Physl 210 B Post Midterm Content
1/201
Earn XP
Description and Tags
Renal, Endocrine, Reproductive
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
202 Terms
Kidney function
Regulators of blood volume and pressure (what we keep/what we excrete).
Kidney excretory products
Urea (from protein breakdown), uric acid (from NT breakdown), creatinine (from muscle use/breakdown) and bilirubin (from Hb breakdown)
Kidneys secrete (endocrine function)
Erythropoietin, Vitamin D (active form = 1, 25 dihydroxy), renin
Kidneys synthesize
Glucose (in a fasting state)
Where is the majority of water?
In the intracellular space (40% of bodyweight out of 60% total)
Where is the remaining water
Circulating in extracellular fluid (interstitial space, 3L is from plasma alone - 20% of 60% bodyweight total)
Compounds/ions commonly found outside of the cell
Na, Cl, HCO3
Compounds/ions commonly found inside the cell
K, Mg, Pi, protein
Osmosis
The net diffusion of water from an area of high concentration to an area of low concentration (from low Osm to high Osm, until they equalize).
Tonicity
Determined by the number of Non-penetrating solutes (Na, K) of an extracellular solution in relation to an intracellular solution
Isotonic
The number of NPS is the same both inside and outside the cell

Hypotonic
The number of NPS is higher inside the cell, causing water to rush in and the cell to swell (eventually bursting)
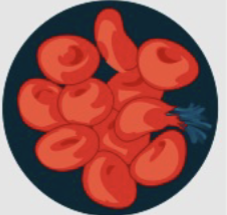
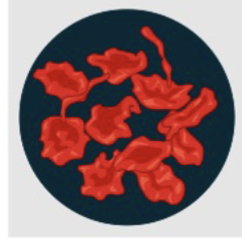
Hypertonic
The number of NPS is higher outside the cell compared to inside, causing the cell to shrink (as fluid rushes out to equalize).
Normal osmolarity number
300 mOsm/L
Ultimate goal of the Kidneys
Maintain homeostasis (balancing the water gained (ingested, produced) vs water lost (sweat, breathing, etc))
Micturition
How the voiding of urine is controlled in the body (ANS)
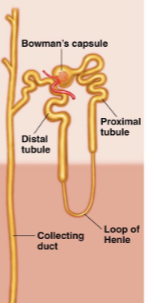
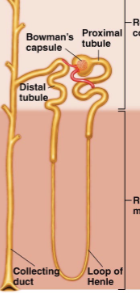
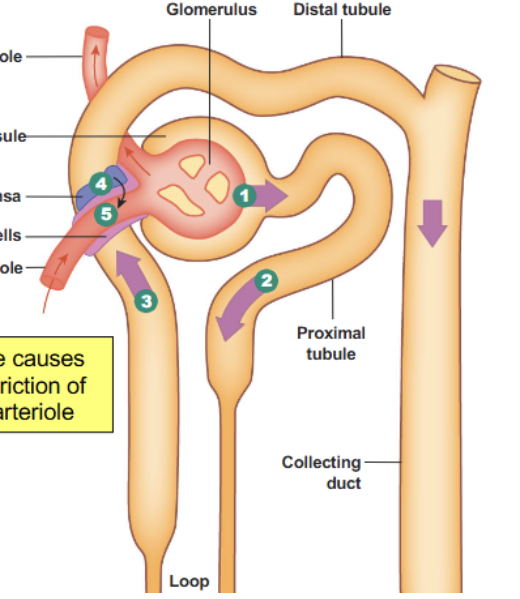
Renal corpuscule
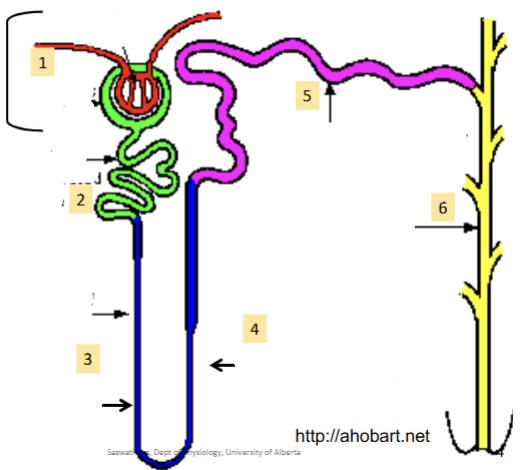
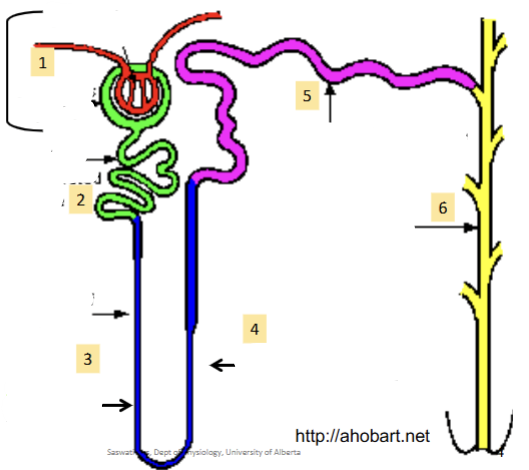
(1). First part of the nephron; contains the glomerulus (bundle of arteries) and bowmen’s capsule (lego hand which cups the capillaries). Always found in the cortex (never in the medulla)
Proximal convoluted tubule
(2). Reabsorbs most of the water and non waste plasma solutes. Major site of solute excretion (H+, NH4-, toxins, drugs)
Loop of Henle (ascending and descending)
(3)/(4). Can dip down into the medulla; how much it dips down determines the nephron type. Hairpin turn. Large amounts of ions reabsorbed (Na, Cl, K, etc)
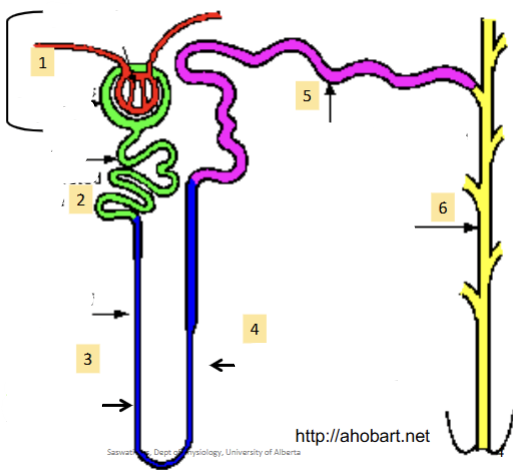
Distal Convoluted tubule
(5). Major homeostatic mechanisms of fine control for water and solute balance to produce urine.
Collecting duct
(6). Many Nephrons drain into a single collecting duct. More fine tuning and homeostatic control occurs here
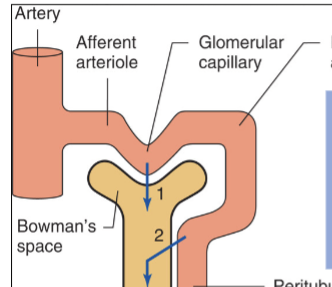
Afferent arteriole
Brings the blood into the Bowman’s capsule
Efferent arteriole
Brings the blood out of the Bowman’s capsule
Podocytes
Inner epithelial cells (AKA visceral layer) of the bowman capsule; specialized with foot-like structures/slits which act as another filtration device. Started off as normal bowman capsule epithelial cells but then became specialized as final structure formed. Slits contain the proteins nephrin and podocins as well as a semiporous membrane.
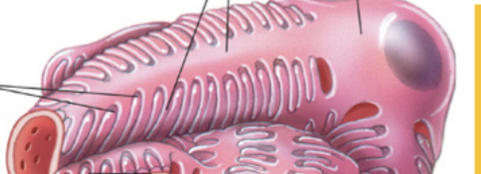
Endothelial cells (fenestrated capillaries) → Basement membrane → Epithelial cells (podocytes)
Order of layers which incoming substrate from the afferent artery must pass through in order to gain access to the inner nephron structures for further filtration. Large molecules (proteins) are kept out! This is because: Pores are too small, Negative charges in BM and Pores repel negatively charged proteins and special semiporous slits in podocytes.
Cortical nephrons
Where the vast majority of the loop of Henle is contained within the cortex, and very little or none of it dips down into the medulla. 85% of nephrons,perform basic functions.
Juxtamedullary nephrons
Where the majority of the Loop of Henle is within the medulla. 15% of nephrons, perform basic functions but primarily involved with regulating urinary concentrations
Cardiac output to the kidneys
Very high (~20% = 1200mL/min).
Glomerular capillaries
Blood vessels which lay within the Bowman’s capsule (inside the corpuscule).

Peritubular capillaries
Blood vessels that surround the proximal convoluted tubule
Vasa recta capillaries
Blood vessel that travels parallel to the loop of Henle.

Glomerular filtration and tubular secretion
Both of these processes allow substances to travel from the blood into the nephron/kidneys (into the lumen) for filtration.
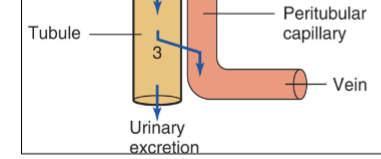
Tubular reabsorption and urinary excretion
Both of these processes allow substances to exit the lumen of the kidneys, either by being reabsorbed into the blood or through external elimination.
Molecules which are able to pass through into kidneys
Water, electrolytes (Na, Cl etc), Glucose, amino acids, fatty acids, vitamins, urea, uric acid and creatine
Molecules which are not able to pass into kidneys
Proteins (ex. albumin), blood cells, plasma proteins, large anions.
Proteinurea
Condition where proteins are found in the blood, means that the filtering techniques are not working and there is a problem.
Net glomerular filtration
Always positive due to PGC (Glomerular capillary pressure = pushing blood out of the capillary into bowman’s capsule) being so much greater than the opposing forces (PBS = pressure in bowman’s space and πGC = osmotic force due to plasma in the protein)
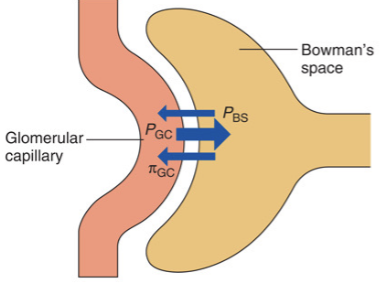
πBS?
Does not exist (assumed to be 0) because the concentration of proteins that are let through into Bowman’s capsule are so low, therefore they do not generate any osmotic pressure
Filtration factor
From the total blood entering the afferent capillary, initially 20% is filtered into the nephron. As it passes through the distal tubules, 19% is reabsorbed, leaving only 1% of that initial blood being excreted in the urine. >99% of plasma is reabsorbed (returned to systemic circulation)
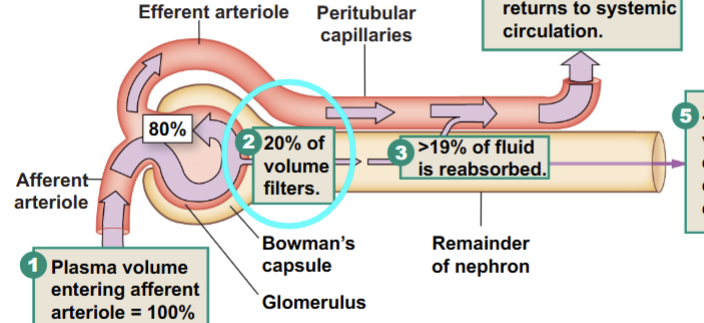
Glomerular filtration rate (GFR)
~125mL/min = 180/day (a lot of fluid - filters the bodies plasma 60x per day). Impacted by net glomerular filtration, permeability of the corpuscular membrane (endothelial fenestrae, basement membrane and podocytes), SA available for filtration (Always impactful). As well as neural/endocrine control.
Neural/endocrine control of GFR
The only factor which can be turned off/on. The rest are all always impacting the GFR in some way.
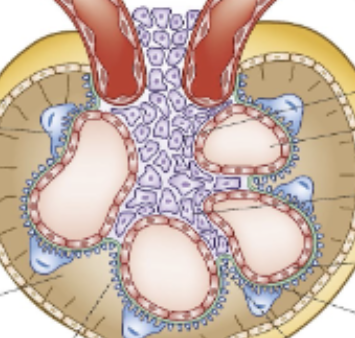
Mesangial cells
Cells found surrounding the glomerulus which are not involved in physically allowing filtrate through, but instead can behave like muscle cells and contract/relax, modifying the available space for solute exchange. Contraction = less space = ↓ GFR (purple cells in diagram)
Constriction of afferent arteriole or dilation of efferent arterioles
Results in a decrease in GFR
Dilation of afferent arteriole or constriction of efferent arteriole
Results in an increase in GFR
Autoregulation of GFR
Manages to stay constant over an incredibly wide range of MAP (want to protect kidney capillaries from too high or low pressures). Occurs via:
Myogenic response (quick = vasodilation/constriction of renal blood vessels)
Tubuloglomerular feedback (increase in flow causes constriction of afferent arteriole via juxtaglomerular apparatus, through paracrine control)
Neuronal and hormonal control
Juxtaglomerular apparatus
Due to the DCT resting against bowman’s capsule, macula densa cells in the DCT can sense increased flow, which causes paracrine release (adenine) to constrict the afferent arteriole. Juxtaglomerular cells in the afferent arteriole can then release renin based on Na+ concentration. Results in a decreased GFR in response to higher blood pressures.
Filtered load
For a specific substance (non protein), how much was filtered into Bowman’s space. Substance (g/L) x GFR (typically 180L/day).
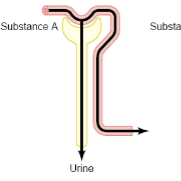
Renal handling of inulin, creatinine
Filtration only (no reabsorption)
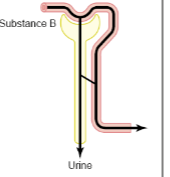
Renal handling of electrolytes
Filtration, partial reabsorption
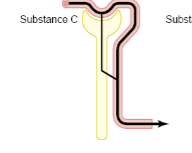
Renal handling of glucose and amino acids
Filtration, complete (100%) reabsorption. This means that at normal function, the clearance of Glc will be 0 (none will be found in the urine)
Renal handling of organic acids and bases
Filtration, secretion
General order of reabsorption for glucose, sodium and urea
Glc (100%) > Sodium (99%) > Urea (44%)
Metabolic secretion
Different from normal secretion; refers to the waste products, produced by cells, being added into the nephron, instead of products from the blood being added to the nephron (normal secretion)
How is reabsorption mediated?
Transepithelial transport = plasma proteins (major)
Diffusion across tight junctions = paracellular (minor)
Always occurring from lumen of tubule (nephron) to the peritubular capillary
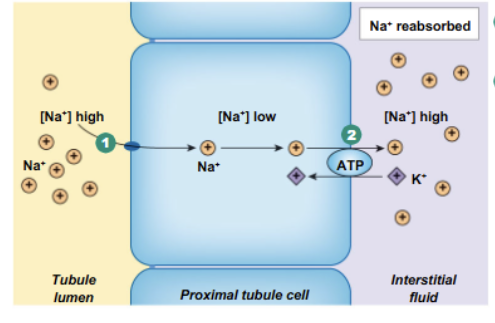
Na reabsorption pathway
Done through transepithelial transport.
Across the luminal side (from inside of nephron tubule into epithelial cell) = varies from nephron segment. Moves down its concentration gradient and contributes to secondary active transport later on, can sometimes diffuse through channels.
Across basolateral side (from tubule cell into interstitial space) = Pumped out via primary active transport (Na/K ATPase)
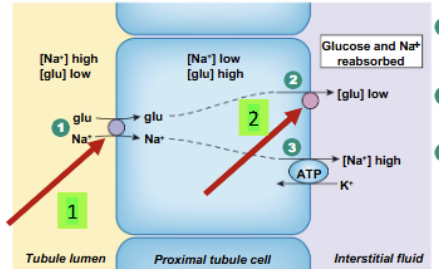
Glc reabsorption pathway
Across luminal side (from inside of nephron tubule into epithelial cell) = SGLT (secondary active transporter, using Na concentration to pull Glc in against its concentration gradient)
Across basolateral side (from tubule cell into interstitial space) = GLUT (facilitated diffusion)
Diabetes mellitus
Potential explanation for glucosuria. Capacity to reabsorb Glc by kidneys is normal but filtered load (amount of Glc) within the blood is abnormally high, exceeding the Glc reabsorption threshold of the tubules (unable to properly reabsorb all the Glc, some escapes and enters the urine).
Renal glucosuria
Genetic mutation of the Na/Glc cotransport (secondary active transport) that would normally allow for the initial reabsorption of Glc to occur.
Reabsorption of urea
Dependent on water being reabsorbed via osmosis. When this occurs, urea is free to diffuse down its concentration gradient, from the tubule lumen, through the epithelium and into the capillary.
Renal clearance
How we quantify the kidneys function in removing a substance from the plasma. Measured in mL/min.
Eq: Clearance of y = [ Uy (urinary concentration) x V (volume of urine in mL/min) ] / Py (plasma concentration)
Filtered Load
GFR x [Plasmay] = [Urinaryy] x volume
*for a given substance y
Inulin clearance
Polysaccharide, not found in the body (100% filtered, no reabsorption). Cin = 180L/day = GFR. Great measure of GFR, but not often used in clinical settings because it has to be injected (since it is not naturally found in the body).
How is GFR related to Pin?
Inversely proportional, therefore when there is lots of Inulin in the plasma, there is very little GFR occuring.
Creatinine clearance
Product of muscle metabolism. Nearly 100% filtered, no reabsorption, slight secretion. Can be used to measure GFR clinically (slight overestimate).
When clearance is greater than GFR
Secretion is occuring
When Clearance is less than GFR
Reabsorption is occuring
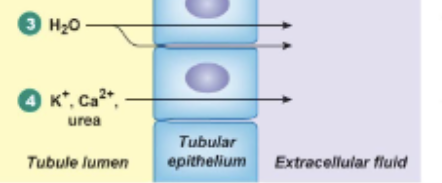
Main area of water reabsorption
Proximal tubules (aquaporins there are always open). Responsible for 67%. Done via passive transport, no hormonal control
Loop of Henle water reabsorption
Responsible for 15%. Done via passive transport through aquaporins, no hormonal control.
Distal tubule water reabsorption
None occurs
Large distal tubule and collecting duct water reabsorption
Variable (8-17%) due to fine tuning occurring here. Controlled by hormone = vasopressin (ADH); passively done via aquaporins
Descending loop of Henle water transport
Permeable to water, therefore water reabsorption occurs, causes the concentration of the filtrate leftover to be hyperosmolar = concentrated. Starts at 300 (normal), increases to 1400.
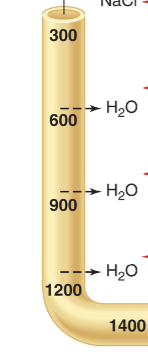
Ascending loop of Henle
Impermeable to water, allows salt reabsorption to occur, generating a hyperosmolar interstisial fluid and restoring tubule concentration = diluted (overshoots, causes it to be ~100 upon exiting, a bit hypo-osmolar)

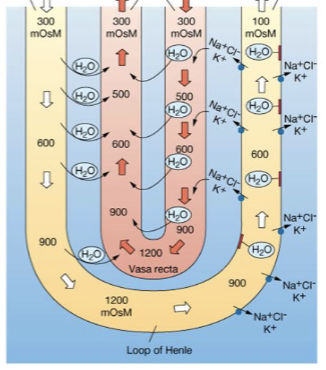
Counter-current multiplier
Name of the mechanism which accounts for the loop of Henle’s role in water reabsorption. Filtrate flowing in opposite directions (counter, asending/descending) and as you travel down, the hyperosmolarity of the filtrate is multipled, and then reversed as you travel up
Difference in osmolarity between ascending and descending loop of Henle
Ascending limb is always 200 mOsml/L less than Descending limb
What happens as you travel deeper into the medulla
The environment gets saltier and saltier (more concentrated, hyperosmolar). This goes for both the loop of henle and the interstisial fluid.
Vasa recta importance for water reabsorption
Permeable to both water and salt. Travels along with the loop, forming its own hairpin turn, with blood flowing in the opposite direction as the filtrate. Responsible for maintaining the gradient generated by the loop of henle, mirrors the osmolarity found at the loops levels
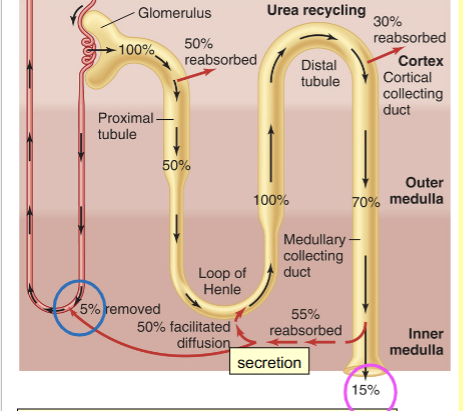
Urea pathway through kidneys
100% filtrated, only 15% excreted, 5% removed via the vasa recta. Reabsorbed in multiple places (proximal tubule, cortical collecting duct and inner medullary collecting duct (ADH important for this step)). Also secreted back into the loop of Henle. Constant recycling helps maintain high osmolarity
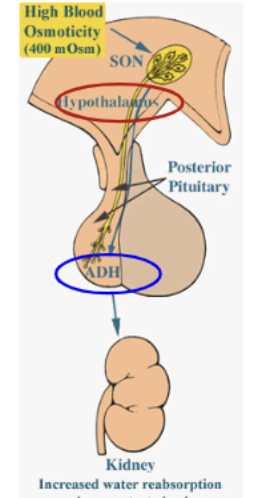
ADH formation and release
Peptide hormone (fast acting). Osmoreceptors in hypothalamus (supraocular nucleus) sense increase in plasma osmolarity and begin production. Then secreted by posterior pituitary, after which it travels down to collecting duct cells.
AQP 2
The specific water channel in the collecting duct which is impacted by ADH levels. Acts on the luminal side (nephron membrane) and is responsible for majority of initial water reabsorption (urine volume ↓)
AQP 3 and 4
Water channels in the collecting duct (on the basolateral side = capillary membrane) that are NOT impacted by ADH
ADH AQP2 mechanism
[IN COLLECTING DUCTS]: ADH binds to receptors → ATP → cAMP (second messenger) → PKA → Protein phosphorylated → induces vesicle and AQP 2 formation and guides it into proper fusion (insertion) with the luminal side → AQP 2 in proper place, water reabsorption increased
Diabetes insipidus
Water diuresis = excess water secreted (increased water loss) in urine due to failure in link between ADH and kidneys. 2 types:
Central = Failure pf ADH release from posteiror pituitary
Nephrogenic = Failure of ADH to act on receptors in nephron collecting duct
Conditions which increase ADH
Heat, dry conditions, water loss/decreased water intake, shock and pain (pee less)
Conditions which decrease ADH
Cold, alcohol, humidity (pee more)
Diabetes Mellitus
Osmotic diuresis = excess solute (sugar) in the urine, which as a byproduct draws out more water due to osmolarity. Both urine volume and solute volume in the urine have been increased
Low Plasma Na (short term)
Handled by baroreceptors (which can sense both low and high Na). Decrease in Na = decrease in blood volume → decrease in stretch → decrease in nerve impusle frequency → activation of sympathetic → constriction of afferent arteriole → (↓GFR, ↓Na excretion, therefore Na reabsorption ↑). Overall = more sodium kept.
High Plasma Na
ANP released, regulates GFR and inhibits Na reabsorption. Also inhibits aldosterone. Overall = more sodium excreted.
Low Plasma Na (Long term)
Aldosterone (steroid hormone) released via RAA pathway. Aldosterone causes increased Na reabsorption, synthesizes new Na transporters and decreases Na excretion (In the cortical collecting duct)
RAA pathway
When NaCl levels drop in the plasam, renin is released. Renin goes on to increase angiotensin I, which is turned into angiotensin II, which in goes on to stimulate the release of aldosterone
Regulation of renin secretion
Done via chemoreceptors in macula densa = distal convoluted (sensitive to NaCl) and mechanoreceptors from juxtaglomerular cells = wall of afferent arteriole (sensitive to plasma volume). Low plasma volume and low NaCl = Low Na, therefore secretion of _____ is increased.
ANP release and impacts
Synthesized by cardiac atria, recognzies high Na (increased blood volume = atrial distension). Inhibits aldosterone, inhibits Na reabsorption, increases GFR (and therefore Na excretion)
High extracellular K+
AKA hyperkalemia. Causes Aldosterone to be produced, increases K+ secretion = increased excretion in the urine (reverse of high Na). Regulated via cortical collecting duct
Low extracellular K+
No Aldosterone produced (opposite of low Na). K+ is mostly reabsorbed in proximale tubule and loop of Henle. Less K+ excreted in urine.
Volatile acid
Only CO2 (Everything else is non volatile)
Extracellular buffer system
Most common in the body. CO2/HCO3-
Intracellular buffer
Proteins or phosphate ions ex) Hemoglobin
Short term Hydrogen ion balancers
Lungs (hyperventilation, hypoventilation etc). Ventilation is adjusted as necessary to maintain homeostasis. ↑ H+ = ↑ ventilation and vice versa
Long term Hydrogen ion balancers
Kidneys. Alkalosis = kidneys excrete bicarbonate to be removed via the urine, bringing pH back down. Acidosis = kidneys synthesize new bicarbonate and release it into plasma to bring pH back up
HCO3- reabsorption
Produced via tubular epithelial cells, depends on the active transport of an H+ ion into the nephron (secretion), where it then combos up with previously filtered bicarbonate to produce H2CO3 (which breaks down into CO2 and H2O and is excreted via urine).
H+ active transporters in kidneys
Could be H+ ATPase, H+/K+ATPase, or H+/Na+ATPase. At different sections of the nephron different transporters will be present on the luminal side.