Chemistry - chemical analysis
1/63
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
64 Terms
What is a pure substance?
a substance in which there is only one type of particle
What is a formulation?
a mixture that has been carefully designed and manufactured to have a specific set of properties
Examples of formulations
fuels, cleaning agents, paints, medicines, alloys, fertilisers and foods
How are formulations made?
by precisely mixing specific ingredients (each with a dedicated purpose) in exact, consistent proportions to achieve a desired function
What is a mixture?
A combination of two or more substances that are not chemically bonded
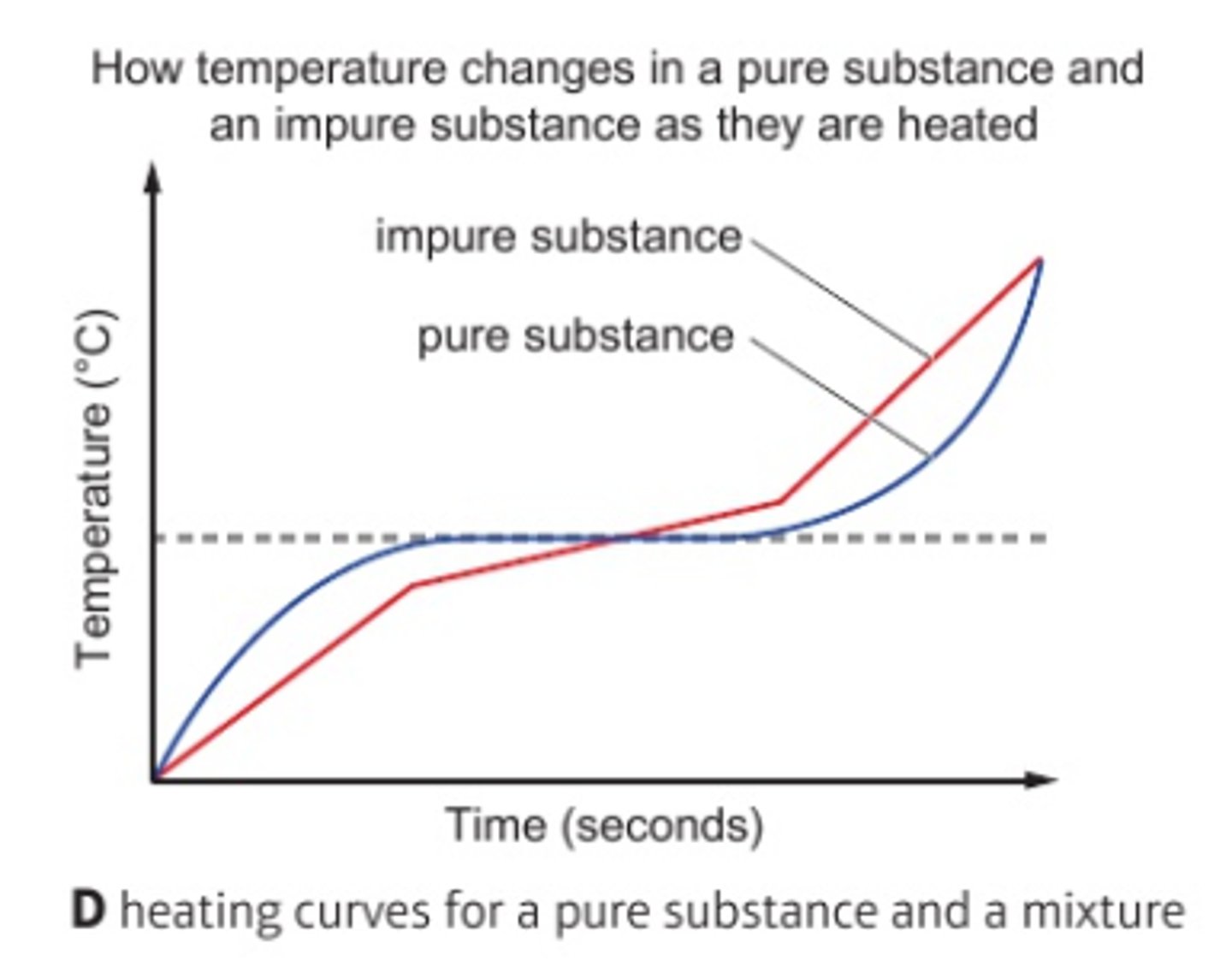
What is the boiling point of pure substances like?
Pure substances have sharp, fixed temperatures
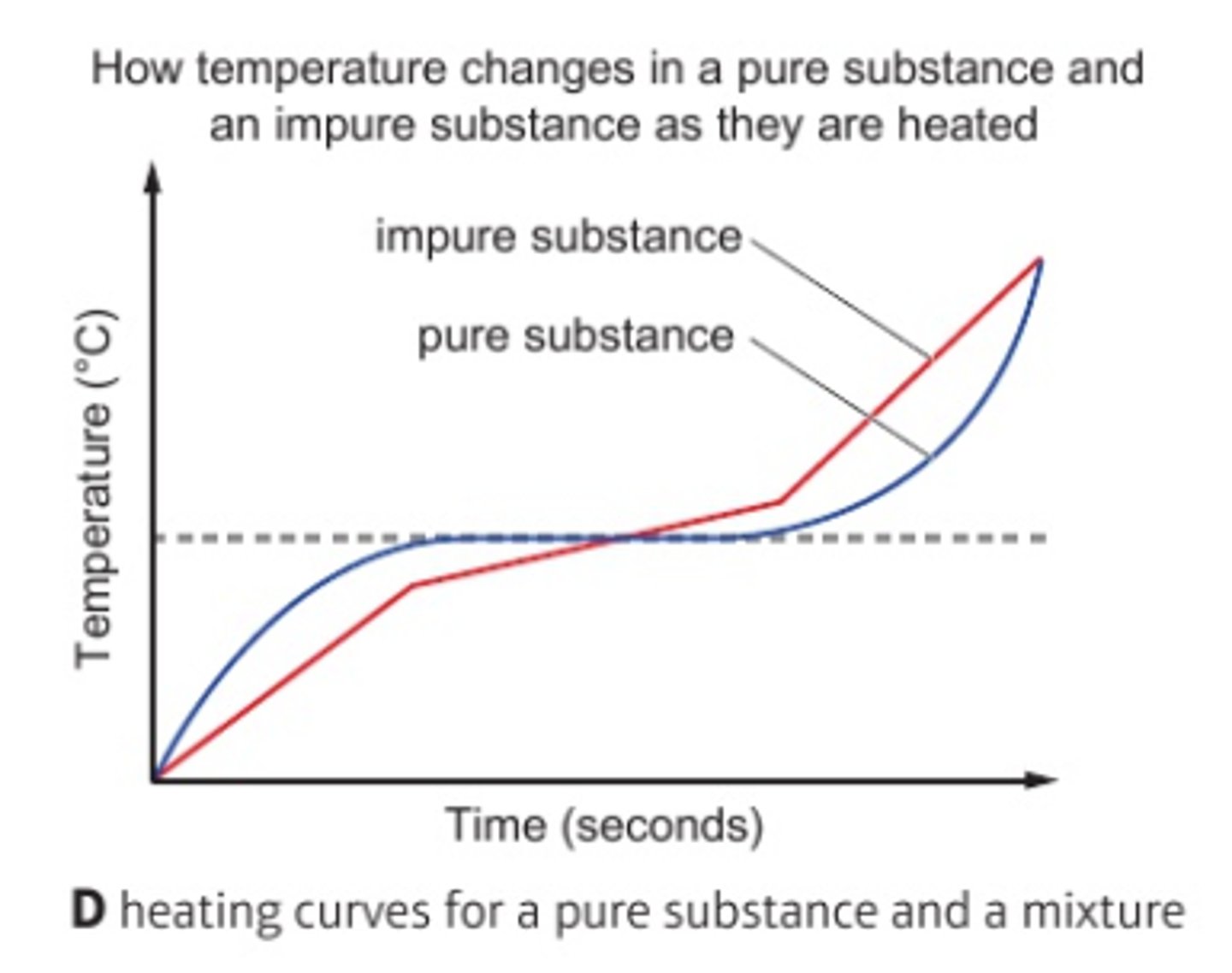
What is the boiling point of impure substances like?
Impure substances melt/boil over a range and usually at a lower temperature than expected
What is a homogeneous mixture?
a mixture in which the composition is uniform throughout
What is a heterogeneous mixture?
a mixture in which the composition is not uniform throughout
How do impurities affect the boiling point of a substance?
It increases the boiling point of a liquid (boiling point elevation) and causes it to boil over a range of temperatures rather than a sharp point.
How are paints formulated?
they have:
- pigments to provide colour
- a binder, to help paint attach itself to an object
- a solvent to help the pigment and binder spread well whilst painting
How are washing-up liquids formulated?
they have:
- a surfactant, which is the actual detergent that removes grease
- water to thin out the mixture
- colouring and fragrance to improve the appeal of the product to customers
- rinse agent to help water drain off
What is chromatography?
Chromatography is a method used to separate mixtures into their different substances.
How does paper chromatography work?
different substances travel different distances based oon their solubility
What is the solvent front?
the furthest point the solvent (mobile phase) reaches on a chromatographic plate or paper (stationary phase)
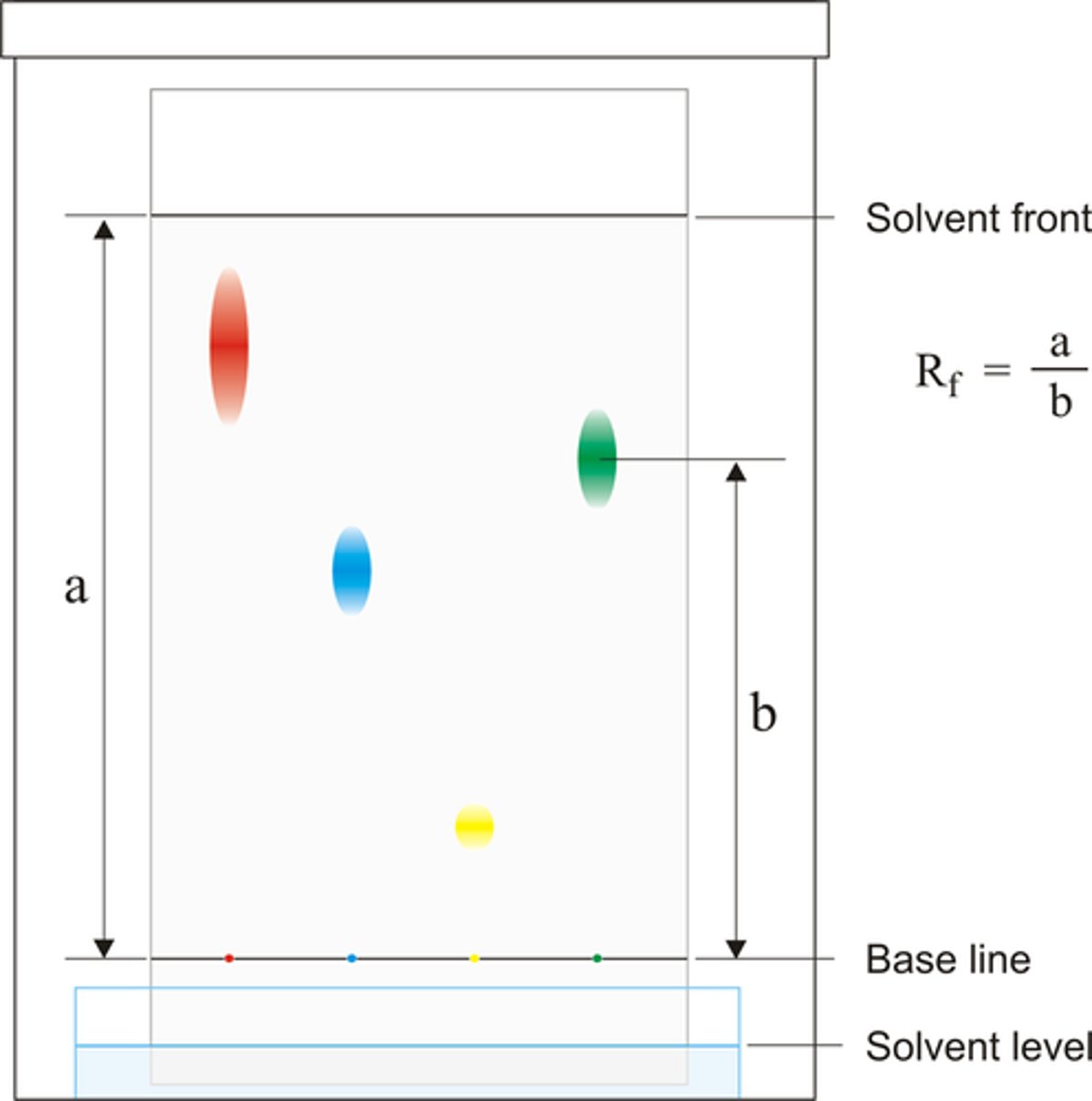
What is the stationary phase in chromatography?
the material that does not move, typically a sheet of absorbent filter paper
What is the mobile phase in chromatography?
The solvent that transports the sample mixture through a stationary phase
What are the steps of chromatography?
1. Draw a pencil line near the bottom of the chromatography paper
2. Put a small spot of the mixture on the line
3. Place the paper in a solvent (e.g. water), making sure the spot is above the solvent
4. The solvent travels up the paper
5. Different substances move at different speeds → they separate
How do more soluble substances travel up the chromatography paper?
they travel further up the paper
How do less soluble substances travel up the chromatography paper?
they travel less
How do insoluble substances travel up the chromatography paper?
they stay on the baseline
Why do more soluble substances travel further in chromatography?
they have a higher affinity for the mobile phase (solvent) and a lower affinity for the stationary phase (paper)
Why do less soluble substances travel less in chromatography?
they have a higher affinity (stronger attraction) to the stationary phase (the paper) than to the mobile phase (the solvent)
In paper chromatography, which molecules move the furthest?
The molecules that are the most soluble
What is the RF value?
The ratio between the distance travelled by the dissolved substance (the solute) & the distance travelled by the solvent
Formula for the RF value
RF value = distance moved by substance/ distance moved by solvent
What numbers are the RF values between?
0 and 1
How can the RF values be used?
They can be used to identify substances by comparing with known values
What is the test for hydrogen?
- Place a lit splint at the opening of a test tube containing the gas
- You will hear a "squeaky pop" if hydrogen is present
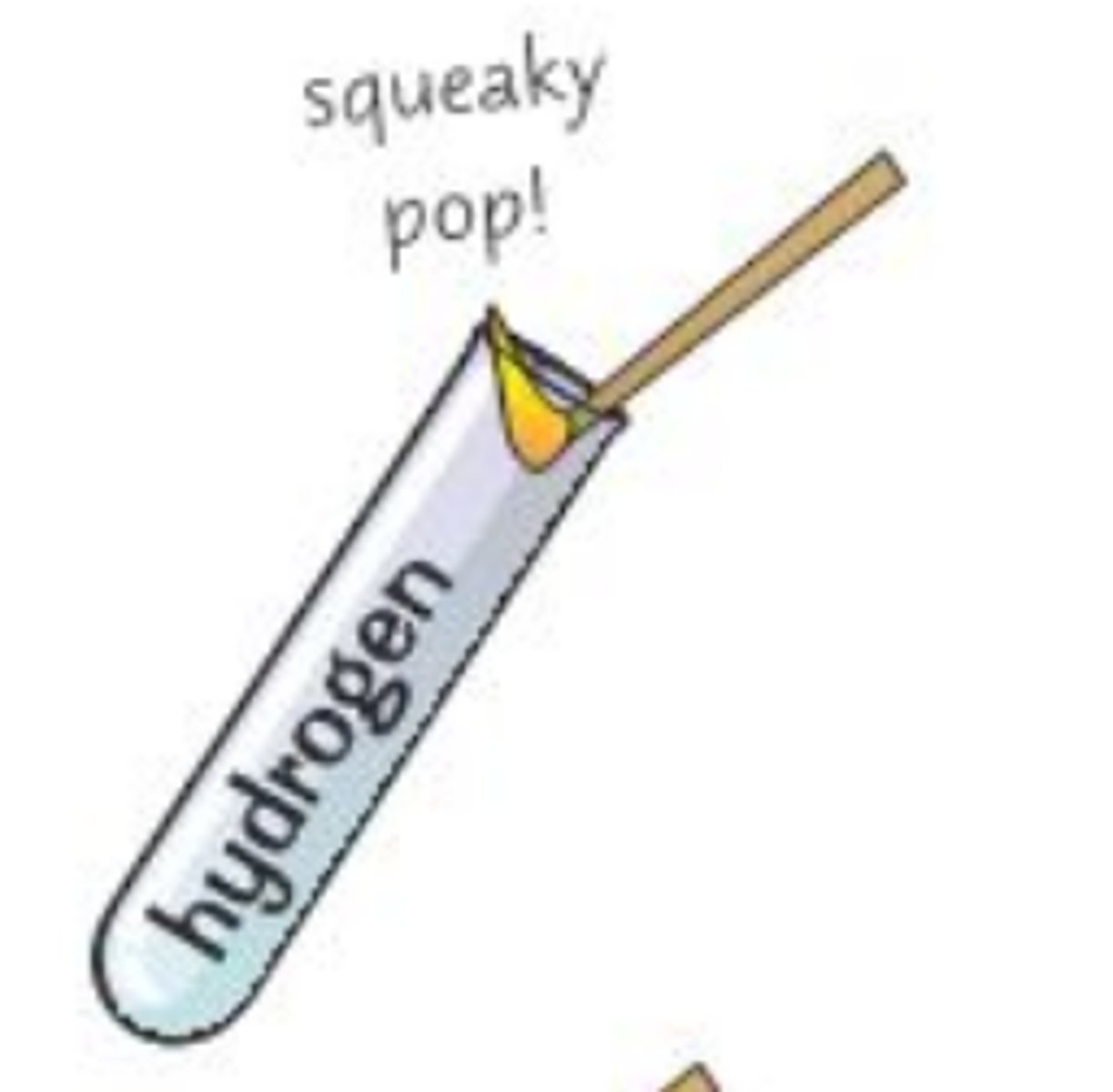
Why do you hear a squeaky pop when hydrogen is present?
because hydrogen burns quickly in air.
What is the test for oxygen?
- Place a glowing splint at the opening of a test tube containing the gas
- if the splint relights, oxygen is present
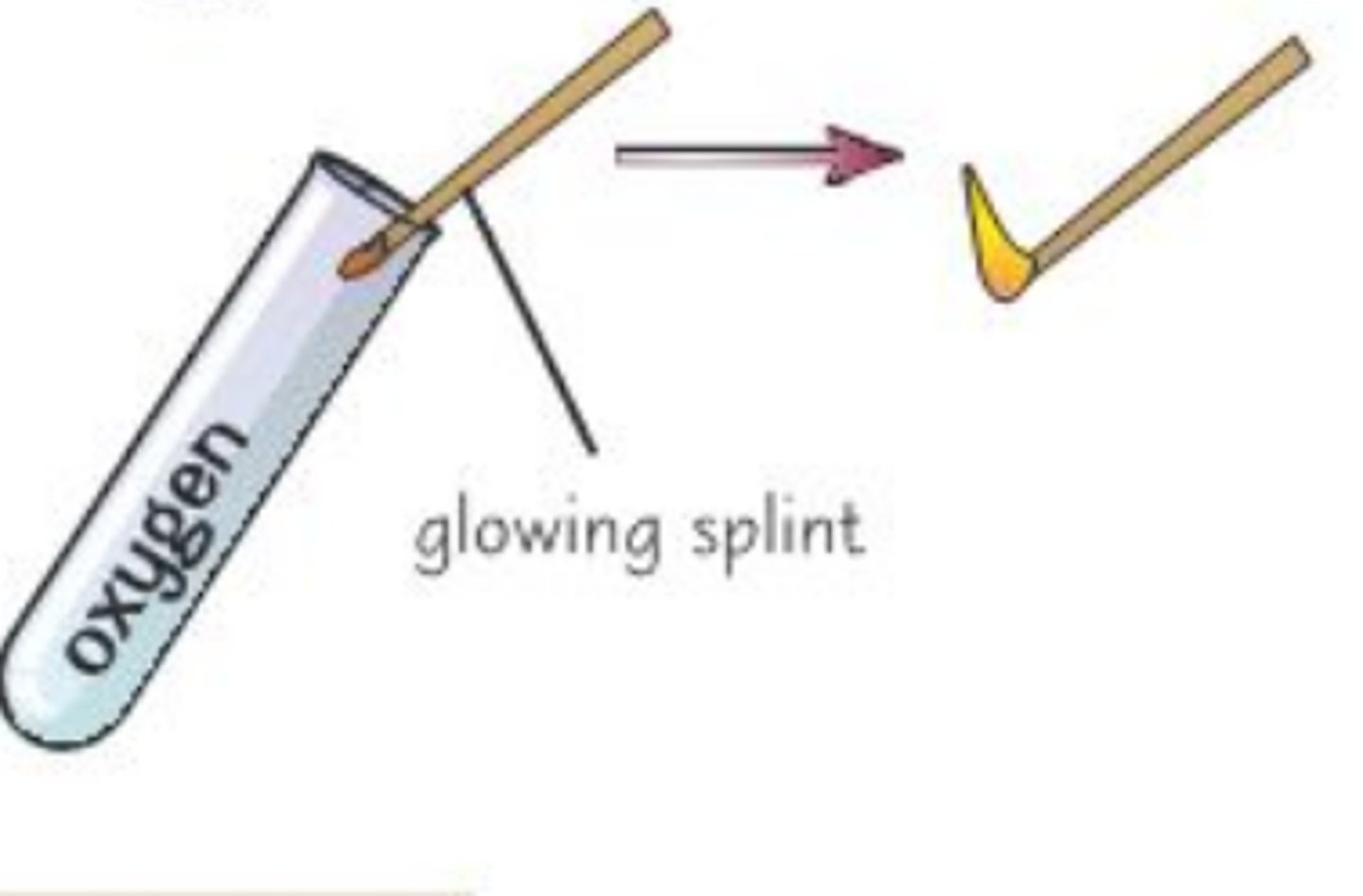
Why does the splint relight when oxygen is present?
Oxygen supports combustion, so it helps things burn
What is the test for carbon dioxide?
Bubble the gas through limewater, if it turns cloudy, carbon dioxide is present.
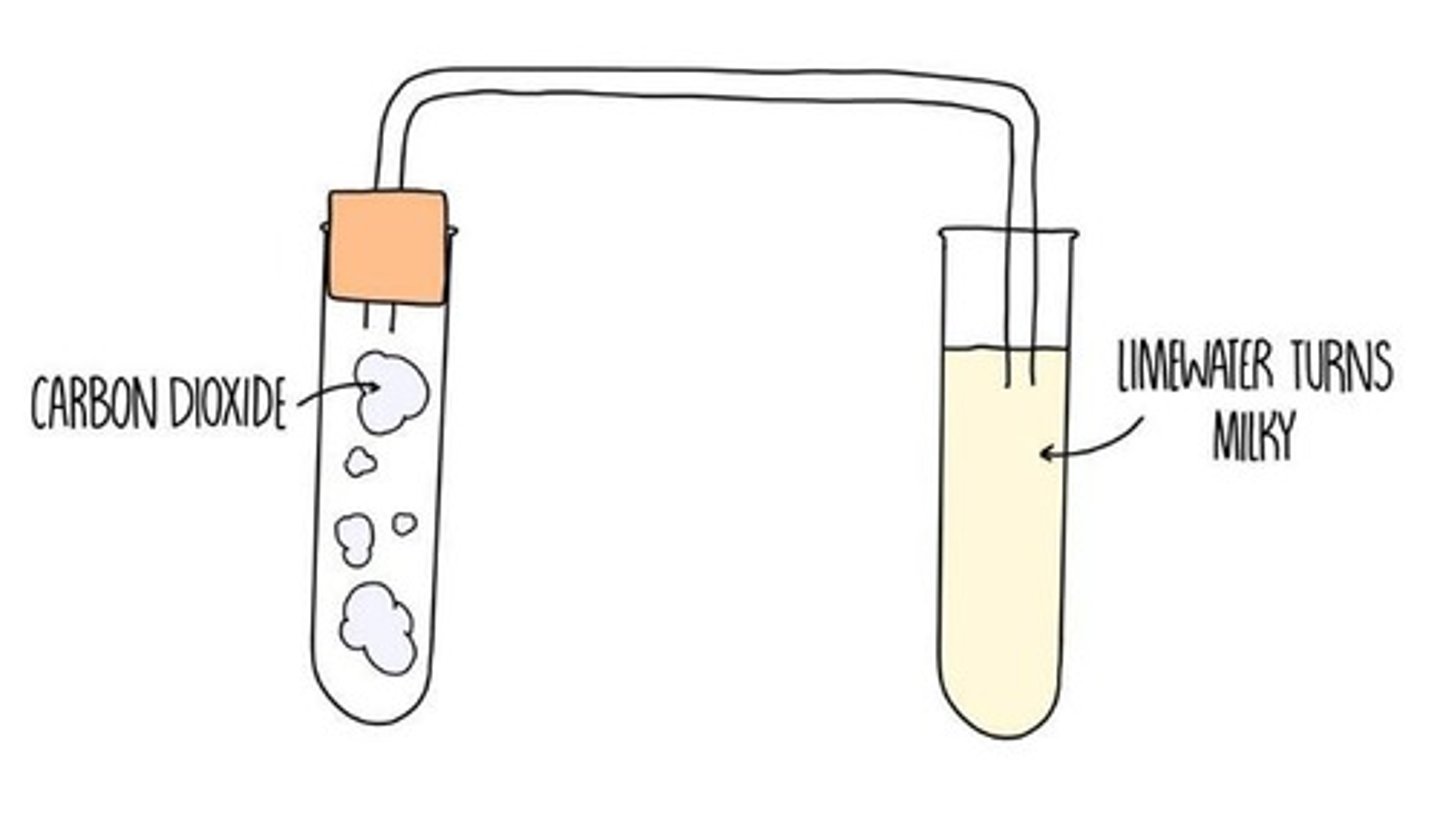
Why does limewater turn cloudy when carbon dioxide is present?
beacuse solid calcium carbonate is made
What is the test for chlorine?
- place a damp piece of litmus paper near the test tube
- if it bleaches white, chlorine is present
How do the flame tests work?
- because the heat excites electrons within the metal atoms to higher energy levels.
- As these electrons fall back to their original, lower energy state, they release energy in the form of light.
- Because each metal has a unique electron structure, the energy gaps differ, releasing unique wavelengths of light that our eyes perceive as distinct colours
What are the steps of the flame tests
1. A Nichrome wire should be dipped in concentrated hydrochloric acid and heated in a Bunsen burner to clean it
2. The wire should then be dipped back into the hydrochloric acid before dipping it into the metal compound being tested
3. The sample is placed in a Bunsen burner with a blue flame
4. The flame changes colour depending on the metal ion present
What colour flame does lithium produce?
Crimson red
What colour flame does sodium produce?
yellow
What colour flame does potassium produce?
lilac
What colour flame does calcium produce?
Brick red
What colour flame does copper produce?
blue-green
What happens if sodium is mixed with another metal in the flame tests?
Sodium's yellow flame is very strong and can mask other colours
What is the test for positive ions?
the flame tests and the sodium hydroxide test
What does the sodium hydroxide test for?
metals in an aqueous solution
What are the steps for the sodium hydroxide tests?
1. Add a few drops of sodium hydroxide solution to your sample
2. Observe if a precipitate (solid) forms
3. Note the colour
4. Add excess NaOH to see if it dissolves
How does the sodium hydroxide test work?
by producing insoluble metal hydroxides that appear as colored or white precipitates
What colour precipitate does calcium form?
White precipitate
What colour precipitate does copper form?
Blue precipitate
What colour precipitate does Iron II form?
Green precipitate
What colour precipitate does Iron III form?
Brown precipitate
What colour precipitate does aluminium form?
A white precipitate that dissolves when excess sodium hydroxide is added
How can you test for carbonate ions?
1. Add a few drops of HCl to the sample in a test tube
2. Connect this test tube to a test tube of limewater using a deviery tube
3. If carbonate ions are present, carbon dioxide will be produced. Limewater will turn cloudy when CO2 is bubbled through.
How can you test for halide ions?
- Add dilute nitric acid and then silver nitrate solution
- check the colour of the precipitate
What colour precipitate does chloride form in the halide ions test?
White precipitate
What colour precipitate does bromide form in the halide ions test?
cream/off-white colour
What colour precipitate does iodide form in the halide ions test?
yellow precipitate
What is the sulfate ions test?
- Add dilute hydrochloric acid, then barium chloride solution
- if sulfate ions are present, a white precipitate forms (barium sulfate)
What is instrumental analysis?
when scientists use machines (instruments) to identify substances and analyse what they are made of
What is flame emission spectroscopy?
An instrumental method used to analyse metal ions in solutions
How does flame emission spectroscopy work?
metal ions, when vaporized in a flame, absorb thermal energy and become excited, emitting light at characteristic wavelengths as they return to ground state
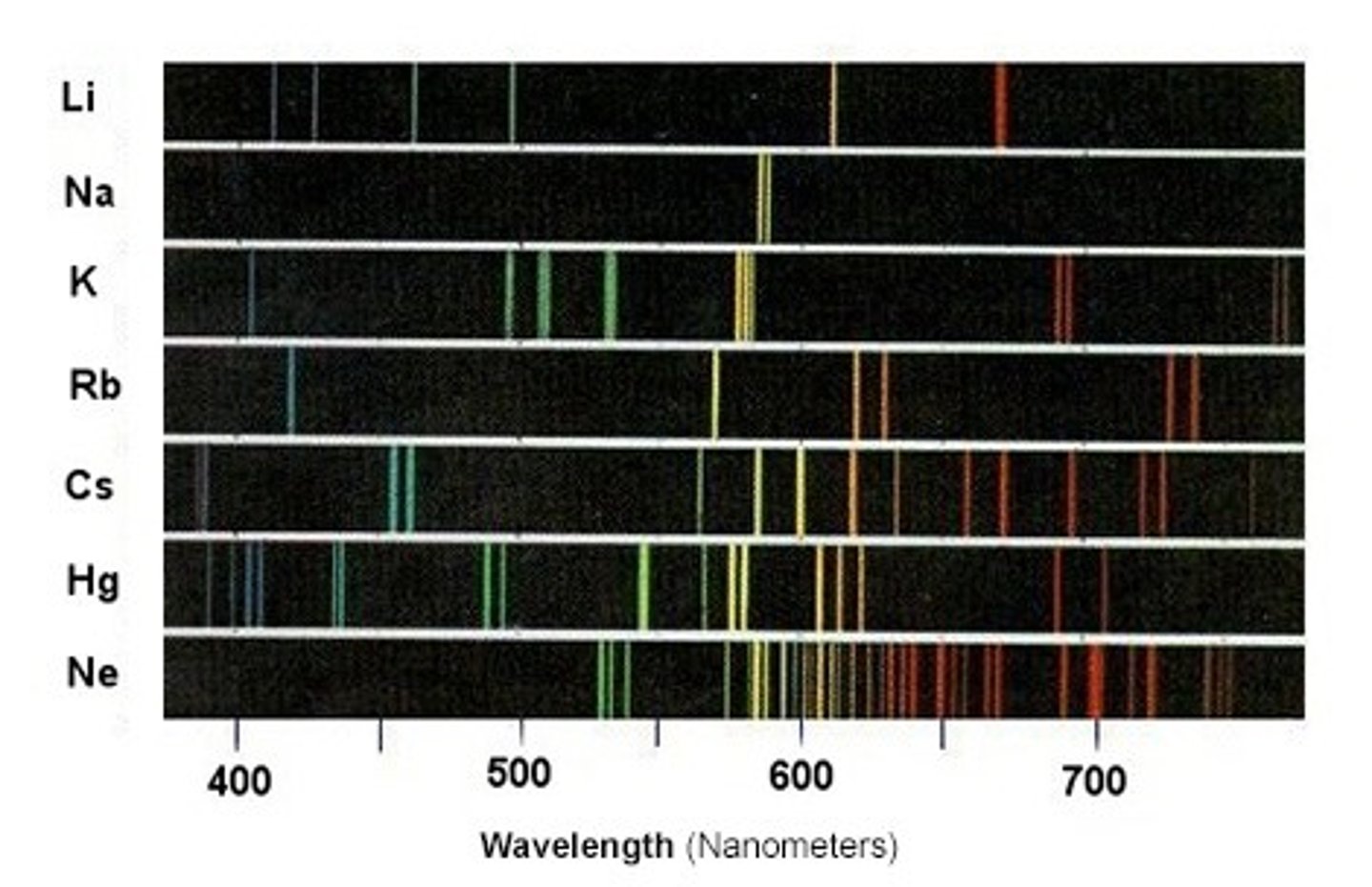
What are the advantages of instrumental analysis?
rapid, accurate, sensitive
What are the advantages of non-instrumental analysis?
low cost, simplicity, and minimal equipment
What are the disadvantages of instrumental analysis?
- the equipment is very expensive
- it requires specialised training to operate
- and results often need to be compared against a database of known substances for identification