A&P CH2 PG14-15
1/45
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
46 Terms
What is a buffer?
A system that:
Resists pH changes
Accepts or releases H+ ions
Keeps pH stable
Why are buffers necessary?
Without it:
Small amounts of acid/base would cause dramatic pH changes
Cells would be damaged
Enzymes would stop working
What are components of a buffer?
They contain:
A weak acid
A weak base
These work together to control H+ levels
What happens if blood becomes too acidic?
The base part of the buffer responds
It binds H+ and removes it (because acidic = too many H+ ions)
Once removed, pH roses back to normal
What is the main buffer system in blood?
Bicarbonate-Carbonic Acid Buffer System
What is the equation for the bicarbonate/carbonic acid buffer system?
CO2 + H2O ←→ H2CO3 ←→ H+ + HCO3-
What are the components of the Bicarbonate -Carbonic Acid Buffer System? (3)
Carbonic acid
Bicarbonate
Acid
Name these components:
H2CO3
HCO3-
H+
Carbonic acid (H2CO3)
Bicarbonate (HCO3-)
Acid (H+)
When blood is too _____ too many _+ present
Acidic
H+
Bicarbonate binds __ when blood is too acidic
H+
what does the equation look like when blood is too acidic (Low pH)
H+ + HCO3- → H2CO3
When blood is too _____, there are too little _+
Basic
H+
What does the equation look like when blood is too Basic (High pH)?
H2CO3 → H+ + HCO3-
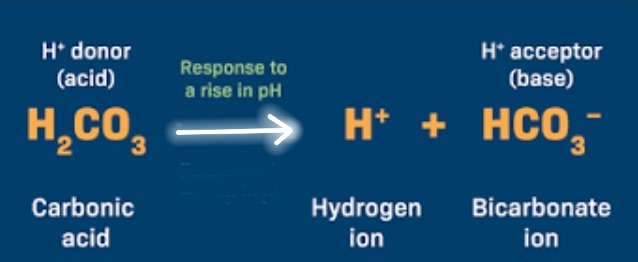
What is:
HCO3-
Bicarbonate
What is:
H2CO3
Carbonic Acid
What does Bicarbonate do?
Removes excess acid
Raises pH
Acts as a base
What does carbonic acid do ?
Releases H+
Lowers pH
Acts as acid
If bicarbonate binds H+, what happens to pH?
pH rises
Less acidic
H+ decreases
If bicarbonate binds to H+ and the pH rises, is this a positive or negative feedback?
Negative feedback
What are inorganic compounds
No carbon back bone
What are examples of inorganic compounds
Water
Salts
Acids
Bases
What are organic compounds?
Compounds that contain carbon + hydrogen
Built around carbon chains
What are examples of carbon compounds
Carbohydrates
Lipids
Proteins
Nucleic acids
Organic chemistry = _ _ _ _
Life
Carbohydrates
C H O
What are the elements in all carbohydrates?
Carbon
Hydrogen
Oxygen
What is the usual ratio for C H O?
1:2:1
Ex: C6H12O6
What is the major function of carbohydrates?
Energy source
Carbohydrates are classified by the number of sugar units they contain. Name three:
Monosaccharides (simple sugars)
Disaccharides (double sugars)
Polysaccharides (many sugars)
What is a monosaccharide?
A single sugar molecule
The simplest form of carbohydrate
Cannot be broken down into smaller sugars
Name three examples of monosaccharides:
Glucose
Fructose
Galactose
What is a simple sugar?
A sugar made of one unit
Easily absorbed
Quickly used for energy
What is a disaccharide?
Two monosaccharides bonded together
Formed by dehydration synthesis
Name three types of disaccharides:
Sucrose (table sugar)
Lactose (milk sugar)
Maltose (grain sugar)
How is SUCROSE formed?
Glucose + fructose
How is LACTOSE formed?
Glucose + Galactose
How is MALTOSE formed?
Glucose + Glucose
What happens when disaccharides are formed?
Water is released in each reaction.
What is a polysaccharide?
A long chain of monosaccharides
Many sugars bonded together
Name three types of polysaccharides:
Glycogen
Starch
Cellulose
What is the function of glycogen?
Energy storage in animals
What is the function of starch?
Energy storage in plants
What is the function of cellulose
Structural support in plants
Anabolic or catabolic?
Building polysaccharides from glucose
Anabolic
Hydrolysis or dehydration?:
Building polysaccharides from glucose
Dehydration synthesis
If starch is broken down to glucose, water is added to starch in a process called __________
Hydrolysis