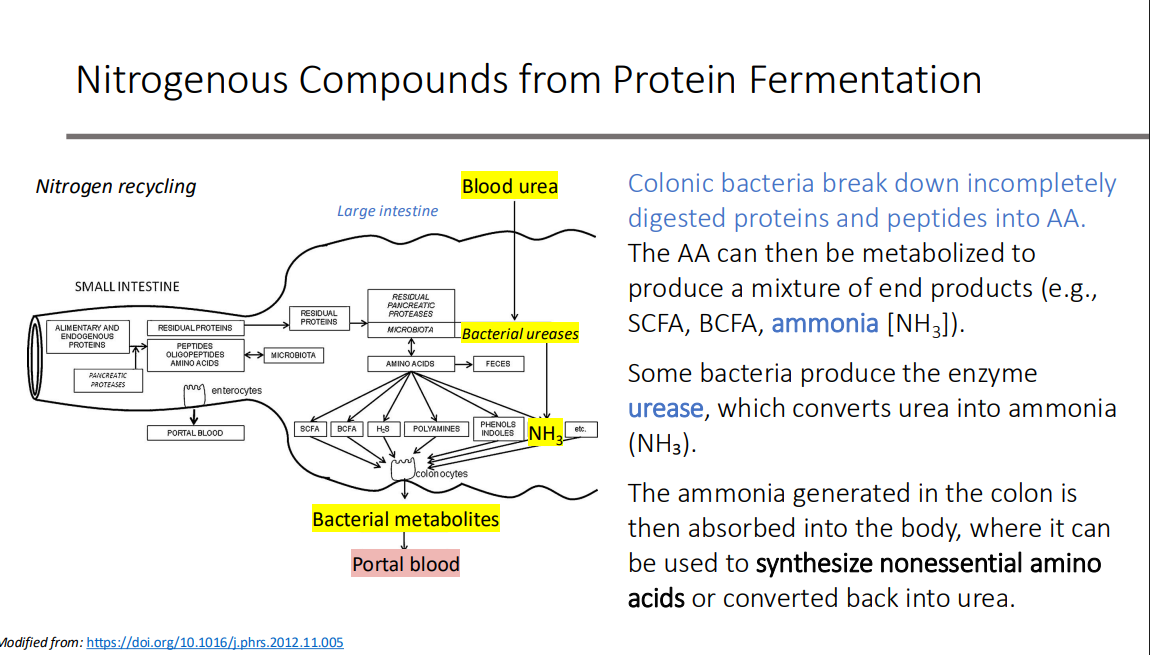
Nitrogenous Compounds from Protein Fermentation: What do Colonic bacteria break down?
incompletely digested proteins and peptides into AA.
Nitrogenous Compounds from Protein Fermentation: The AA can then be metabolized to produce?
a mixture of end products (e.g., SCFA, BCFA, ammonia [NH3 ]).
Nitrogenous Compounds from Protein Fermentation: Some bacteria produce the enzyme?
urease, which converts urea into ammonia (NH₃).
Nitrogenous Compounds from Protein Fermentation: The ammonia generated in the colon is then absorbed into the body, where it can be used to synthesize?
nonessential amino acids or converted back into urea.
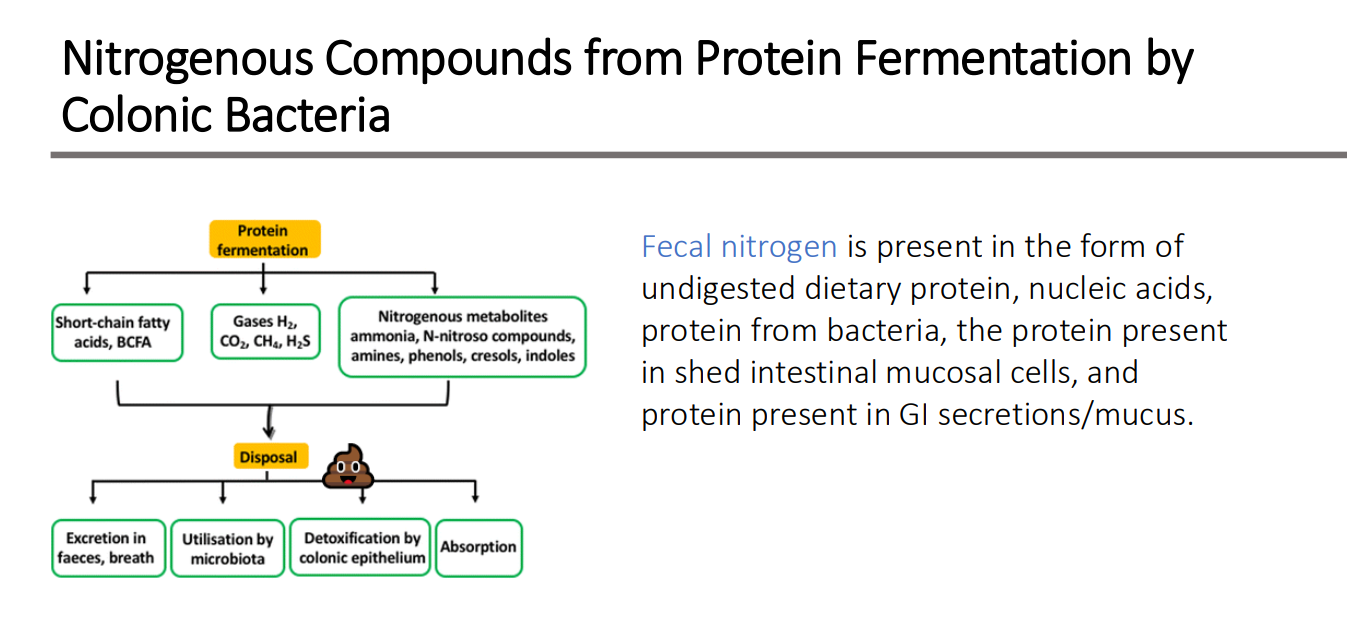
Nitrogenous Compounds from Protein Fermentation: Fecal nitrogen is present in the form of what 5 things?
undigested dietary protein,
nucleic acids,
bacteria,
shed intestinal mucosal cells
GI secretions/mucus

Which of the following are events that occur during protein digestion?
E. i, ii, v

Select TRUE statements about protein absorption and utilization in enterocytes.
E. i, ii