General Chemistry 1 - Chapter 5 - Thermochemistry
1/29
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
30 Terms
Energy
The ability to do work or transfer heat.
Thermodynamics
Study of energy and its transformations.
Thermochemistry
Study of chemical reactions and the energy changes that involve heat.
Electrostatic Potential Energy
The most important form of potential energy in molecules (Eel = (kQ1Q2)/d).
Energy is _____ when chemical bonds are formed.
Energy is _____ when chemical bonds are broken.
Released, consumed
Electrostatic Potential Energy (Eel) is between ______ charged ions.
Oppositely
First Law of Thermodynamics
Energy can be converted from one form to another, but it is neither created nor destroyed.
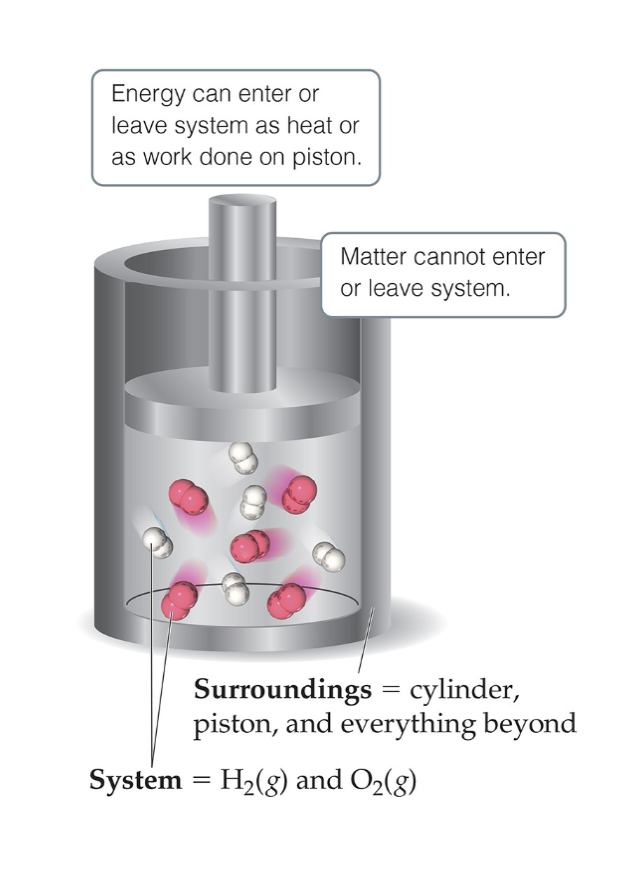
Explain the relationship between the system and the surroundings.
The system is the portion of the universe we single out to study, while the surroundings are everything else.
System + Surroundings = The Universe.
Where the universe can be as big or as small as we make it.
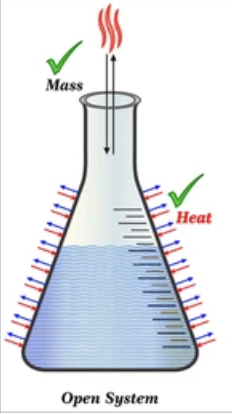
Open System
Exchanges heat and mass with its surroundings.
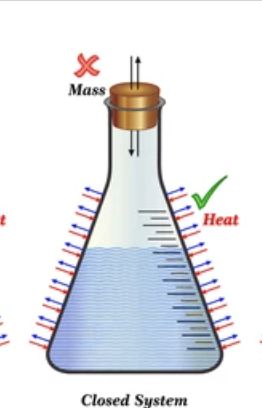
Closed System
Exchanges ONLY heat with its surroundings.
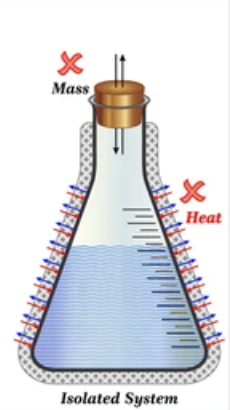
Isolated System
CANNOT exchange heat or mass with its surrounds.
Internal Energy (E)
The sum of all kinetic and potential energy of a system.
When you have /\ (delta) of anything, it is equal to the final minus the initial. So, /\X = Xfinal - Xinitial. This can also be written as /\X = Xproducts - Xreactants.
/\E = Efinal - Einitial
/\H = Hproducts - Hreactants
And so on….
A positive /\E (/\E > 0, Efinal > Einitial) results when the system _____ energy from the surroundings.
A negative /\E (/\E < 0, Efinal < Einitial) results when the system _____ energy to the surroundings.
gains, loses/releases
What are q and w in the equation, /\E = q + w?
q is the term for heat, and w is the term for work.
In the equation /\E = q + w,
1.) If q is positive, the system….
2.) If q is negative, the system…
1.) GAINS heat
2.) LOSES heat
In the equation /\E = q + w,
1.) If w is positive, work is…
2.) If w is negative, work is…
1.) done ON the system
2.) done BY the system
In the equation /\E = q + w,
1.) If /\E is positive, the system….
2.) If /\E is negative, the system…
1.) Has a net GAIN of energy
2.) Has a net LOSS of energy
Because energy is always conserved, the numerical value of q for the system and surroundings is always…
The same, just with different signs (±).
Endothermic Exchange of Heat
When the system absorbs heat.
qsys > 0
/\H > 0
Exothermic Exchange of Heat
When the system loses heat.
qsys < 0
/\H < 0
State functions
A function that depends only on initial and final states (like internal energy, E), rather than the path by which the system arrived at that state.
Path Functions
A function that depends on the path by which the system arrived at that state, (like heat, q, and work, w), rather than just the initial and final states.
Formula for work?
W = -P*/\V,
Where /\V = Vfinal - Vinitial
Usually, the work done by a chemical or physical change is the mechanical work associated with a change in volume of ___.
gas
Define enthalpy.
The internal energy plus the product of pressure and volume.
H = E + P*V
When a system changes at constant pressure, enthalpy can be written as /\H = /\E + P*/\V. By substituting the values of /\E and w, you can simplify this equation to be….
/\H = q
Or the change in enthalpy is the heat gained or lost
REMEMBER THIS IS ONLY VALID AT CONSTANT PRESSURE
Is enthalpy an extensive or intensive property?
Extensive, because it depends on the sample size.
What is the relationship of the enthalpy change between a reaction and its reverse reaction?
The reactions are equal in magnitude, but opposite in sign (±).
True or false: The enthalpy change for a reaction depends on the states of the reactants and products.
True.