Materials Science Flashcards
1/26
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
27 Terms
Gray Iron
Carbon: 2.5–4.0% C, 1.0–3.0% Si
Structure: Graphite as flakes in ferrite or pearlite matrix
Properties:
Weak and brittle in tension (flake tips = stress concentrators)
Strong under compression
Excellent vibration damping
High wear resistance
Cheap and easy to cast
Uses: Machine bases, engine blocks, cylinder heads, clutch plates, pipes
Ductile (Nodular) Iron
Carbon: 3.5–3.8% C, with Mg/Ce addition
Structure: Graphite as spherical nodules in pearlite or ferrite matrix
Properties:
High tensile strength (414–827 MPa)
Good ductility (up to 18% elongation)
Approaches steel in mechanical performance
Uses: Crankshafts, gears, valves, pump bodies, automotive components
White Iron
Carbon: locked as cementite (Fe₃C), no free graphite; <1.0% Si, fast cooled
Structure: Cementite dominant, no graphite present
Properties:
Extremely hard and wear resistant
Very brittle, virtually unmachinable
No plastic deformation
Uses: Rolling mill rollers; intermediate for malleable iron production
Malleable Iron
Carbon: 2.3–2.7% C; white iron reheated 800–900°C
Structure: Temper carbon rosettes/clusters in ferrite or pearlite matrix
Properties:
Better ductility than white iron
Good strength
Limited to thin sections (must solidify white first)
Uses: Connecting rods, transmission gears, pipe fittings, railroad components
Compacted Graphite Iron (CGI)
Carbon: 3.1–4.0% C, 1.7–3.0% Si, lower Mg/Ce than ductile iron
Structure: Wormlike/vermicular graphite (up to 20% nodules) in pearlite or ferrite matrix
Properties:
Strength between gray and ductile iron
Higher thermal conductivity than other cast irons
Better thermal shock resistance
Lower oxidation at high temperatures
Uses: Diesel engine blocks, exhaust manifolds, brake discs, flywheels
Ferrous alloy’s
Ferrite (α iron)BCC (cubic) stable at < 912˚C
• Austenite (γ iron) FCC (cubic) stable at 727 ˚C -1493 ˚C
• Cementite (Fe3C) Compound (orthorhombic ceramic, ratio 3:1)
• Delta iron (δ iron) BCC (cubic) stable at 1394 ˚C -1538 ˚C
• Pearlite (α + Fe3C)
• Ledeburite (γ + Fe3C)
Steel (General)
Carbon: <2.14% C
Structure: Iron-carbon alloy, carbon content determines properties
Properties: High tensile strength, ductile, durable, but rusts/corrodes
Uses: Structural applications across all industries
Types of Carbon Steel
Low Carbon Steel (<0.25% C)
Structure: Ferrite and pearlite matrix; cannot form martensite
Properties:
Soft and weak but excellent ductility and toughness
Machinable and weldable
Least expensive steel
Uses: Car body panels, I-beams, pipelines, bridges, tin cans
Low Carbon Steel - HSLA variant
Structure: Low carbon with added Cu, V, Ni, Mo (up to 10 wt%)
Properties:
Higher strength than plain low carbon
Ductile, formable, machinable
Better corrosion resistance
Uses: Bridges, towers, high-rise columns, pressure vessels
Medium Carbon Steel (0.25–0.60% C)
Structure: Tempered martensite after heat treatment (austenitize, quench, temper)
Properties:
Stronger than low carbon but less ductile
Good wear resistance and toughness
Heat treatable with Cr, Ni, Mo additions
Uses: Railway wheels, gears, crankshafts, machine parts
High Carbon Steel (0.60–1.4% C)
Structure: Hardened and tempered; carbide compounds with alloying elements
Properties:
Hardest and strongest of carbon steels
Least ductile
Excellent wear resistance; holds sharp edge
Uses: Cutting tools, knives, razors, hacksaw blades, springs, dies
Stainless Steel
Carbon: Varies; minimum 11% Cr required
Structure: Martensitic, ferritic, or austenitic depending on composition
Properties:
Highly corrosion and rust resistant
Wide range of mechanical properties
Strengthened by precipitation hardening
Uses: Gas turbines, steam boilers, aircraft, nuclear power units, medical equipment
Cast Iron (General)
Carbon: >2.14% C (typically 3.0–4.5% in practice)
Structure: High carbon causes graphite formation; type depends on cooling rate and composition
Properties:
Lower melting point than steel (1150–1300°C); easy to cast
Some types very brittle
Note: Parent category for gray, ductile, white, malleable, and CGI
Ceramics (General)
Structure: Ionic and/or covalent bonding; anions form scaffold, cations fill interstices; anions are larger than cations
Properties: Hard, brittle, high melting point, chemically inert
Key concept: The shape and size of interstices (tetrahedral vs octahedral) determines what structures are possible
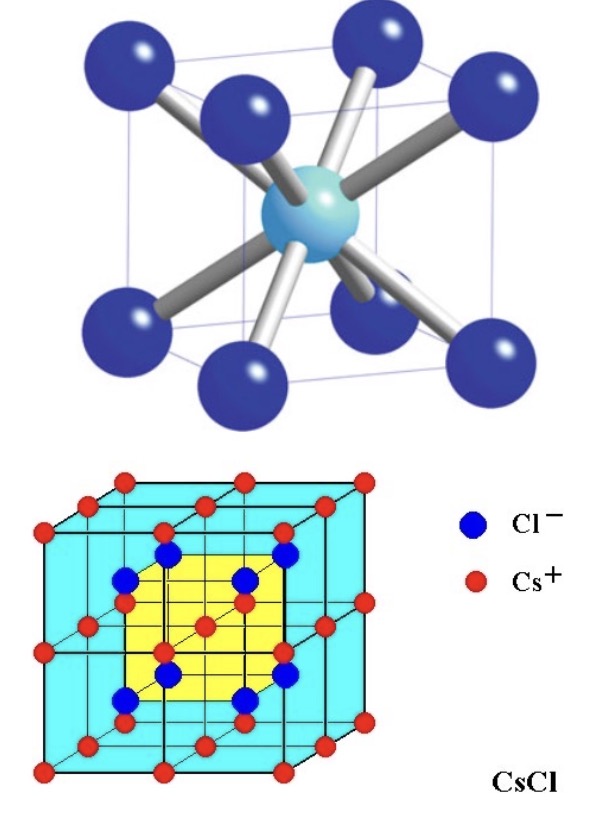
CsCl
Structure: Simple cubic (sc); CN = 8 for both ions
Key concept: Simplest ceramic structure; does not occur in oxides
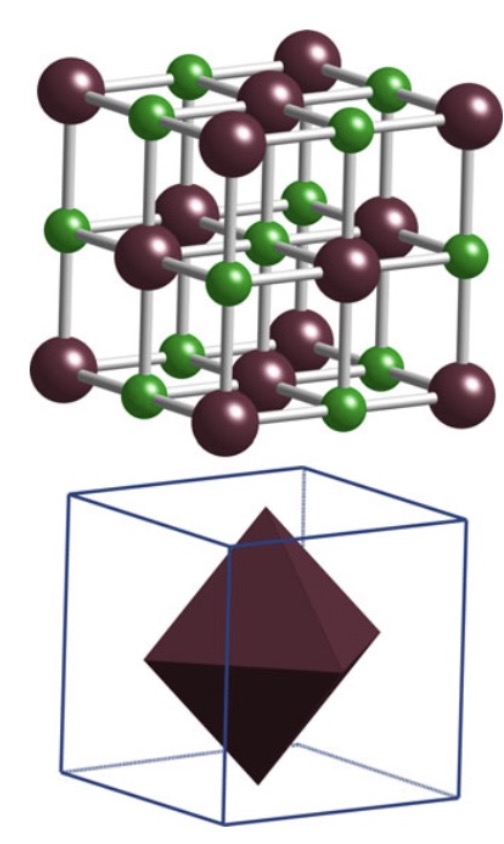
NaCl / Rock Salt (MgO, TiC, PbS)
Structure: fcc anion arrangement; cations fill all octahedral interstices; CN = 6 for both ions
Properties: Carbides with this structure are hard, chemically inert, high melting point
Examples: MgO, CaO, FeO, NiO, TiC
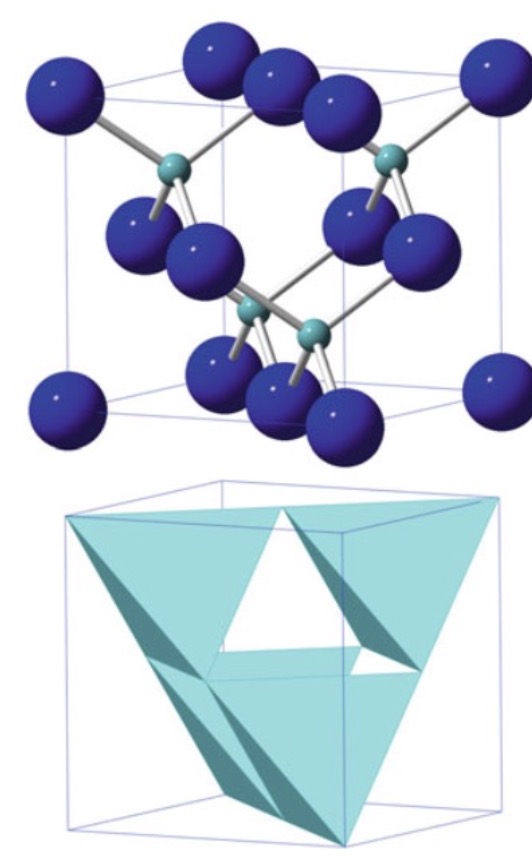
GaAs / Zinc Blende (β-SiC)
Structure: fcc anions; cations fill half the tetrahedral interstices; CN = 4 for both ions; open structure (APF = 0.41)
Properties: Usually semiconductors due to covalent bonding; band gap increases with more ionic character
Key concept: Replacing all atoms with same element gives diamond-cubic structure (C, Si, Ge)
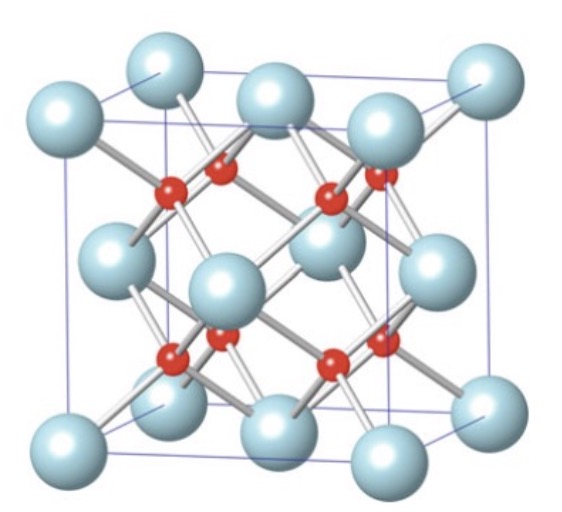
CaF₂ / Fluorite
Structure: fcc cations; anions fill all tetrahedral interstices; Ca CN = 8, F CN = 4
Properties: Transparent to deep-UV light
Uses: Semiconductor lithography lenses; cubic zirconia (CZ) is a diamond simulant
Key concept: UO₂ has this structure; the empty interstice at center can hold nuclear fission byproducts without straining the lattice
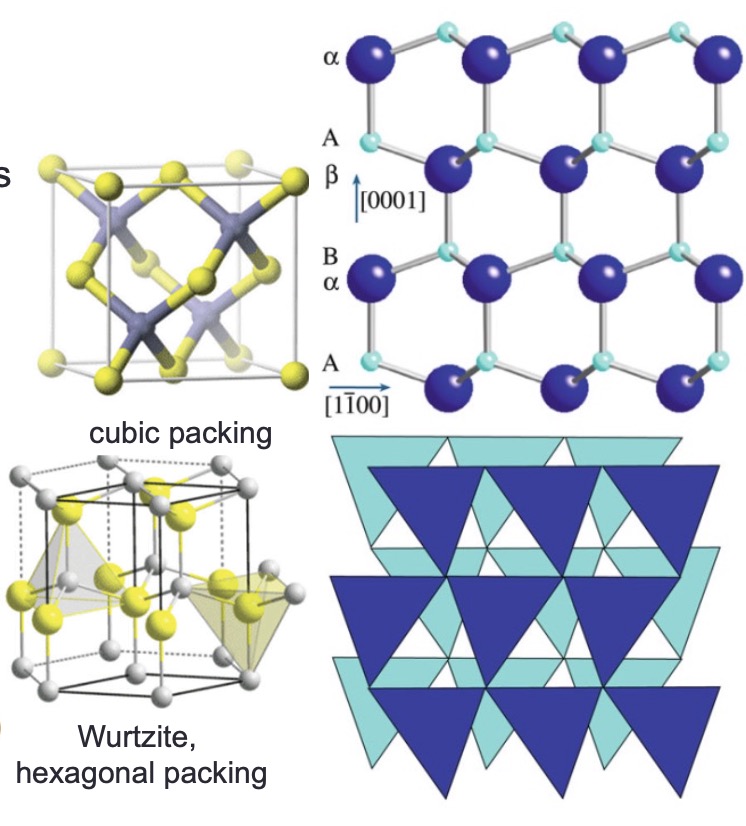
AlN / Wurtzite (BeO, ZnO)
Structure: hcp anions; cations fill half tetrahedral interstices; CN = 4; tetrahedra stack ABABAB (vs zinc blende ABCABC)
Properties: High thermal conductivity (BeO, AlN); ZnO is a semiconductor used in varistors
Key concept: Same nearest-neighbor environment as zinc blende but only one unique stacking direction [0001]
Types of Polymers
Homopolymer - A-A-A-A-A
Alternating Copolymer - A-B-A-B-A-B
Periodic Copolymer - A-B-B-A-B-B-A-B-B
Statistical Copolymer - A-B-B-B-A-B-A-B
Block Copolymer - A-A-A-A-B-B-B-B
Grafted Copolymer (A-A-A) with side chains of (B-B-B…)
What is a cross-linked polymer?
A polymer where bonds form between chains (usually covalent); requires at least 4 chains to emanate from a junction point
What are examples of complex polymer architectures?
Star, comb, brush, dendronized, dendrimer, and ring polymers
Polyethylene (PE)
(C₂H₄)ₙ; nonpolar, saturated, thermoplastic; highest global production
Low strength, high ductility, low friction, prone to creep
Melts 120–180°C; excellent electrical insulator; resists acids/bases; absorbs almost no water
Transparency varies with crystallinity level
HDPE vs LDPE
HDPE (≥0.941 g/cm³): low branching → tight packing → stronger intermolecular forces → stiffer, stronger; used in pipes, jugs, toys
LDPE (0.910–0.940 g/cm³): high branching → loose packing → weaker forces → flexible, ductile; used in bags, film wrap
PE-X (Cross-linked PE)
Cross-links convert PE from thermoplastic to thermoset
Improved heat resistance, chemical resistance, reduced creep/flow
Methods: peroxide (200–250°C), silane (Si–O–Si bridges), irradiation (β/γ), azo compounds
Used in plumbing, cable insulation, automotive
Polypropylene (PP)
Thermoplastic; carbon backbone with CH₃ on alternating carbons; lowest density commodity plastic (0.895–0.92 g/cm³)
CH₃ group improves mechanical strength and thermal resistance vs PE, but reduces chemical resistance (tertiary carbon is more reactive)
Isotactic (crystalline, Tm ~171°C) vs atactic (amorphous, lower Tm)
Resists fats/solvents at room temp; dissolves in xylene/tetralin when hot
Tough, flexible, good fatigue resistance; translucent to opaque
PVC
Carbon backbone with Cl on alternating carbons; polar C–Cl bond; thermoplastic; 3rd highest global production
Rigid (uPVC, 1500–3000 MPa) or flexible (plasticized, 1.5–15 MPa)
Cl atoms give excellent flame retardancy (LOI ≥45; releases HCl suppressing combustion)
Poor inherent thermal stability; decomposes at 140°C; requires heat stabilizers
Electrical insulation inferior to PE/PP due to polarity
Resists acids, bases, salts, fats; some solvents (THF, acetone) can damage it
PTFE (Teflon)
Carbon backbone fully surrounded by F atoms; nonpolar fluoropolymer; thermoplastic
C–F bond = second strongest single bond in organic chemistry → extremely chemically inert
Lowest coefficient of friction of any solid; hydrophobic due to weak London dispersion forces
Melts at 327°C; maintains toughness down to −268°C
Only attacked by alkali metals and strong fluorinating agents
Used in non-stick coatings, chemical pipework, surgical grafts, catheter coatings
PMMA (Acrylic/Plexiglas) Polymethyl methacrylate
Atactic, completely amorphous thermoplastic; transparent (no crystalline regions to scatter light)
Transmits up to 92% visible light; density 1.17–1.20 g/cm³ (less than half of glass)
Good impact strength vs glass; inferior to polycarbonate
Ester groups easily hydrolyzed → poor solvent resistance; swells/dissolves in many organics
Superior UV/environmental stability vs polystyrene and PE → preferred for outdoor use
Max water absorption 0.3–0.4% by weight; tensile strength decreases with moisture
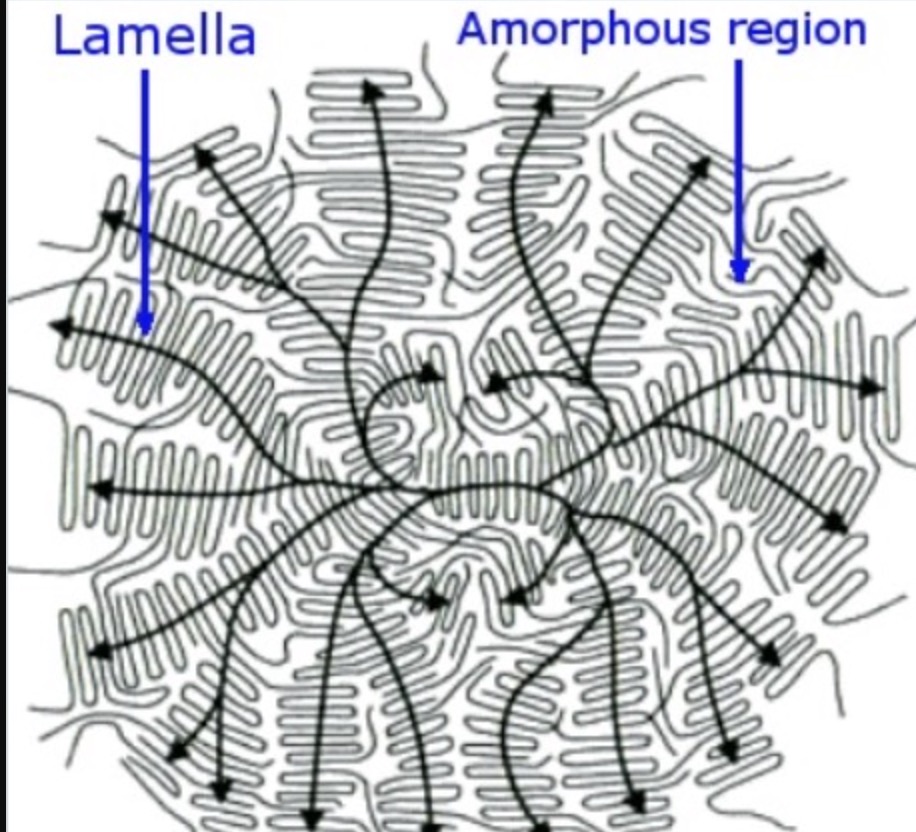
Ordered region in a polymer
lamellae