md7.2 - spindle fibres and spindle poisons
1/113
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
114 Terms
what is the 3rd step of mitosis?
anaphase
what happens to the duplicated chromosomes during anaphase?
the duplicated chromosomes separate from each other
where do the chromosomes move to during anaphase?
they move towards the two spindle poles at opposite ends of the cell
what is the purpose of chromosome movement during anaphase?
to ensure each future daughter cell receives a complete and equal set of chromosomes.
: What drives chromosome movement towards the spindle poles during anaphase?
A: Spindle microtubules shorten and pull the chromosomes towards the poles.
What are the two types of microtubule movement that occur during anaphase?
Anaphase A — where the kinetochore microtubules shorten, pulling chromosomes towards the poles, and
Anaphase B — where the spindle poles themselves move further apart, increasing the separation between the two sets of chromosomes.
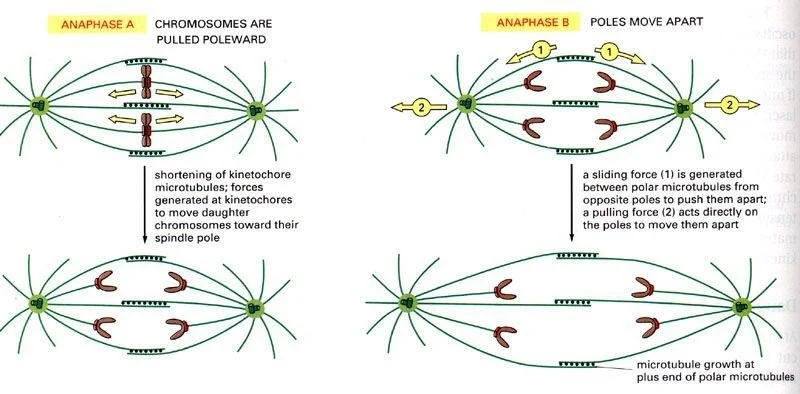
: What is the overall outcome of anaphase?
Two complete sets of chromosomes are pulled to opposite ends of the cell, ready for two new daughter cells to form.
Why is accurate chromosome separation during anaphase so important?
If chromosomes fail to separate correctly, daughter cells may end up with too many or too few chromosomes, which can lead to cell death or diseases such as cancer.
What is the fourth step of mitosis?
Telophase.
What has happened to the chromosomes by the start of telophase?
The duplicated chromosomes have reached their new spindle poles at opposite ends of the cell.
What is happening to the cell during telophase?
The cell is in the process of dividing.
What happens to the chromosomes during telophase?
They begin to decondense, returning from their compact state back to a more relaxed form.
What happens to the nuclear envelope during telophase?
A new nuclear envelope reforms around each set of chromosomes, creating two new nuclei.
What happens to the spindle microtubules during telophase?
They begin to break down as they are no longer needed to separate the chromosomes.
: What follows telophase?
Cytokinesis — the physical splitting of the cell into two separate daughter cells.
What is the overall outcome of telophase?
Two new nuclei have formed, each containing a complete set of chromosomes, and the cell is ready to fully divide into two genetically identical daughter cells.
What are antimitotic drugs?
Drugs that target microtubules to disrupt mitosis, ultimately leading to mitotic arrest and cell death.
Where do antimitotic drugs bind on tubulin?
They bind to binding sites on the surface of the globular part of tubulin.
What are microtubule targeting agents (MTAs)?
Agents that bind to tubulin and alter microtubule dynamics, ultimately leading to mitotic arrest and cell death.
How do MTAs cause cell death?
: By altering microtubule dynamics, they disrupt the normal process of mitosis, causing the cell to arrest in mitosis and eventually die.
Where do many antimitotic compounds originate from
Many come from natural sources including plants, fungi and invertebrates.
How many MTA binding sites are known?
6 binding sites are known in total.
How are the 6 MTA binding sites distributed between α and β tubulin?
4 are on β-tubulin and 2 are on α-tubulin.
What are the four binding sites found on β-tubulin?
The taxane, laulimalide/peloruside, vinca, and maytensine binding sites.
What are the two binding sites found on α-tubulin?
The colchicine and pironetin binding sites.
hat are the two overall types of microtubule targeting agents?
Microtubule stabilising agents (MSAs) and microtubule destabilising agents (MDAs).
What is the difference between MSAs and MDAs?
MSAs stabilise microtubules and prevent them from shrinking, whilst MDAs destabilise microtubules and promote their breakdown.
Q: Why are antimitotic drugs useful in medicine?
Because by disrupting mitosis and causing cell death, they can be used to target and kill rapidly dividing cancer cells.
Microtubule Stabilizing Agent (MSA’s) | Microtubule Destabilizing Agents (MDA’s) |
Taxane ligands | Vinca ligands |
Laulimalide/peluroside ligands | Maytensine ligands |
Colchicine ligands | |
Pironetin ligands |
What is paclitaxel?
A tetracyclic diterpenoid and a widely used microtubule targeting agent in cancer therapy.
hat is paclitaxel used to treat?
It is used in diverse cancer therapies, not just ovarian cancer.
: What are the side effects of paclitaxel?
Myelosuppression (suppression of bone marrow activity, which is reversible) and peripheral neuropathy (damage to the peripheral nerves).
What is myelosuppression?
A reduction in bone marrow activity leading to fewer blood cells being produced. In the case of paclitaxel this side effect is reversible.
What is peripheral neuropathy?
Damage to the peripheral nerves, which can cause pain, numbness or weakness, particularly in the hands and feet.
: Where is the taxane binding pocket located on tubulin?
: It is located near the internal (lumen-facing) surface of the microtubule, on the inside of the tubulin.
How does paclitaxel stabilise microtubules?
: It strengthens the lateral contacts between adjacent protofilaments, leading to microtubule stabilisation.
what are profilaments?
Is the mode of action the same for all compounds that bind to the taxane pocket
No, the exact mode of action is compound specific.
What is the difference between direct stabilisation and an allosteric mechanism in relation to paclitaxel?
Direct stabilisation involves paclitaxel physically strengthening contacts between protofilaments,
whereas an allosteric mechanism means paclitaxel causes a conformational change elsewhere in the tubulin that prevents the dimer from compacting, indirectly stabilising the microtubule.
Do taxanes bind well to soluble (free floating) tubulin?
No, taxanes bind poorly to soluble tubulin but with high affinity to the binding site on the β-tubulin subunit within assembled microtubules.
Where specifically does paclitaxel bind on the microtubule?
: It binds to the β-tubulin subunit on the inside (lumen-facing surface) of the assembled microtubule.
Why is it significant that paclitaxel binds to assembled microtubules rather than free tubulin?
It means paclitaxel specifically targets microtubules that are already built, rather than preventing tubulin from assembling in the first pla
How does paclitaxel access its binding site on the inside of the microtubule?
It gains access through small gaps or by taking advantage of natural fluctuations in the microtubule lattice structure.
What conformational change does paclitaxel cause upon binding?
It causes a shape change in tubulin that increases the lateral binding affinity between neighbouring protofilaments, though the exact mechanism is not fully understood.
How many paclitaxel binding sites are there per tubulin dimer?
There is one binding site per tubulin dimer.
What is meant by near 1:1 stoichiometry in relation to paclitaxel binding?
It means that in principle, full stabilisation would require approximately one paclitaxel molecule for every tubulin dimer in the microtubule.
Does paclitaxel need to occupy every binding site to have a significant effect?
No, just one paclitaxel molecule per several hundred tubulin dimers is enough to reduce the rate or extent of microtubule shortening by 50
What concentration of paclitaxel is needed to block mitosis by 50% in HeLa cells?
Just 8 nanomoles per litre (8 nM) is sufficient to block mitosis in 50% of HeLa cells.
What is the key mechanism by which paclitaxel disrupts mitosis and causes cell death?
It is the disruption to microtubule dynamics, not any increase in polymer mass, that is crucial for interrupting mitosis and ultimately causing cell death.
Why is disruption of microtubule dynamics so damaging to the cell?
ecause dynamic microtubules are essential for correct chromosome separation during mitosis. Without proper dynamics the cell cannot divide correctly and ultimately dies.
What does this tell us about the importance of microtubule dynamics in mitosis?
It shows that microtubules must be able to grow and shrink dynamically for mitosis to proceed — simply having more or larger microtubules is not enough, and disrupting their dynamics alone is sufficient to kill the cell.
What are laulimalide and peloruside,
They are microtubule stabilising agents (MSAs)
Where is the laulimalide/peloruside binding site located, and how does this compare to the taxane site?
: It is located on the outside of the microtubule wall, on the opposite side to the taxane binding site which is on the inside (lumen-facing) of the microtubule.
How do laulimalide and peloruside stabilise microtubules?
hey strengthen lateral contacts between neighbouring protofilaments, and uniquely there is evidence they can interact with both their host tubulin dimer and the adjacent tubulin dimer, potentially bridging neighbouring units.
What are the natural vinca alkaloids?
Vincristine and vinblastine.
What was the clinical significance of the first generation vinca alkaloids?
They achieved significant clinical success, most notably against childhood leukaemia.
What is a notable example of a semi-synthetic vinca alkaloid analogue?
Vinorelbine is a notable semi-synthetic analogue derived from the first generation vinca alkaloids.
What are the side effects of vinca alkaloids
Peripheral neuropathy and myelosuppression, which is reversible.
: How does the side effect profile of vinca alkaloids compare to taxanes?
They share a similar side effect profile, both causing peripheral neuropathy and reversible myelosuppression.
Why was the success of vinca alkaloids against childhood leukaemia significant?
It demonstrated that plant-derived compounds could be highly effective cancer treatments, paving the way for the development of diverse semi-synthetic analogues and further natural product drug discovery.
Where is the vinca binding site located on tubulin?
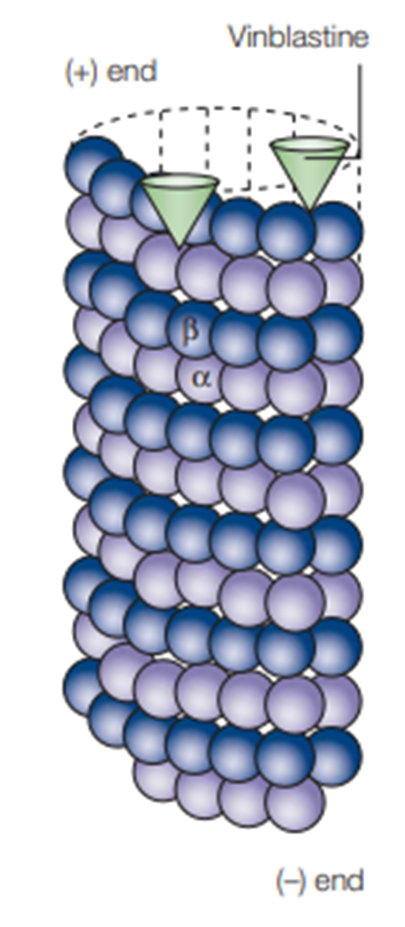
t the plus end surface of β-tubulin.
What structural change does vinca binding cause at the plus end?
It alters the surface of the (+) end, forming a "wedge" shape.
How does the "wedge" formation affect microtubule growth?
It interferes with the incorporation of new heterodimers, preventing the microtubule from growing.
What happens to the (+) ends of microtubules when vinca alkaloids bind?
The (+) ends remain curved, preventing the microtubule wall from forming correctly.
What additional effect can vinca site ligands have on free tubulin?
They can cause tubulin to form oligomers, which decreases the pool of free soluble tubulin available for microtubule growth.
Why is the formation of tubulin oligomers significant?
By reducing the free tubulin pool, it further limits the cell's ability to build new microtubules, compounding the destabilising effect.
What happens to microtubules at high concentrations of vinca alkaloids?
At high concentrations (e.g. 10-100 nM in HeLa cells) microtubules depolymerise and break down completely.
How many vinca ligand molecules are needed to significantly reduce microtubule dynamics?
Just one or two ligands at the end of a microtubule are sufficient to reduce dynamics by 50%.
What is the key mechanism by which vinca alkaloids exert their biological activity?
It is the suppression of microtubule dynamics, rather than complete depolymerisation, that is the key to their biological activity.
How does the mechanism of vinca alkaloids compare to that of paclitaxel?
Both work primarily by suppressing microtubule dynamics rather than simply increasing or decreasing the total amount of microtubule polymer. However vinca alkaloids are destabilising agents (MDAs) that prevent growth, whilst paclitaxel is a stabilising agent (MSA) that prevents shrinkage.
Are vinca alkaloids classed as MSAs or MDAs?
: They are microtubule destabilising agents (MDAs), as they prevent microtubule growth and promote depolymerisation at high concentrations.
maytansine site:
How does the location of the maytansine binding site compare to the vinca site?
The maytansine site is close to the vinca binding site but is structurally distinct, being formed from different parts of the tubulin protein, making it a genuinely separate binding site.
: What is the normal function of the maytansine binding pocket in a growing microtubule?
A: In a normally growing microtubule, the maytansine binding pocket at the (+) end is the space where the (-) end of the next α-tubulin unit slots in during growth, meaning it is the site of tubulin addition.
How does maytansine impede microtubule elongation?
by binding to and occupying the pocket where the next tubulin unit would normally attach, maytansine physically blocks new tubulin from being added, preventing the microtubule from growing.
What is the current clinical status of maytansine and its derivatives?
Maytansine and its derivatives are currently under clinical evaluation as potential cancer treatments.
: Where specifically does eribulin bind on microtubules?
It mainly binds with high affinity to the (+) end of existing microtubules.
What cancers is eribulin currently approved to treat?
Eribulin is used in the treatment of metastatic breast cancer and unresectable liposarcoma.
Eribulin is used in the treatment of metastatic breast cancer and unresectable liposarcoma.
Eribulin also shows non-mitotic effects including vascular remodelling which reduces tumour hypoxia, as well as decreased metastasis and invasion.
What is tumour hypoxia and why is it significant that eribulin reduces it?
Tumour hypoxia is when cancer cells are deprived of oxygen, which can make them more aggressive and resistant to treatment. By reducing hypoxia through vascular remodelling, eribulin may make tumours more vulnerable to treatment.
Why are eribulin's non-mitotic effects significant?
They suggest eribulin can attack cancer through multiple mechanisms beyond just disrupting cell division, potentially making it a more effective and wide ranging treatment.
What range of solid tumours is eribulin currently being investigated for
t is being investigated for breast cancer, non-small cell lung cancer (NSCLC), prostate, brain, cervical, and other solid tumours.
How does eribulin differ from other microtubule destabilising agents such as vinca alkaloids?
Binding affinity and specificity — eribulin binds with particularly high affinity to the (+) end, whereas vinca alkaloids bind to a specific vinca pocket nearby
Additional effects — eribulin has non-mitotic anticancer effects that vinca alkaloids do not
Vinca alkaloids (e.g. vincristine, vinblastine)
Bind to the vinca site on β-tubulin at the (+) end
Form a "wedge" that prevents new heterodimers from being incorporated
At high concentrations cause full depolymerisation
Are microtubule destabilising agents (MDAs)
Eribulin
Also binds to β-tubulin at the (+) end
But binds with uniquely high affinity specifically to the (+) end of existing microtubules
Has additional non-mitotic effects such as vascular remodelling, reducing tumour hypoxia, and decreasing metastasis and invasion
Is also a microtubule destabilising agent (MDA)
What is colchicine currently used clinically to treat?
: It is used clinically to treat gout.
why is colchicine not widely used as an anti-tumour agent?
Because the doses required to achieve anti-tumour effects cause severe toxicity, making it too dangerous for cancer treatment.
Where is the colchicine binding site located on tubulin
It is located between the α and β tubulin subunits, at the intradimer interface.
Q: How would you describe the binding kinetics of colchicine?
Colchicine binds slowly but the binding is practically irreversible, meaning once bound it stays bound.
What conformational effect does colchicine binding have on the tubulin heterodimer?
It stabilises the heterodimer in a curved conformation.
: What normally happens to tubulin conformation during polymerisation?
During normal polymerisation the tubulin transitions from a curved conformation at the tip of the microtubule to a straight conformation in the body of the microtubule.
How does colchicine binding prevent polymerisation?
By stabilising the curved conformation of the heterodimer, colchicine prevents the transition from curved to straight that is necessary for the tubulin to be incorporated into the microtubule wall, thereby inhibiting polymerisation.
Where does colchicine bind first — at the microtubule tip or in the soluble pool?
it is thought that colchicine binds to tubulin in the soluble pool first, and the colchicine-tubulin complex is then incorporated into the microtubule, rather than colchicine binding directly at the microtubule tip.
What is the effect of tubulin-colchicine units being incorporated into the microtubule?
Their presence slows down polymerisation but does not prevent it entirely, meaning the microtubule can still grow but at a reduced rate.
Q: How does colchicine's mechanism differ from vinca alkaloids?
vinca alkaloids bind to the (+) end of existing microtubules and form a wedge that blocks new heterodimer addition, whereas colchicine binds to tubulin in the soluble pool first, locking it in a curved conformation that impairs polymerisation when incorporated into the microtubule.
: Why is the irreversible nature of colchicine binding significant?
Because once colchicine binds to a tubulin unit it permanently locks it in the curved conformation, meaning that tubulin unit will always impair microtubule polymerisation whenever it is incorporated, making colchicine a particularly potent and long lasting inhibitor.
Why don't microtubule targeting agents work against all cancers?
Different cancers have different properties and mechanisms of resistance, meaning a drug that is highly effective against one cancer type may be essentially ineffective against another.
Which cancers is paclitaxel particularly effective against?
Paclitaxel is very effective against ovarian, mammary (breast) and lung tumours.
Which cancers is paclitaxel largely ineffective against?
Paclitaxel is essentially ineffective against many solid tumours such as colon and kidney carcinomas.
Which cancers are vinca alkaloids most effective against?
Vinca alkaloids are effective against many haematological (blood) cancers such as leukaemia.
What is a notable limitation of vinca alkaloids?
They are usually ineffective against solid tumours.
What are the four main possible mechanisms of resistance to microtubule targeting agents?
Overexpression of membrane transport proteins (ABC-transporters), microtubule associated proteins, post-translational modifications, and up and/or down regulation of different tubulin isotypes.
What are ABC-transporters and how do they cause resistance?
ABC-transporters are membrane proteins that pump drugs out of cells. Overexpression of these proteins in cancer cells means the drug is removed before it can have its effect, reducing its efficacy.