polymers of life
1/18
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
19 Terms
How does mass spec work
an organic compound is bombarded with electrons causing it to break up into different fragments some of these fragment are charge ions and are picked up by a mass spec and create a fragmentation pattern
what happens with the fragmentation pattern
the pattern is recorded on a mass spec with the mass/charge ion on the x axis and the abundance of each ion on the y axis
what does the fragmentation pattern tell us
can be used to determine the structure
if the charge of the fragment is +1 it will be equal to the mass of the fragment
what does the largest peak mean
the largest peak is the molecular mass of the whole organic molecule
what is the M+1 peak
the peak caused by the small proportion of organic molecules in a sample with cause carbon-13
what could peaks higher than M+1 mean
depend on isotopes in a compound
for example chlorine has to isotopes of 35Cl and 37Cl
what doesn’t mass spec detect
uncharged particles and particles that are two unstable and break down to quickly
how do you find the ‘lost fragments‘
by looking at the difference between two peaks and subtracting the smaller one from the bigger one
(Remember this wont have a charge)
what is different about high resolution mass spec
m/z values are given to 4 or more decimal places
why is high resolution mass spec useful
allows you to compare elements with similar Mrs as only carbon 12 is a whole number
or when several different molecules could have caused the same peak
how would you lay out mass spec in the exam

what is nuclear magnetic resonance (NMR) spectra
an analytical technique used to work out the structure
a sample of the compound is exposed to to a range of frequencies and wave lengths
how much is absorbed depends on the environment its in
the pattern can tell you about where it is
what happens to carbon in different environments
a nucleus is partly shielded from the radiation depending on what is around it so it absorbs different frequency
to be in the same environments carbons must be bonded to the same things in the entire molecule
what does the number of peaks tell you
the number of carbon environments there is
what is the chemical shift used for
to show what carbon environments there is
how would i show this in an exam
a grid with one row for the type of environment and another for the ppm
Write the number of carbons and the number of peaks
write about symmetry (if there is 4 carbons and two carbon enviroments)
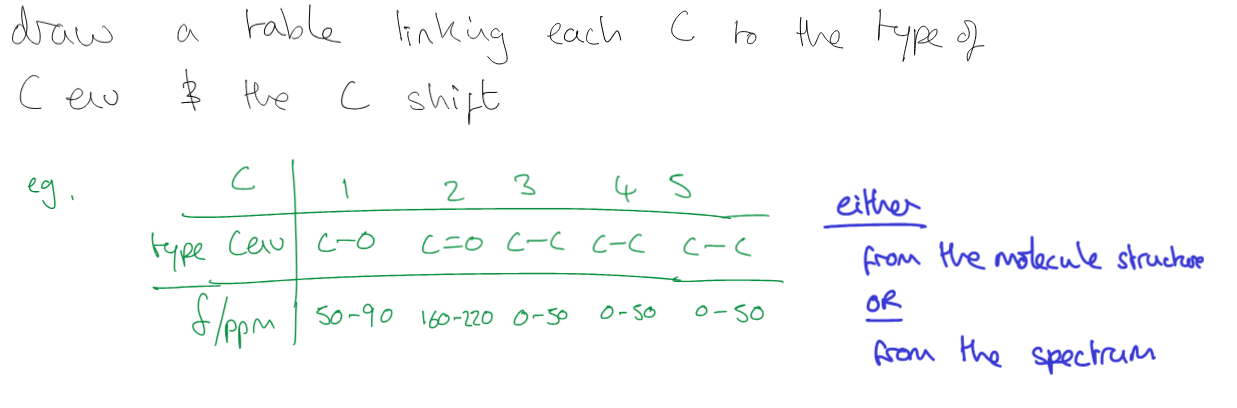
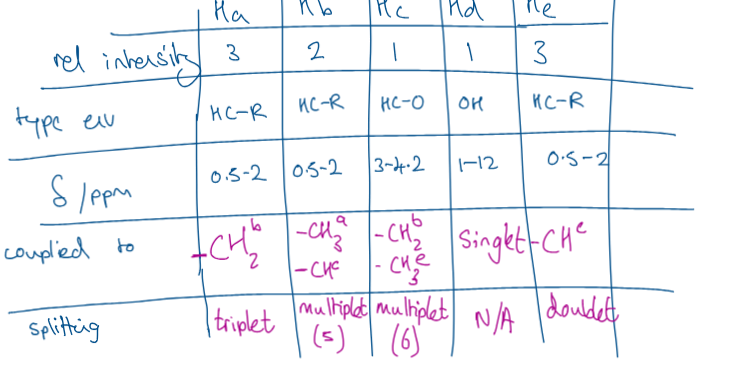
what 4 things do we look for in proton NMR (H1 NMR)
number of peaks=number of H environments’
ratio of peak areas (relative intensity)= number of H in each environment
chemical shift value = type of H environment
splitting pattern = number of H on next door carbon
what is splitting
tells us how many hydrogens are on the neighbouring carbon
O-H don’t get split or cause splitting

how would i show HNMR in and exam
relative intensity
type of environment
ppm
splitting
coupled to