Properties of Water
- Cohesion: the tendency of molecules of the same kind to stick together
\
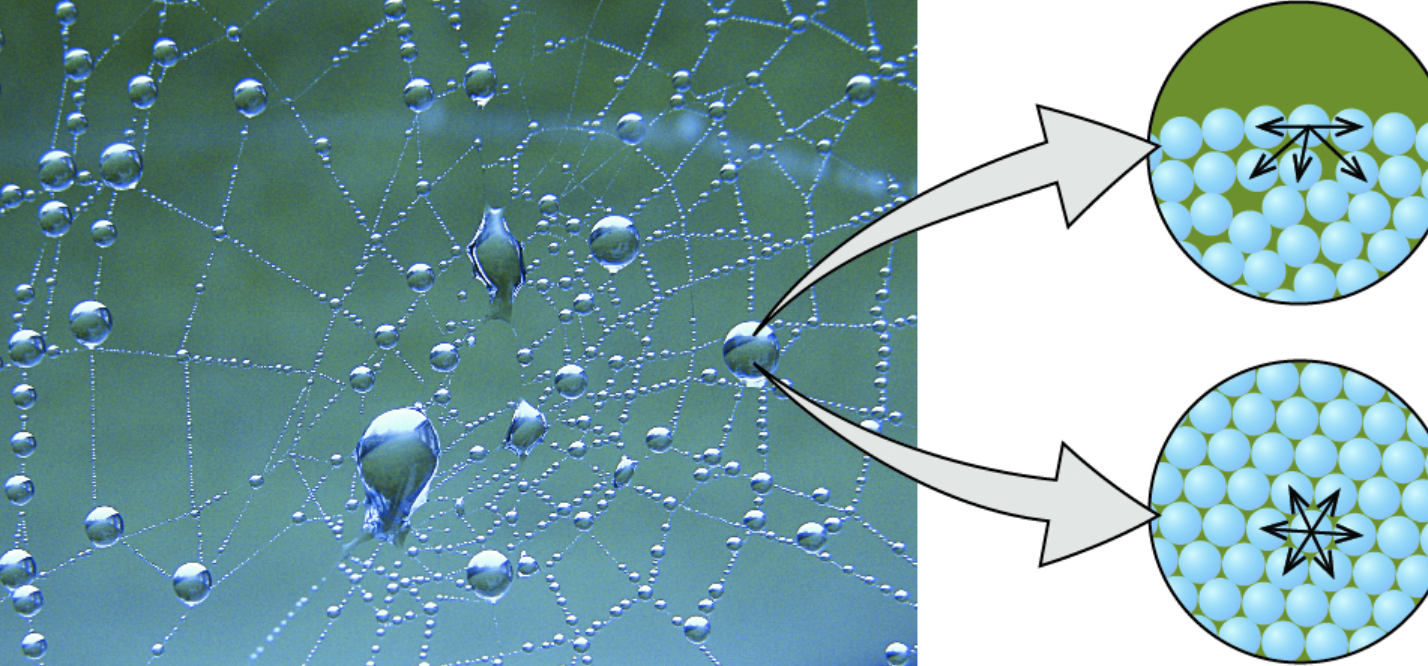
- Adhesion: the tendency of two different kinds of molecules to stick together * Capillary action: the ability to move something up a surface * Ex: a straw in a glass of water has water slightly higher than the rest: the water rises using adhesion towards the straw, and the rest of the water rises using cohesion
- Surface tension: a measure of how difficult it is to break the surface of a liquid * Hydrogen bonds cause water to stay together
\


\
- Moderate temperature * Thermal energy: energy associated with the random movements of atoms and molecules * Heat: transfer of thermal energy from hot → cold * Temperature: measure of intensity of moving atoms * A lot of heat needed to be absorbed for water to change temperature * High heat capacity * Raising temperatures → bonds must break to vibrate faster, despite high surface tension * Lowering temperatures → bonds must break to evaporate, despite high surface tension
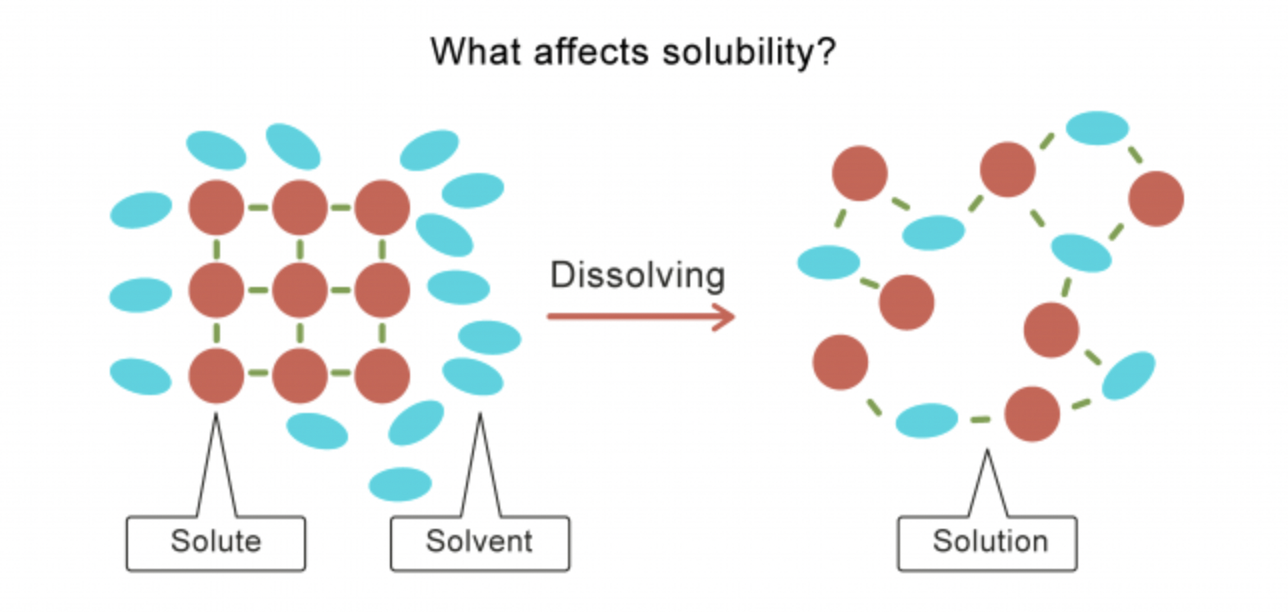
- Major solvent * Solvent: dissolving agent * Solute: dissolved substance * Solution: solvent + solution * Aqueous solution: solution where the solvent is water * Charge of the water molecules breaks about other molecules, causing them to dissolve
\
\