Chymotrypsin mechanism
1/6
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
7 Terms
Step #1
SUBSTRATE BINDING: The peptide side chain fits into a hydrophobic pocket, aligning the peptide bond for attack
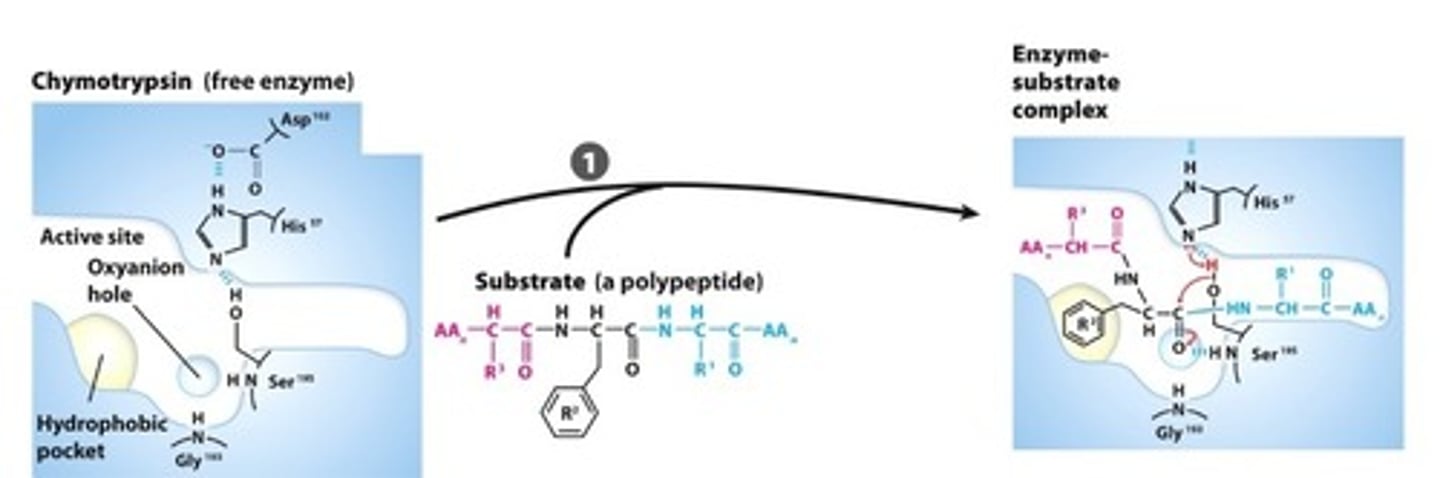
Step #2
NUCLEOPHILIC GENERATION: The Ser and His residue interaction create a Ser alkoxide, this nucleophile attacks the carbonyl carbon forming a tetrahedral acyl-enzyme intermediate that is stabilized bythe oxyanion hole
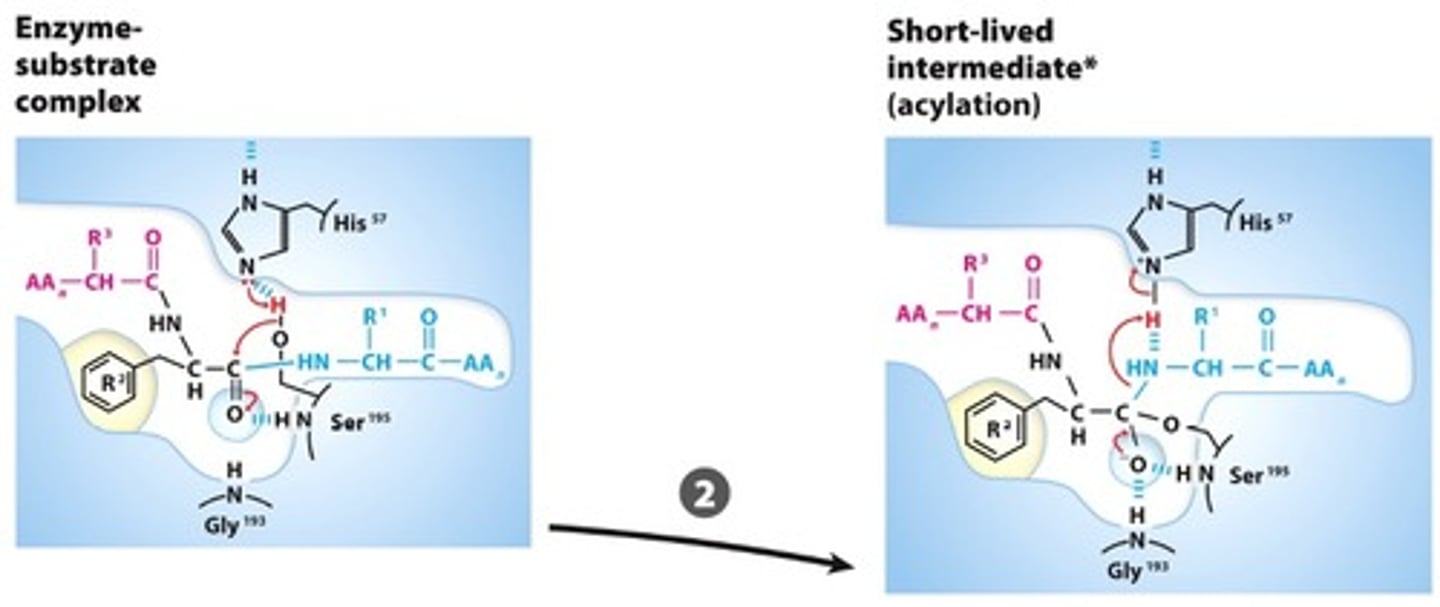
Step #3
ACYLATION: Collapse of the tetrahedral intermediate reformsthe carbonyl, releasing the amino terminal fragment and leaving an acyl Ser-95 ester
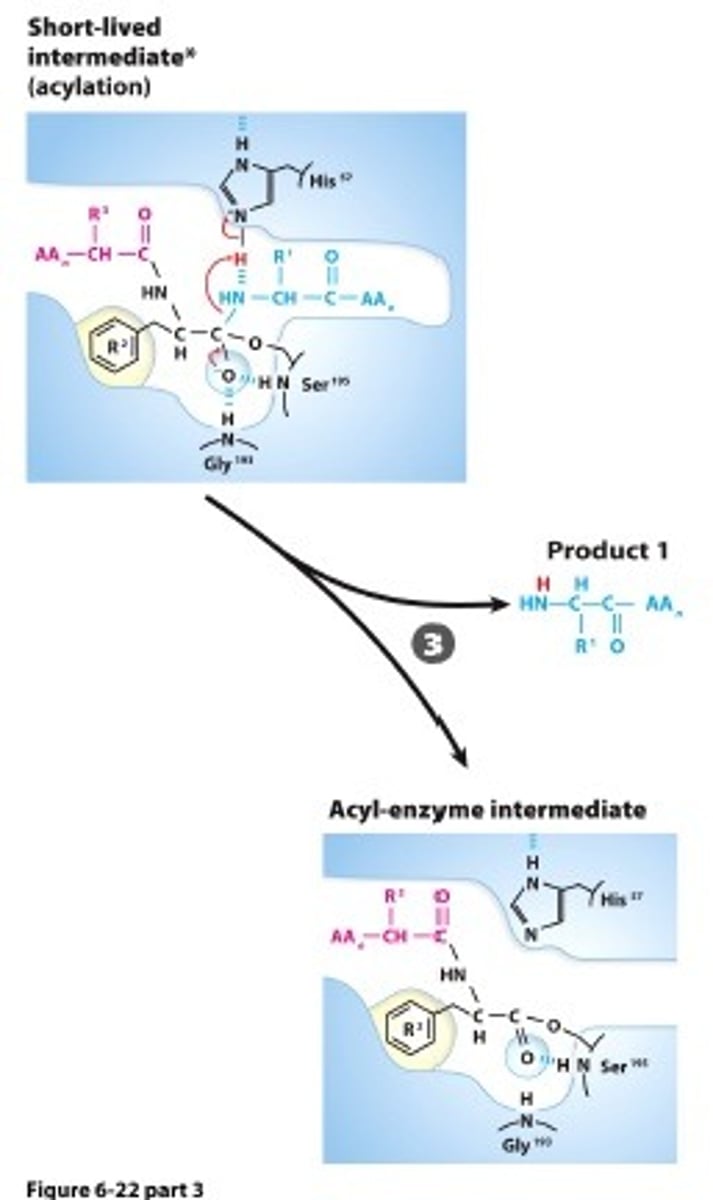
Step #4
WATER ACTIVATION: A water nolecule is deprotonated by the catalytic base that produces a hydroxide ion
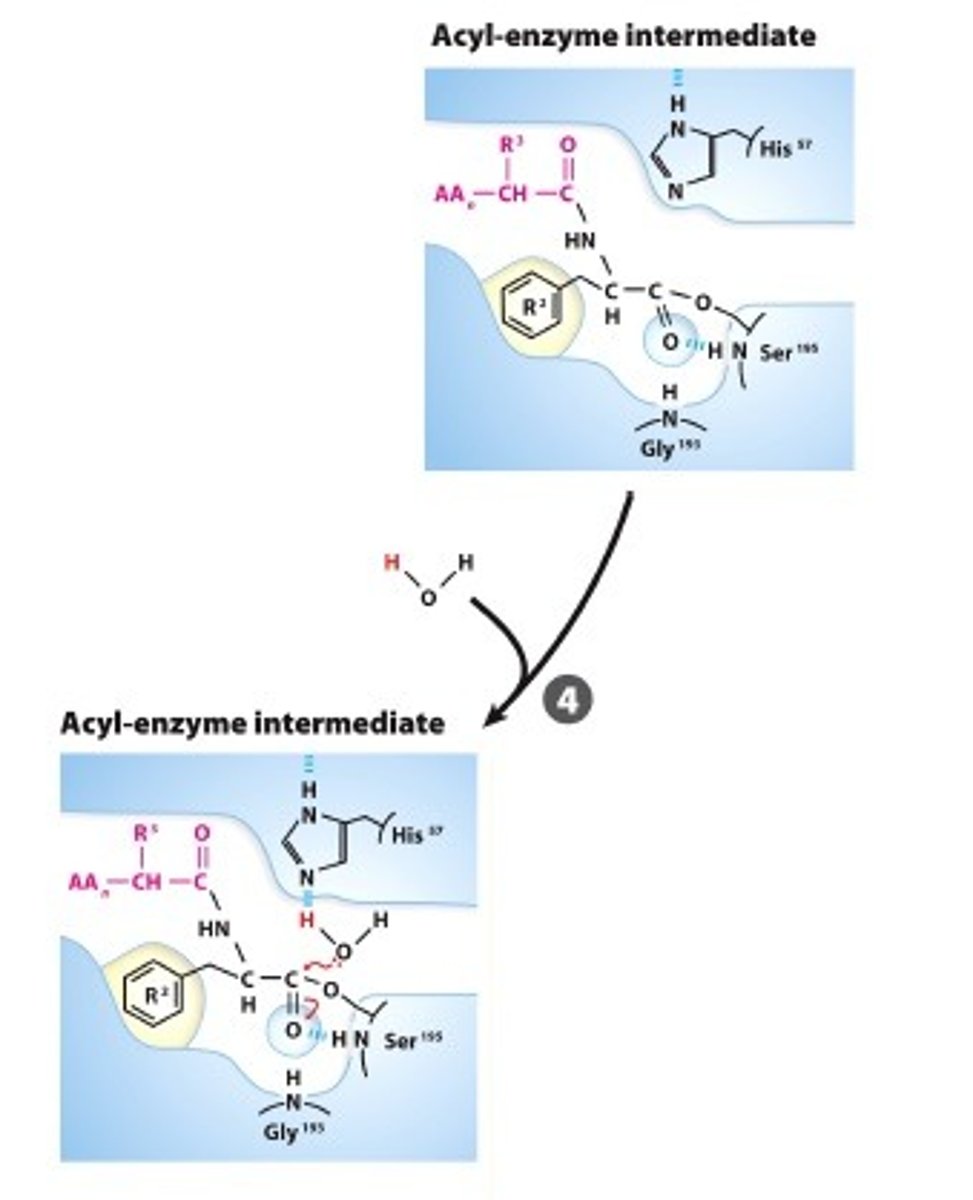
Step #5
DEACYLATION:The hydroxide attacks the acyl-Ser ester creating a 2nd tetrahedral intermediate that is stabilized in the oxyanion hole.
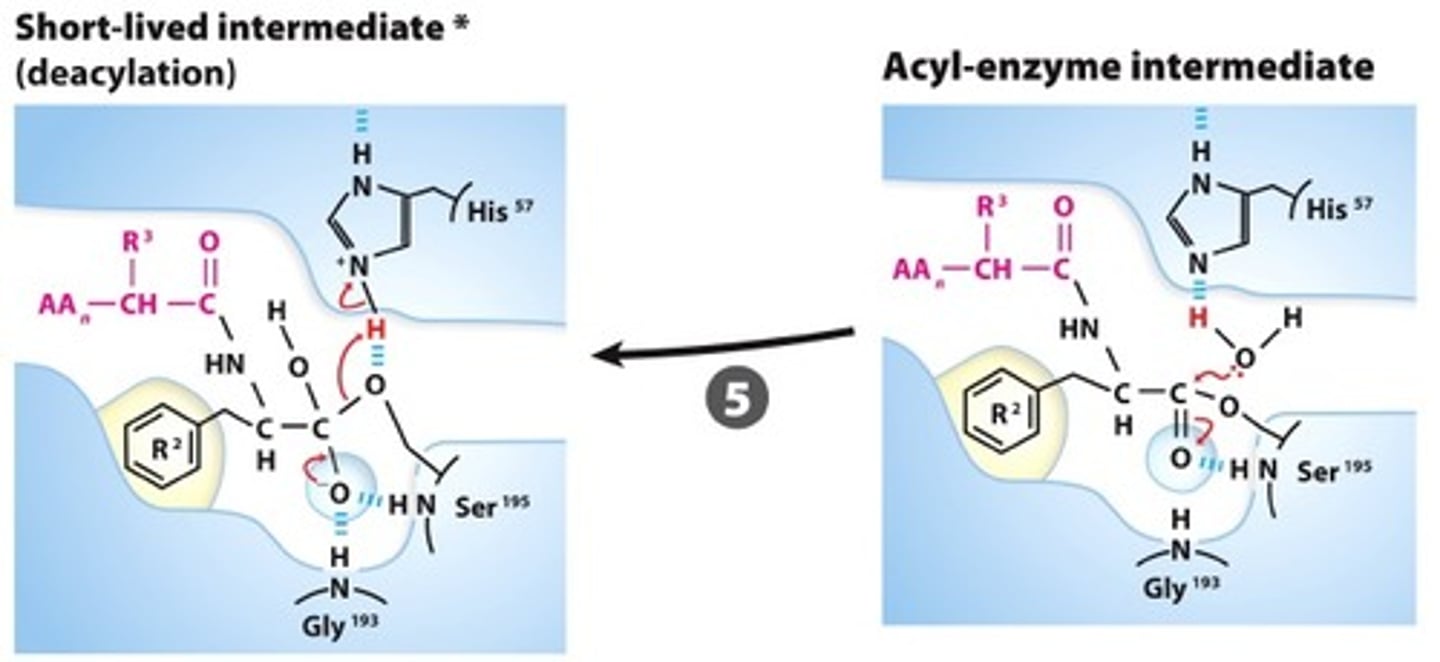
Step #6
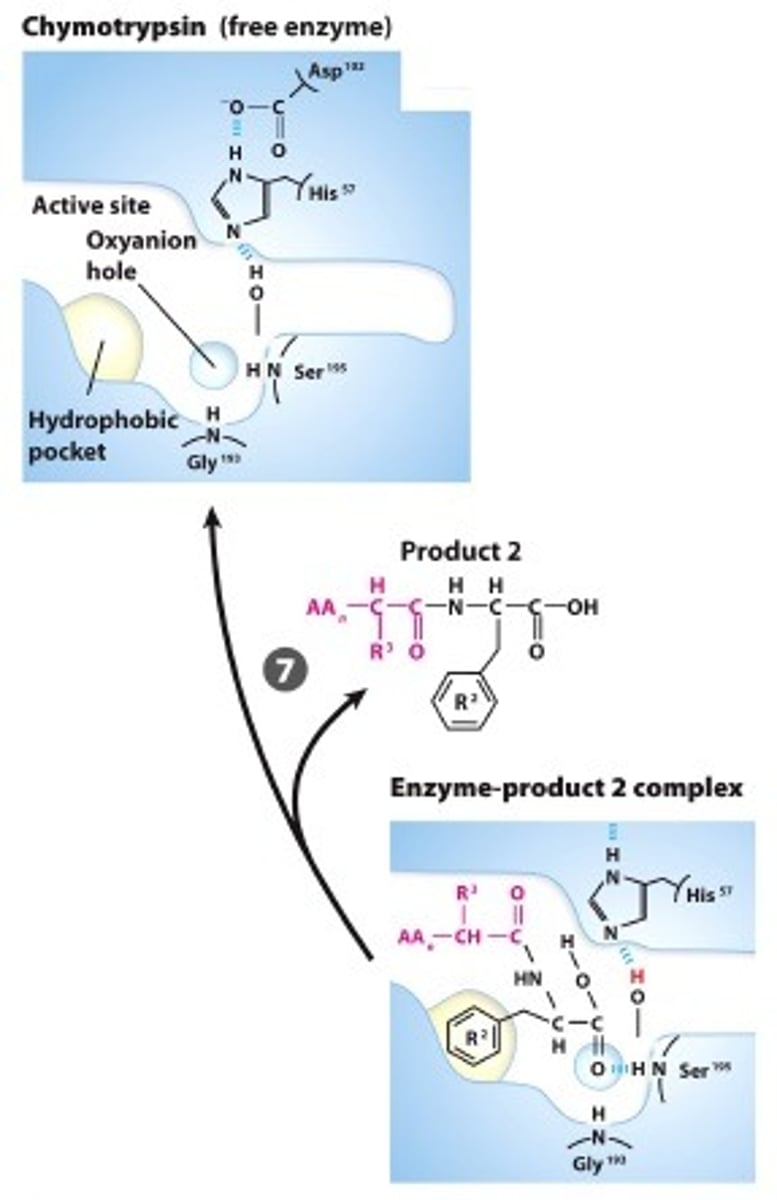
BREAK-OFF FROM THE ENZYME: The intermediate collapses, releasinf the carboxyl terminal fragment andrestoring the free Ser, reasy for another catalytic round
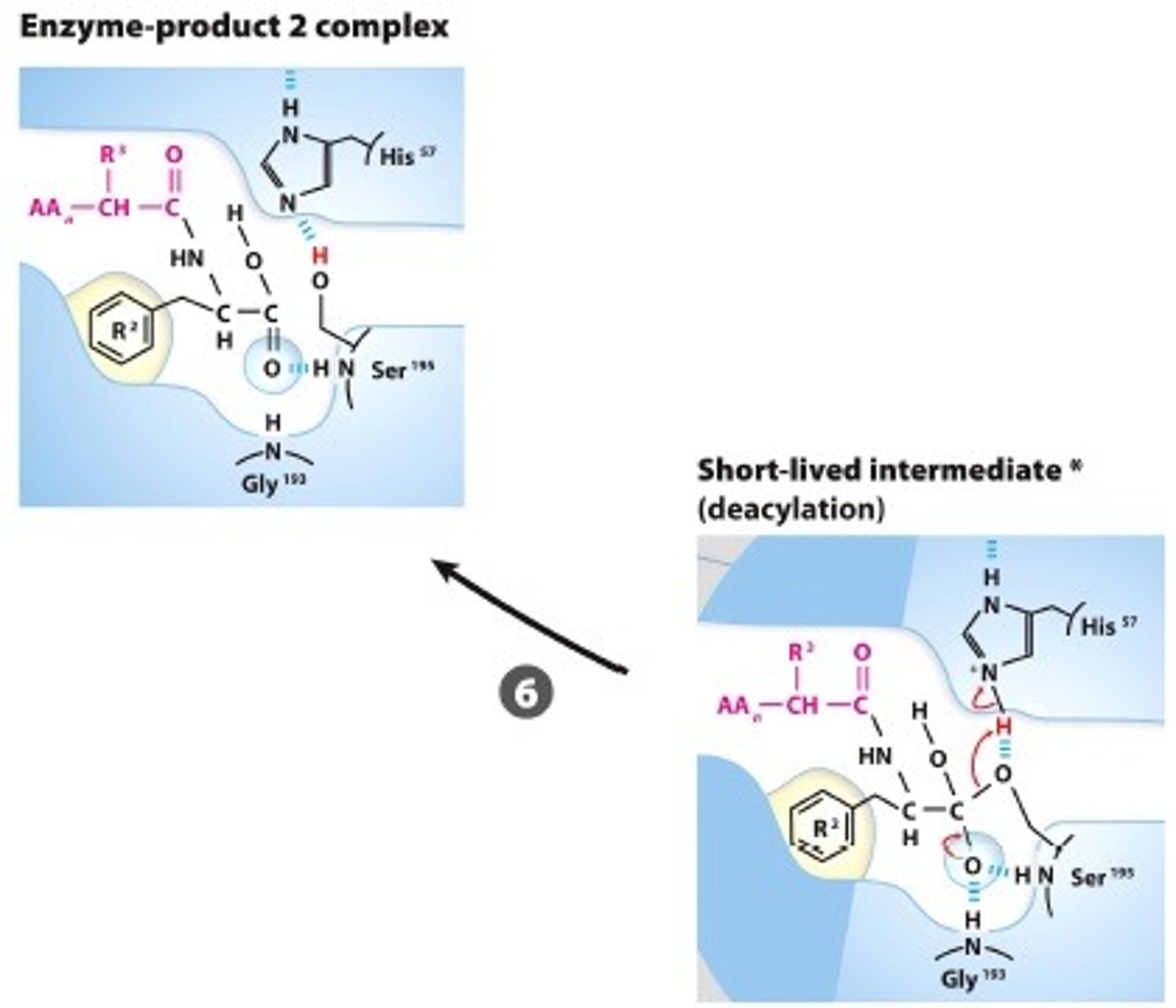
Step #7
PRODUCT DISSOCIATES:The product dissocates an the process starts over with a new substrate