CHE 2B- ALL OF IT
1/271
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
272 Terms

Groups, EG, # of Lone Pairs, MG, Angles, “P” or “NP”, hybridization
2, linear, 0, linear, 180, NP only if both groups are the same, sp
intensive vs extensive properties
Intensive properties do not depend on the amount of substance, whereas extensive properties do depend on the quantity. Examples include density (intensive) and mass (extensive).

what is Z
atomic charge of the protons
periodic trend for Zeff
INC, TO THE RIGHT ONLY.
AS SHEILDING DEC, ZEFF MUST INC!!
what is Zeff
while Z is the atomic charge of the protons, which is what the core electrons will experience, the Zeff is what valence electrons experience, because the shielding causes the val e-’s to experience a reduced effective nuclear charge due to the presence of core electrons.
zero-point energy
its the “ground state” for energy, aka the LEAST energy possible, however due to the Heisenberg uncertainty principle, the position/momentum will not = 0
for the equation E=hv, how else can it be changed?
second bullet point doesn’t matter here

Groups, EG, # of Lone Pairs, MG, Angles, “P” or “NP”, hybridization
3, trigonal planar, 0, trigonal planar, 120, NP only if all 3 groups are the same, sp2
Groups, EG, # of Lone Pairs, MG, Angles, “P” or “NP”, hybridization
3, Trigonal planar, 1, bent, 120, P, sp2
Groups, EG, # of Lone Pairs, MG, Angles, “P” or “NP”, hybridization
4, tetrahedral, 0, tetrahedral, 109.5, NP only if all 4 groups are the same, sp3
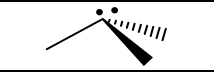
Groups, EG, # of Lone Pairs, MG, Angles, “P” or “NP”, hybridization
4, tetrahedral, 1, trigonal pyramidal, 109.5, P, sp3
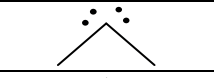
Groups, EG, # of Lone Pairs, MG, Angles, “P” or “NP”, hybridization
4, tetrahedral, 2, Bent, 109.5, P, sp3
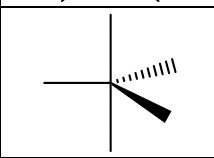
Groups, EG, # of Lone Pairs, MG, Angles, “P” or “NP”, hybridization
5, trigonal bipyramidal, 0, trigonal bipyramidal, 120 and 90, NP only if Linear and Trig. Planar parts are both NP, sp3d
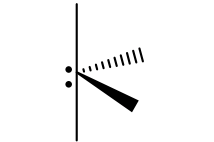
Groups, EG, # of Lone Pairs, MG, Angles, “P” or “NP”, hybridization
5, trigonal bipyramidal, 1, seesaw, 120 and 90, P, sp3d
Groups, EG, # of Lone Pairs, MG, Angles, “P” or “NP”, hybridization
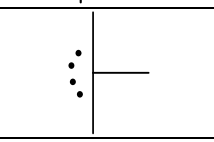
5, trigonal bipyramidal, 2, T-shaped, 90, P, sp3d
Groups, EG, # of Lone Pairs, MG, Angles, “P” or “NP”, hybridization
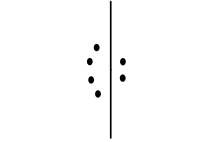
5, trigonal bipyramidal, 3, linear, 180, NP only if both groups are the same, sp3d
Groups, EG, # of Lone Pairs, MG, Angles, “P” or “NP”, hybridization
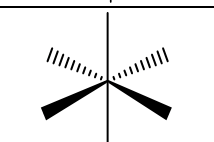
6, octahedral, 0, octahedral, 90, NP only if groups on same axis are the same, sp3d2
Groups, EG, # of Lone Pairs, MG, Angles, “P” or “NP”, hybridization
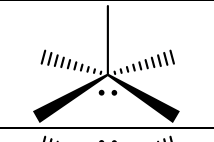
6, octahedral, 1, square pyramidal, 90, P, sp3d2
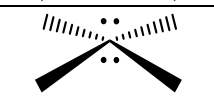
Groups, EG, # of Lone Pairs, MG, Angles, “P” or “NP”, hybridization
6, octahedral, 2, square planar, 90, NP only if groups on the same axis are the same, sp3d2
Hunds Rule
A principle in quantum chemistry stating that electrons will occupy degenerate orbitals singly before pairing up, maximizing total spin.
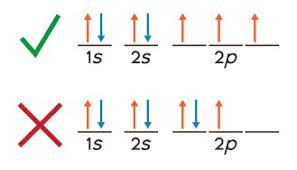
Aufbau principle
The Aufbau principle states that electrons fill lower-energy orbitals before occupying higher-energy ones, following a specific order based on increasing energy levels.

Bohr Theory
A model of the atom proposed by Niels Bohr, which describes electrons orbiting the nucleus in fixed paths with quantized energy levels. (a simple model, not accurate, but easier to work with!)
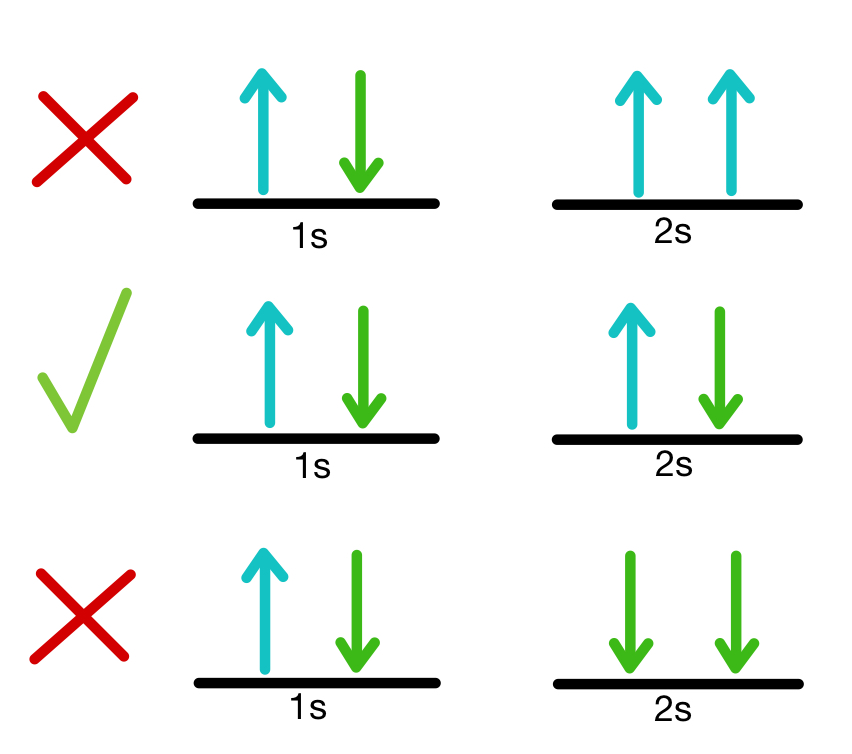
Pauli Exclusion Principle
States that no two electrons in an atom can have the same set of quantum numbers, ensuring that each electron occupies a unique state.
Heisenberg Principle
Also known as the Uncertainty Principle, it states that it is impossible to precisely measure both the position and momentum of a particle simultaneously.
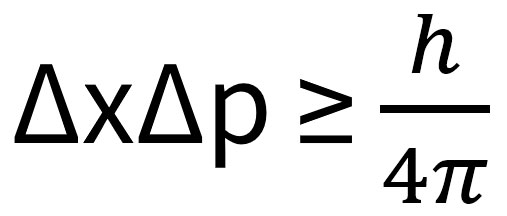
covalent radius
the measure of the size of an atom's covalent bond, defined as half the distance between the nuclei of two bonded atoms.
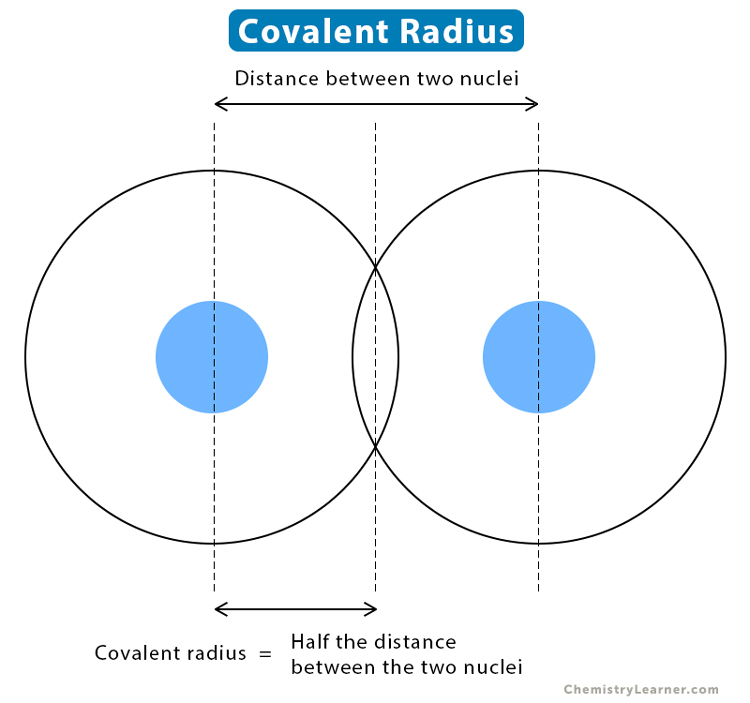
metallic radius
the measure of the size of an atom when it forms metallic bonds, typically defined as the distance from the nucleus to the outermost electron shell in a metallic lattice.
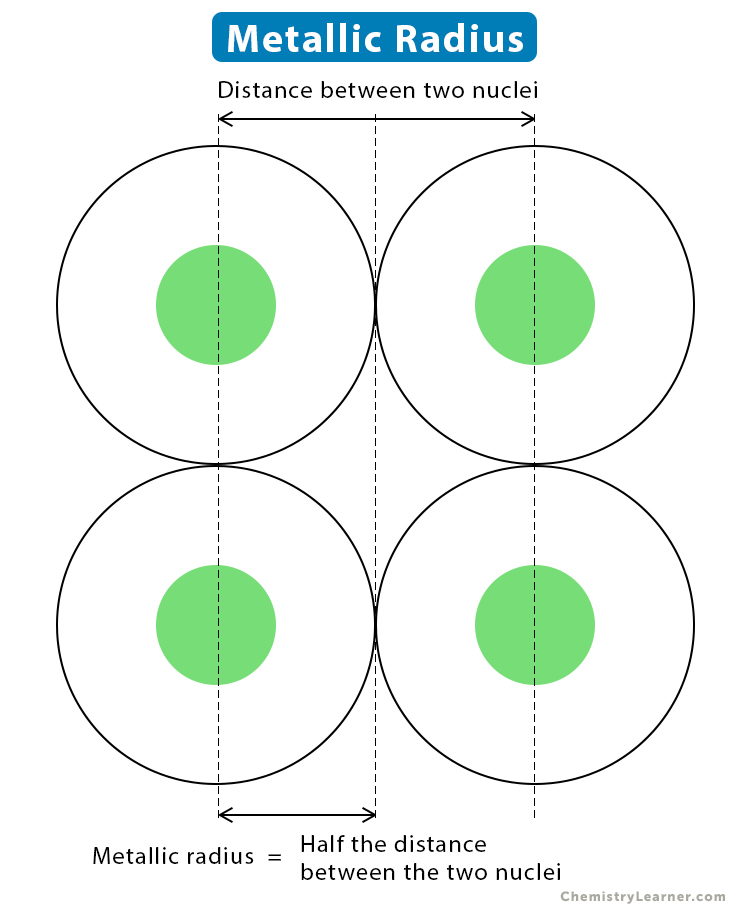
ionic radius
the measure of the size of an ion, typically defined as the distance from the nucleus to the outermost electron shell in an ionic compound.
Are metals…
1.) Wanting to GAIN or LOSE e-’s?
2.) REDUCING or OXIDIZING agents?
3.) Making BASIC or ACIDIC oxides?
1.) Lose e-’s
2.) Reducing agents
3.) Basic oxides
Are nonmetals…
1.) Wanting to GAIN or LOSE e-’s
2.) REDUCING or OXIDIZING agents?
3.) Making BASIC or ACIDIC oxides?
1.) Gain e-’s
2.) Oxidizing agents
3.) Acidic oxides
Quantum numbers- what is n mean?
The principal quantum number (n) indicates the energy level and size of an atomic orbital, representing the shell in which an electron resides.

Quantum numbers- what is l mean?
The angular momentum quantum number (l) determines the shape of an atomic orbital and can take integer values from 0 to n-1. It also lets you know which orbital it is (spdf)
Quantum numbers- what is ml mean?
The magnetic quantum number (ml) specifies the orientation of an atomic orbital in space and can take integer values ranging from -l to +l, including zero. It corresponds to which subshell an electron is in (see picture)
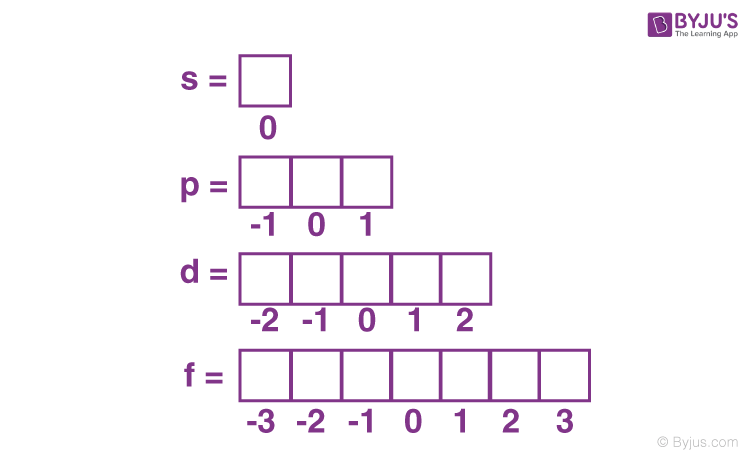
Quantum numbers- what is ms mean?
The spin quantum number (ms) describes the intrinsic spin of an electron within an atomic orbital, which can have a value of +1/2 or -1/2.
What does an s orbital look like?
sphere shaped
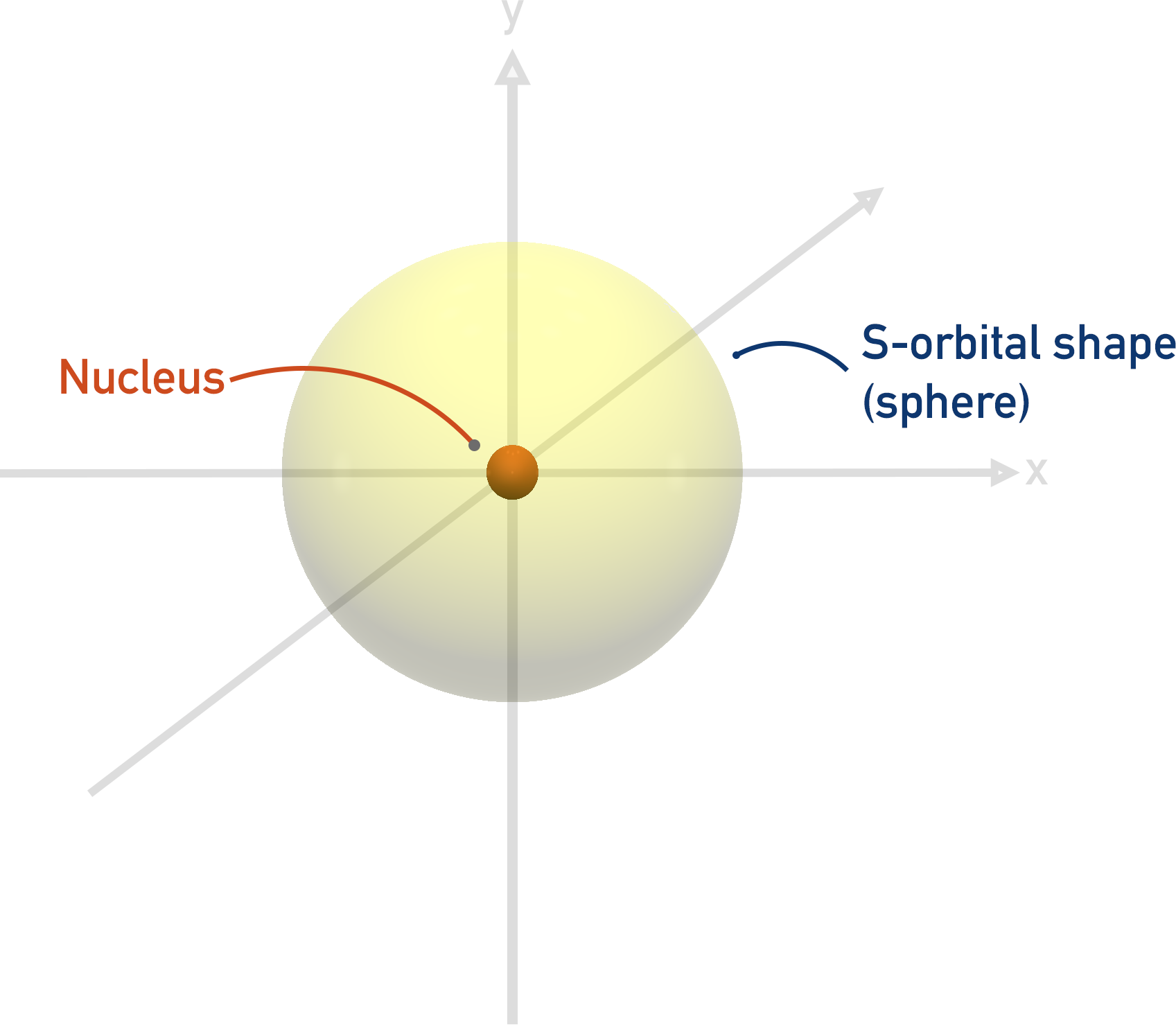
What does a p orbital look like?
dumbbell shaped
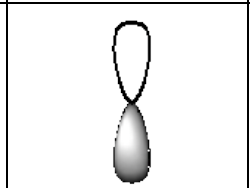
What does a d orbital look like?
cloverleaf shaped
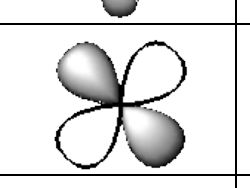
Electron config- what does an expanded diagram look like?
A visual representation showing the arrangement of electrons in an atom, with lines or arrows indicating occupancy and additional layers or subshells. (ONE ORBITAL FOR EVERY ML VALUE)
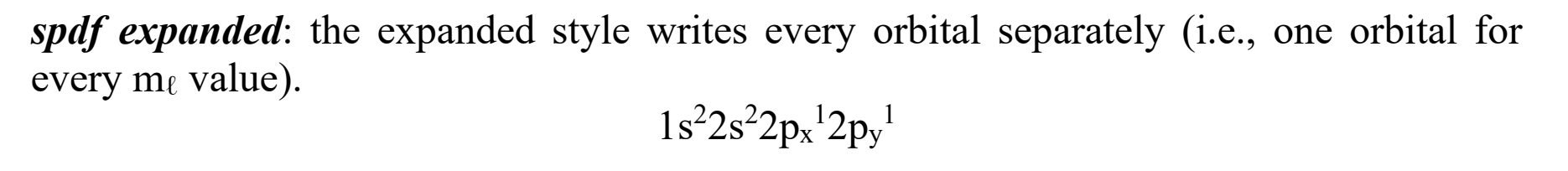
Electron config- what does a condensed diagram look like?
Lists all the spdf orbitals, but less complicated than the expanded diagram (you can also include noble gases to shorten it more too)
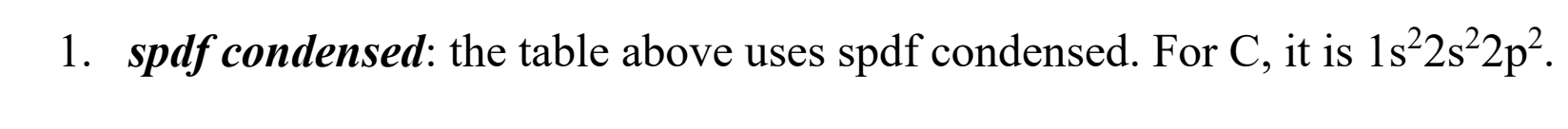
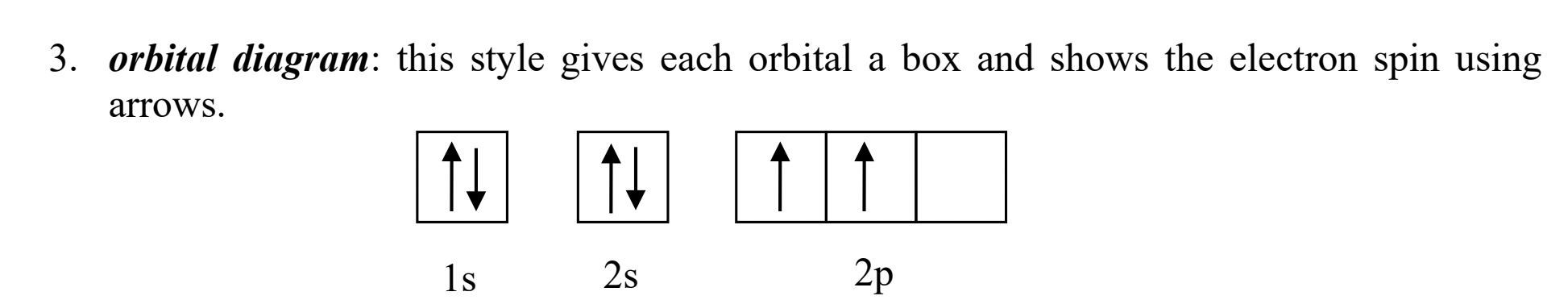
Electron config- what does an orbital diagram look like?
the one with the boxes and arrows
exceptions to the Aufbau process- 4th and 9th columns
This happens in the 4th and 9th column of the trans. metals (ex, Cr and Cu columns). since they are so close to having a half-full/full orbital for their d orbital, they put one of the s-orbital e-’s, (having it half filled, so it’s still stable enough) and puts that electron into the d orbital to achieve greater stability.
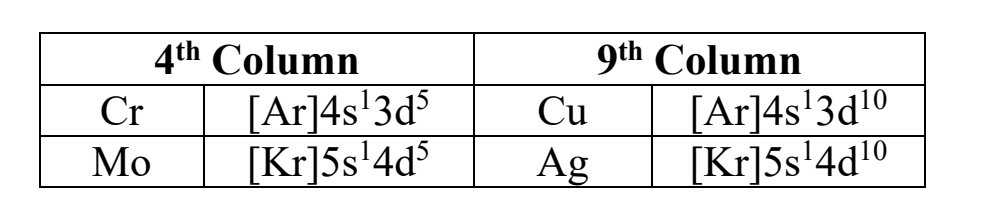
exceptions to the Aufbau process- trans. metal cations
When forming cations, transition metals often lose electrons from their s orbitals before their d orbitals.(This is because the s electrons are higher in energy than the d electrons once the transition metal is ionized, allowing the cation to achieve more stability. )

WHY are half filled and filled shells stable?
half-filled: bc there will be no electron-electron repulsion (each e- gets its own box)
filled- bc it mimics a noble-gas type config
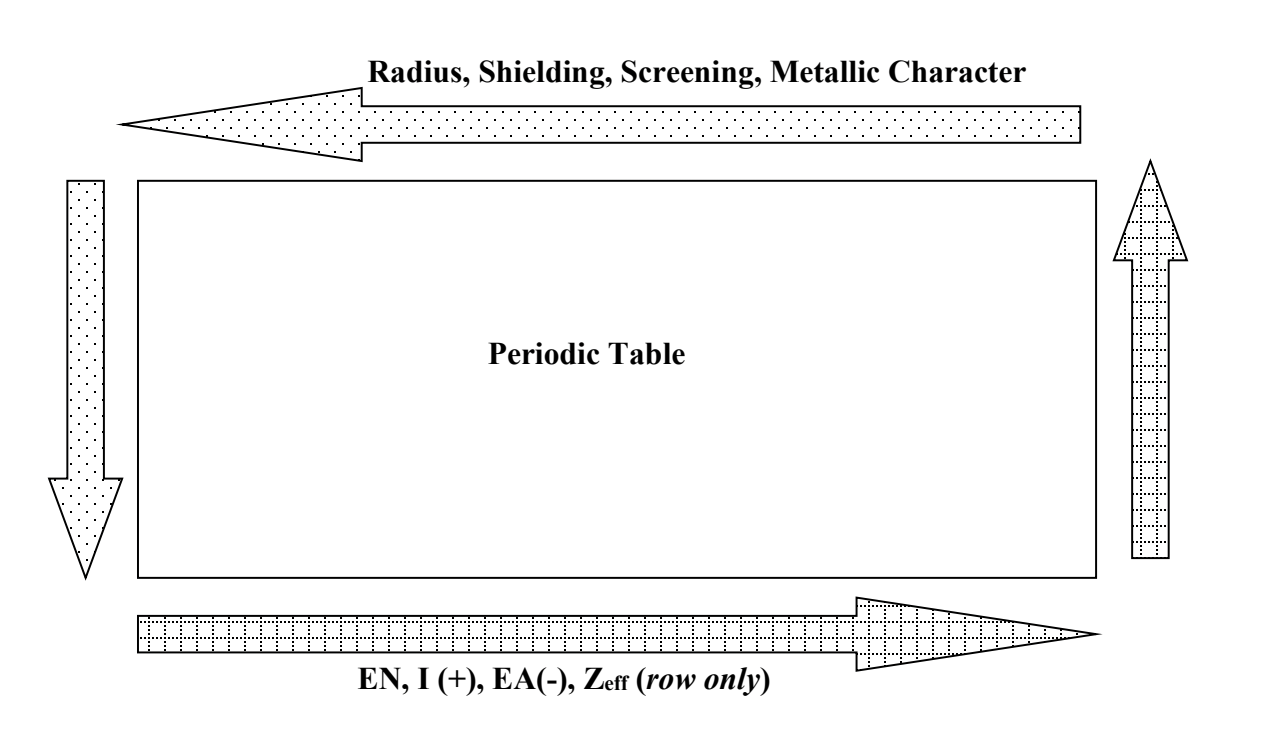
Trends for: Atomic radius, shielding, screening, metallic character, electronegativity, ionization energy, electron affinity, Zeff
Electronegativity, Electron affinity, Ionization energy inc when going toward the top right (NOTE: e- affinity kinda doesn’t have a consistent trend up or down. its more by row)
Radius, shielding/screening, metallic character inc when going toward the bottom left
what is electron affinity (EA (-), and what does it mean if it’s ±?
EA is the energy change associated for when an atom gains an electron
(also, with the periodic trend, when we say the EA ‘increases’ we mean that the value is getting more negative (meaning it’ll release a bigger amount of energy when it gains an e- )
exceptions- columns when electrons would be half-fill (bc they’re stable, they don’t want to add/gain e-) and noble gases. These elements will have positive or barely negative EA values.
+EA= less favorable electron gain, energy will be required to add the e- (endo!)
-EA= more favorable electron gain, energy will be released when an e- is added. (exo!)
NOTE: Anions will take increasingly amounts of energy to add electrons (big positive E), since the anion (NEGATIVE) and e- (NEGATIVE) will want to repeal each other.
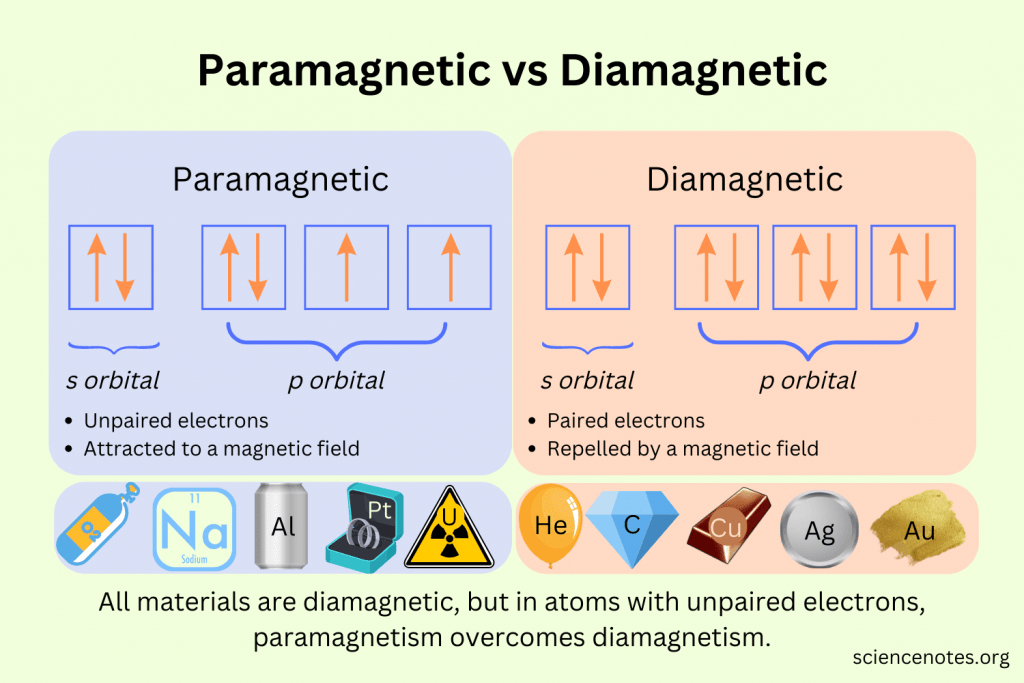
Paramagnetic vs Diamagnetic
Paramagnetic: atom have unpaired electrons and are attracted to magnetic fields. It only takes ONE unpaired e- to make the whole thing paramagnetic.
Diamagnetic: all electrons paired and are unaffected (or BARELY weakly repelled) by magnetic fields.
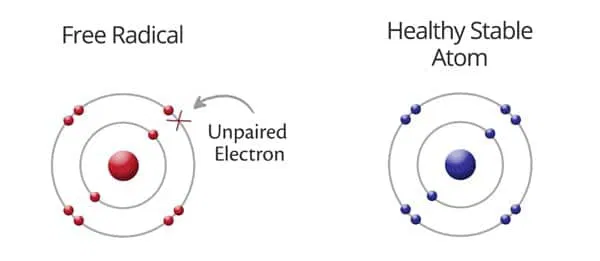
radical electron
a single, unpaired electron
ionic bond
bond between 2 ions, the TRANSFER electrons
Coordinate Covalent bond
a bond formed when one atom donates both electrons in a shared pair to another atom, resulting in a stable molecule.
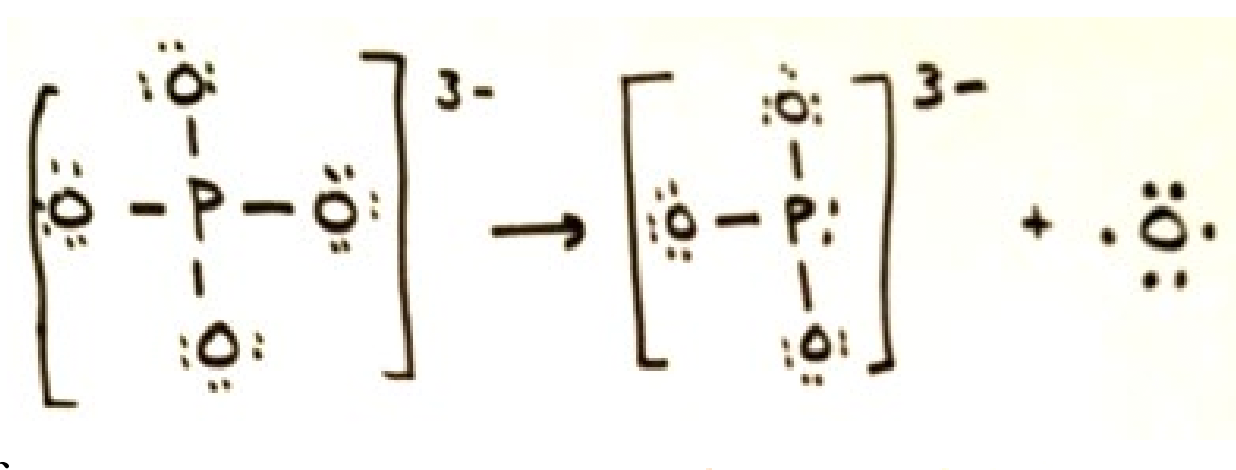
Multiple bonds
Covalent bonds form double or triple bonds sharing more than two electrons.
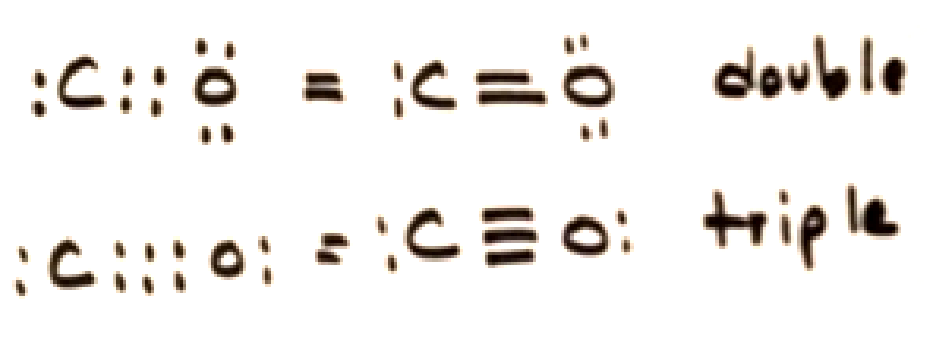
Polar Covalent bonds
bonds where the electrons are not shared equally between the two atoms because of differences in electronegativity.
“Polar means that there is a pole or dipole because electrons are unequally distributed on the molecule. In contrast, molecules can be nonpolar”
Molecular geometry vs Electron geometry
Molecular geometry: the 3D arrangement of atoms in a molecule
electron geometry: the spatial arrangement of all electron groups, including lone pairs.
covalent bond
when 2 atoms SHARE an e-
bond length
distance between nuclei/ the sum of the covalent radii
Bond order
the measure of multiple bonds. It is calculated as the difference between the number of bonding and antibonding electron pairs, divided by two.
Rules for assigning oxidation numbers
1.) Elements in their elemental forms= 0
2.) Group 1 metals= +1, Group 2 metals= +2
3.) Hydrogen= +1, EXECPT when it’s bonded to a metal (then it’s -1)
4.) Trans. elements are determined from anion’s charge
5.) the most electronegative elements get their typical ox. state (like since Carbon wants 4 e-, C’s ox. state= -4)
6.) the last element is forced to have what it has to balance out the compund/ion
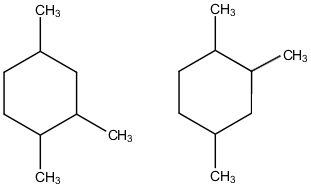
In these organic structures, where will u find C atoms?
all the ends and bends!
What is polarity?
The distribution of electrical charge across a molecule. Also, means that there is a pole or dipole because electrons are unequally distributed on the molecule
what are bond angles
angle of two adjacent covalent bonds that share a central atom
What is a charge, and which type of bonding does it involve
its where an atom’s total # of e- is not equal to the # of protons. Has to do with ionic bonding
What is oxidation state, and which type of bonding does it involve, how does it relate to charge
It’s the charge given to an atom, assuming the bond to other elements are ionic, showing the degree of e-’s lost or gained (or the theoretical charge)! The Ox state charge= overall charge
What is ionization energy/ would would ± values mean?
Ionization energy is the energy present to remove an electron from a molecule.
+ value= energy must be put in to remove the electron (endo!)
- value= energy will be released when an electron is added (exo!).
electronegativity
is the measure of an atom's ability to attract and hold onto electrons in a chemical bond. It plays a crucial role in determining bond character, whether covalent or ionic. (also, it’s just a value, don’t worry about it being ±)
what is formal charge, and how does it relate to overall charge?
the FC is assigned specifically to an atom in a molecule, assuming the electrons are equally shared among atoms. The sum of the FCs of each atom = overall charge too!
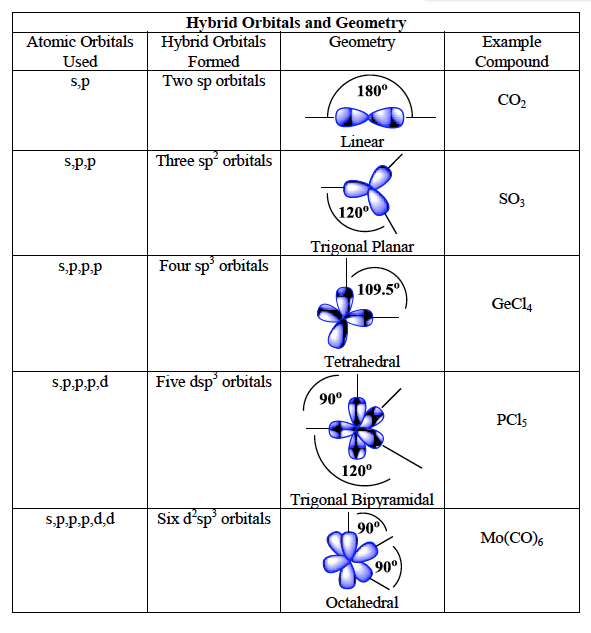
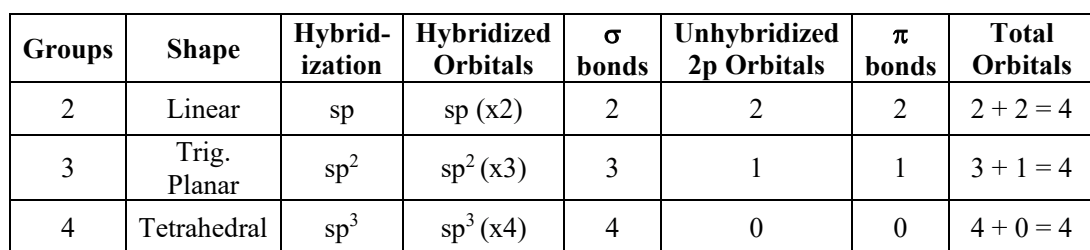
What is the hybridization of groups 1-6 (and their shape?)
Group 1, linear, s or 2p
Group 2, linear, sp
Group 3, trigonal planar, sp2
Group 4, tetrahedral, sp3
Group 5, trigonal bipyramidal, sp3d
Group 6, octahedral, sp3d2
What does bond length mean and how does it relate to bond strength?
Bond length is the distance between the nuclei of two bonded atoms, which influences bond strength; shorter bonds tend to be stronger due to increased nuclear attraction between the atoms.
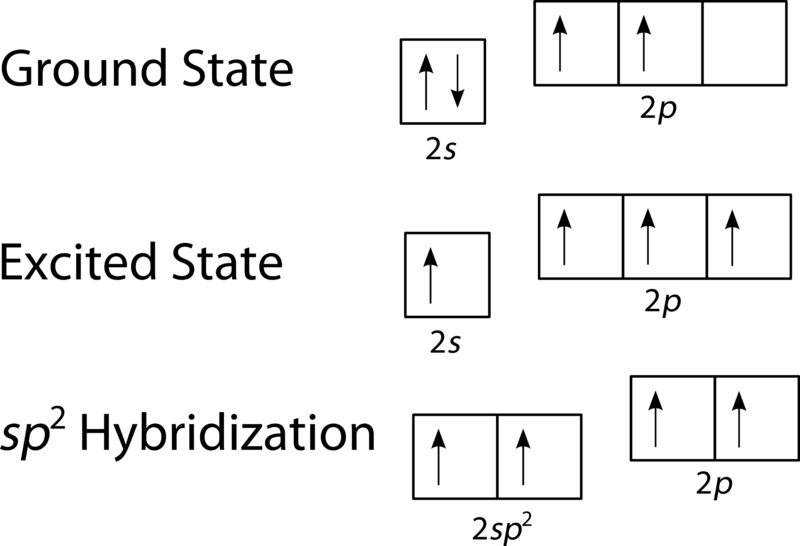
What is hybridization, and how do you find it?
is the concept of combining atomic orbitals to form new hybrid orbitals, relevant for understanding molecular geometry and bonding. To find hybridization, count the number of electron groups around the central atom and match the count to hybridization types: 2 domains = sp, 3 = sp2, 4 = sp3, etc.
hybridization schemes
describes the process by which atomic orbitals mix to form new hybrid orbitals suited for bonding. Common schemes include sp, sp2, sp3, and their respective geometries based on electron domain count. These schemes help predict molecular shapes and bond angles in various compounds.
electron-sea model
e-’s on metals are free to move around. This explainsthe conductivity and malleability of metals, as the delocalized electrons allow metal atoms to slide past each other without breaking bonds.

hybridization theory
is a model that explains how atomic orbitals mix to create hybrid orbitals for bonding. It helps in predicting molecular geometry and the arrangement of atoms in a molecule.
valence-bond method
is a quantum mechanical model that describes the formation of covalent bonds through the overlap of atomic orbitals. It emphasizes the role of electron pairs in bond formation and provides insights into molecular structure and bonding interactions.
VSEPR method
is a model used to predict the geometry of molecular structures based on the repulsion between electron pairs around a central atom. It helps in determining the spatial arrangement of atoms in a molecule.
Band theory
theory that states that bands form when orbitals are overlapping from multiple bonds/MOLES worth of orbitals of the same element overlap
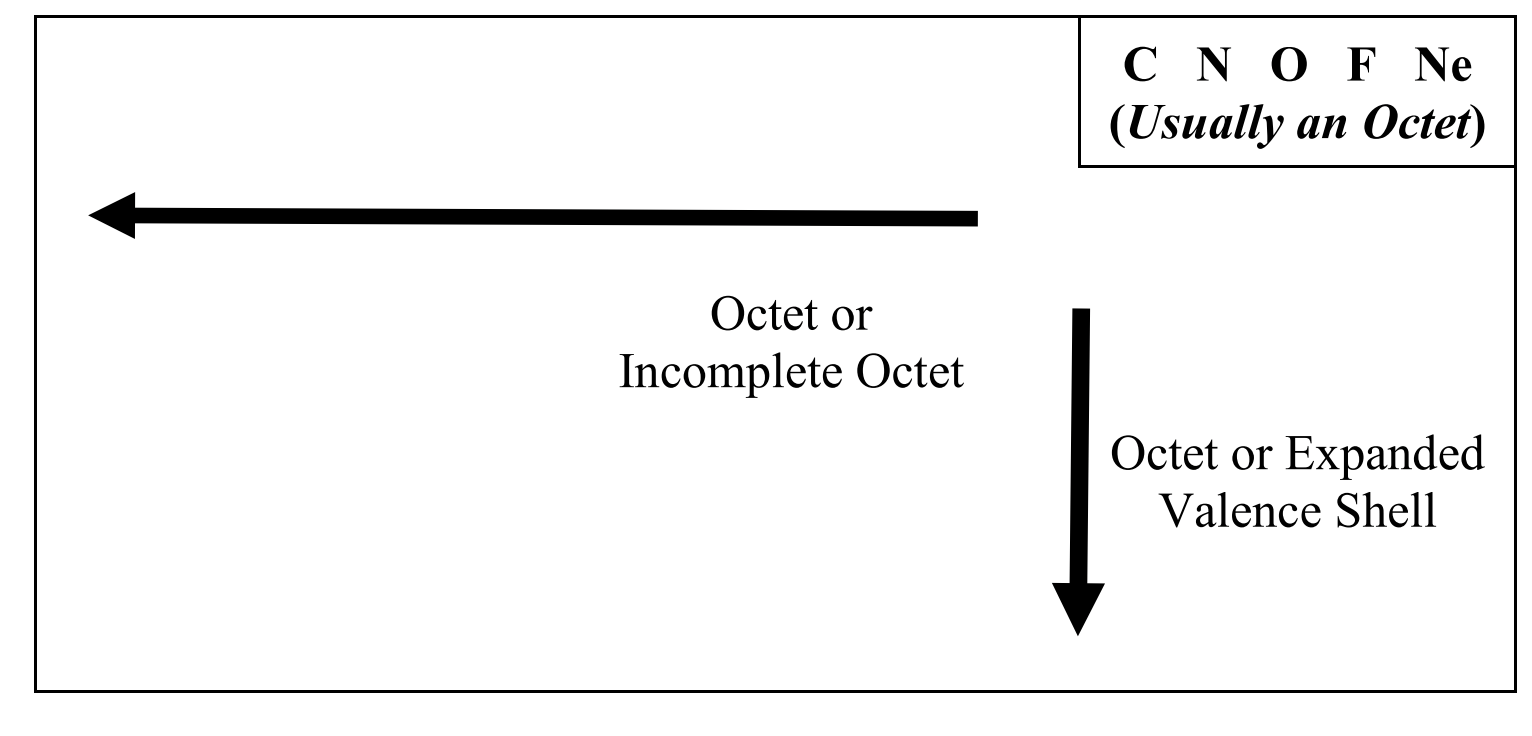
Octet rule and exceptions
is a principle in chemistry stating that atoms tend to form stable molecules by having 8 electrons in their valence shell.
EXCEPTIONS:
The compound NO, free radicals (reactive lone atoms with an unpaired e-)
Incomplete octets- any element to the LEFT OF THE C COLUMN can have less that 8 e- in their val shell. (theres some exceptions, like how C can have a formal charge of +1) (?)
Expanded val. shells: the bigger atoms under the C row can have more than 8 e- in their val shell (for example, P in PCl5 )
Resonance and delocalization
describe a phenomenon where certain molecules can be represented by two or more valid Lewis structures, highlighting the distribution of electrons across different bonds and structures. This results in a more stable configuration due to the delocalization of electrons. (bc where those bonds are? the delocalized e-s are moving back in forth there.)
Drawing lewis structures- steps
1.) find the total # of val e-
2.) draw skeleton w/ single bonds
3.) fill the rest of the val e-s, starting with the terminal atoms
4.) if you have extras, put them on atoms with an atomic number thats bigger than 10
5.) AND if you have more than 1 atom with a Z>10, put it on the atom in the center
6.) Check: do u have an octet? If u do, ur almost done, just see if there’s resonance structures now
7.) IF YOU DON’T: recognize some atoms don’t have an octet (like H, trans. metals (bc they would have empty d-orbitals). If you have this, try to find a lone pair on an atom that’s close by, remove it, and make another bond. Consider resonance structures
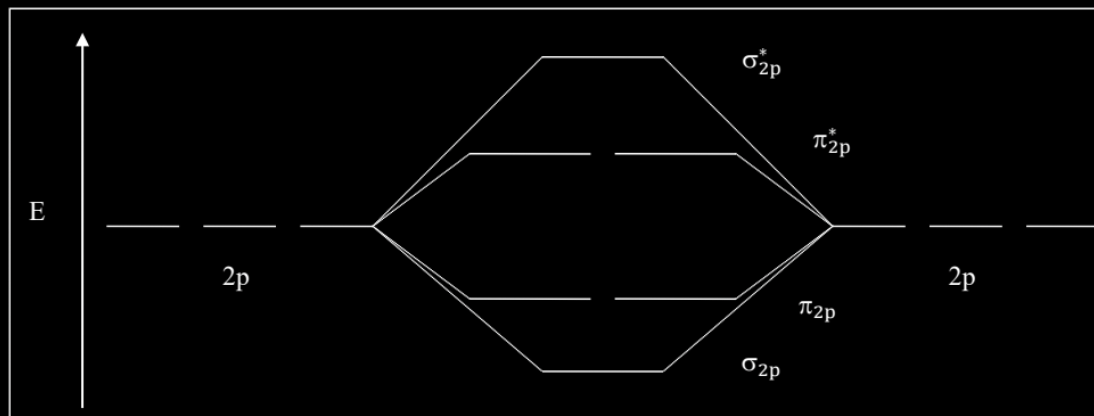
For MO diagrams- what would they look like if it’s an element to the LEFT of O? (N atoms or lower)
The middle orbitals would have an orbital sequence of 2-1-2-1 (alternating). (remember, sigmas have 1 orbital, and pi’s have 2)
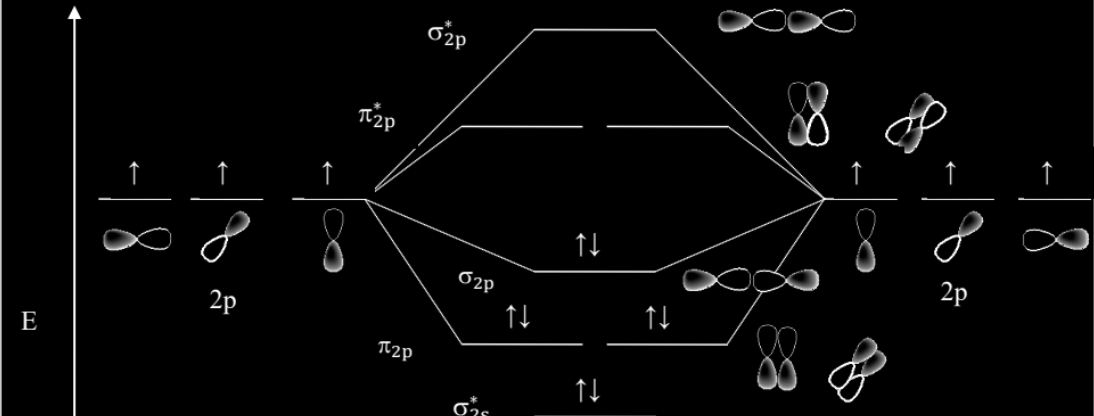
For MO diagrams- what would they look like if it’s an element to the RIGHT of O? (O atoms or higher)
The middle orbitals would have an orbital sequence of 1-2-2-1. (remember, sigmas have 1 orbital, and pi’s have 2)
atomic orbital theory
A theory that describes the behavior of electrons in atoms as waves, leading to the concept of atomic orbitals. It explains how electrons are distributed in an atom and how their arrangements influence chemical bonding.
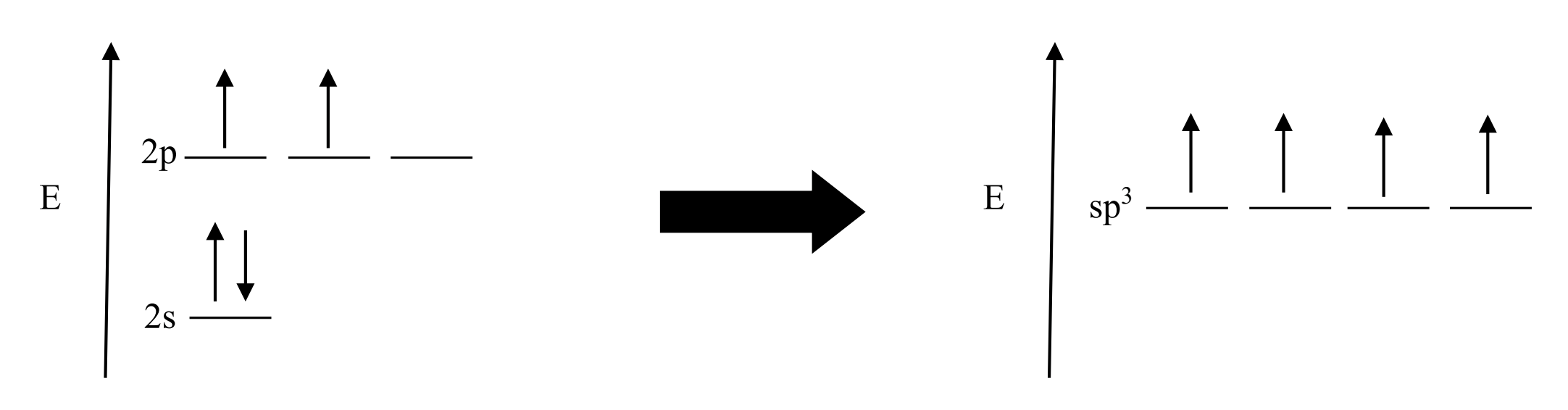
what would a sp3 hybrid orbital look like
An sp3 hybrid orbital results from the mixing of one s orbital and three p orbitals. (ALL IN THE SAME E LEVEL, THERES NO 2P ORBITALS)
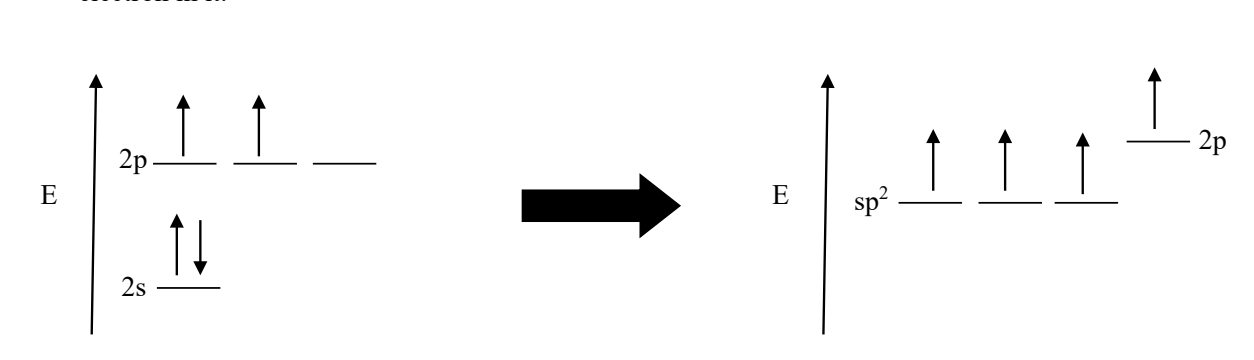
what would a sp2 hybrid orbital look like
results from the mixing of one s orbital and two p orbital. One unhybridized p orbital remains for pi bonding. (you’ll have 3 sp² orbitals and ONE 2p orbital (that has a high E level))
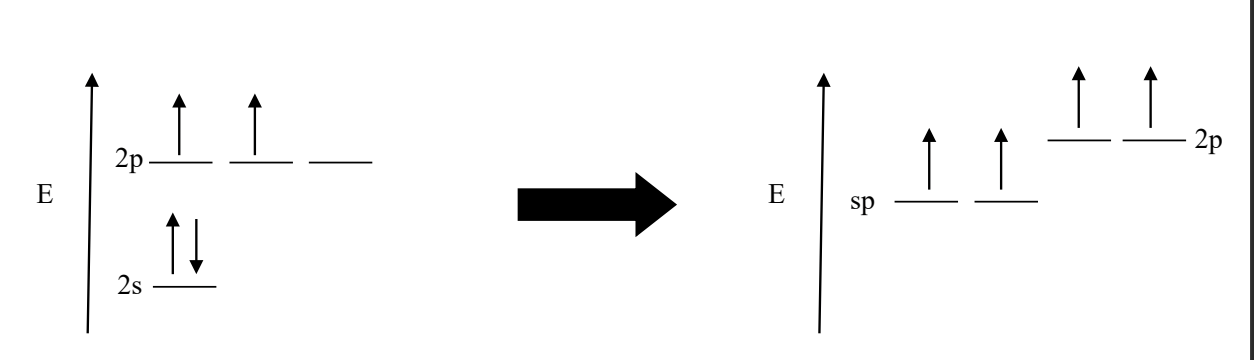
what would a sp hybrid orbital look like
An sp hybrid orbital results from the mixing of one s orbital and one p orbital. This hybridization typically occurs in molecules with triple bonds or in molecules with a linear geometry. (You’ll have TWO sp orbitals and then TWO 2p orbs (on a slightly higher E level)
What bonds do hybridized/unhybridized orbitals usually make? (describe each of the 3 hybs with their bonds) and what’s a good way to remember?
Hybridized orbitals typically form sigma bonds, while unhybridized orbitals usually form pi bonding.
sp³= 4 sigma bonds
sp²= 3 sigma bonds and 1 pi bond;
sp= 2 sigma bonds and 2 pi bonds.
THE NUMBER OF HYBRIDIZED ORBITALS = THE NUMBER OF SIGMA BONDS
THE NUMBER OF 2P ORBITALS = THE NUMBER OF PI BONDS
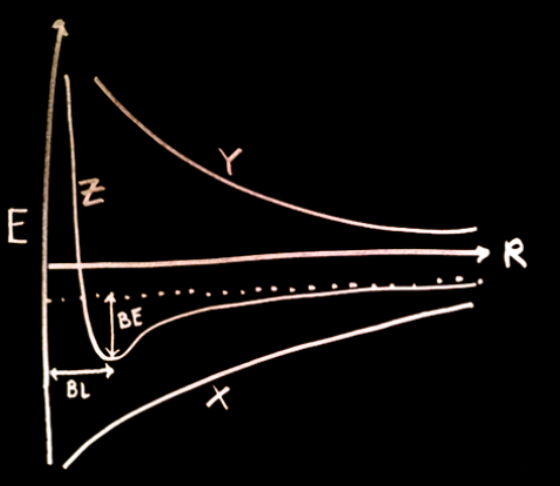
What do all the variables on this graph mean?
E= energy
R= the distance between 2 bonding nuclei
X= the bonding interaction. As the distance between the nuclei (R) decreases, the energy decreases (meaning it’s more stable). This curve is also representing the constructive interference/orbital overlap (and as u know, the more that happens, the more stable it gets.)
Y= the REPLUSION that happens when the nuclei get too close together (which is unfavorable!) which causes there ti be an INCREASE of E at low R. This is the anti bonding curve, showing the destructive interference
Z= mathematical sum of bonding/anitbonding curves, called the SUM CURVE. if ur looking at the dip of the curve: the bottom part is where the bond occurs. The BL label shows the equilibrium bond length, and the BE label shows the equilibrium bond energy
when putting the electrons into hyb. schemes, what’s important to remember?
Even while the sp/p orbitals are on different E levels, they are so close that you fill them like they are all in the same row!
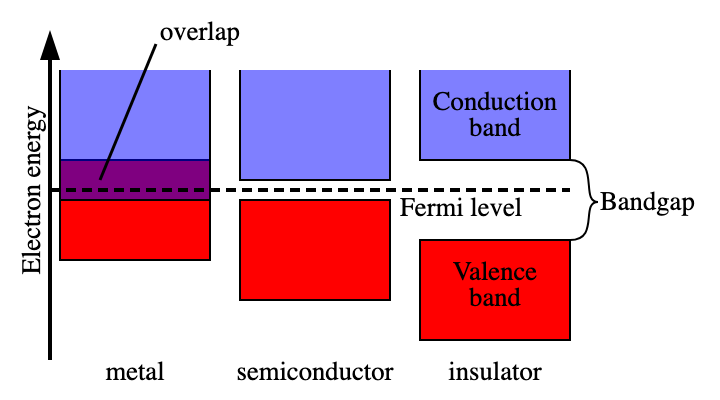
Regarding band theory- What do the bands look like for metals, semiconductors, and insulators?
metals have a small gap/overlap, semiconductors/semi-metals have a bigger gap but its not huge, but insulators have really big gaps between the valence band and the conduction band. This gap determines their electrical conductivity, with metals conducting electricity easily, semiconductors requiring some energy to conduct, and insulators preventing conduction.
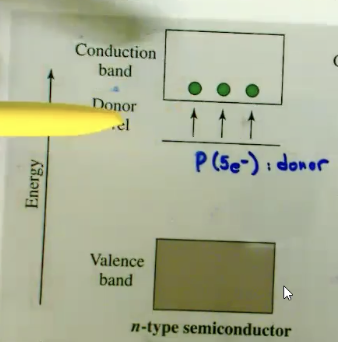
What is a n-type semiconductor?
type of semiconductor that has been doped with elements that provide extra electrons (AKA ELECTRON RICH) (like Silicon, Phosphorus), increasing its electron concentration. It puts an anergy level just below the conduction band to make it easier for e-’s to move into there.
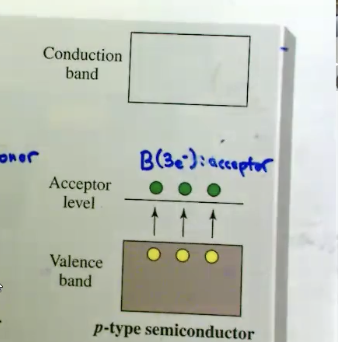
What is a p-type semiconductor?
type of semiconductor that has been doped with elements that are ELECTRON POOR (like boron, gallium) just above the valence band, creating holes that facilitate electron movement, enhancing its conductivity.
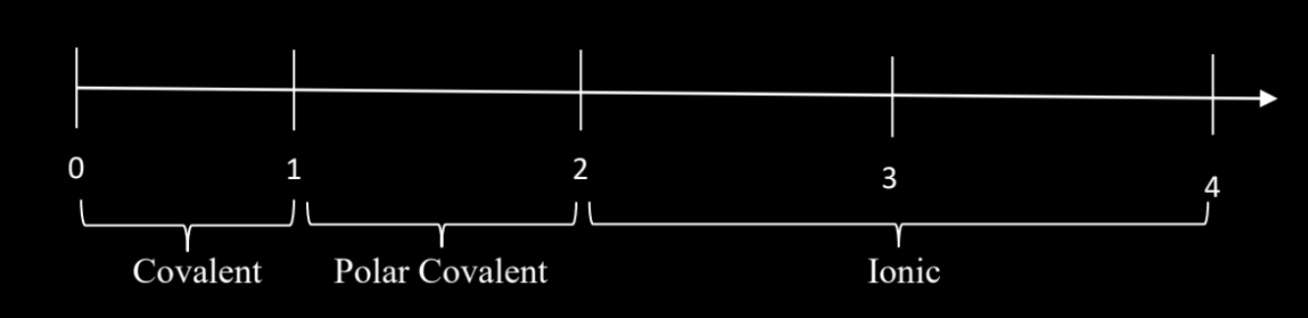
How to calculate the POLARITY of a bond
find the difference in EN (|ΔEN|) between 2 atoms
Scale of polarity (how to know if something is covalent/polar covalent/ionic based on polarity values)
covalent= (|ΔEN|) < 1 and ionic if (|ΔEN|) >2
What is the system, surroundings, boundary, and universe?
System- The part of the universe for study
surroundings- anything that’s no defined in the system
boundary- line/surface that seperates the system and surroundings
universe- both the system and surroundings
what are the 3 types of systems? (and examples for each, in terms of cups of water)

energy- definition
The capacity to do work or produce heat
What are the 3 ways the energy of an object can be described?

What are the 2 ways energy can be transferred between objects? And which of the 3 types of energy of an object is associated with what of the 2 ways?

SI units of J, Nm, and N
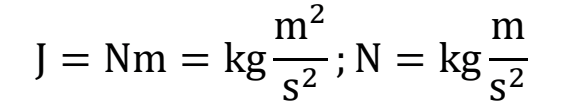
what is the zeroth law of thermodynamics
if two systems are each in thermal equilibrium with a third system, then they are in thermal equilibrium with each other
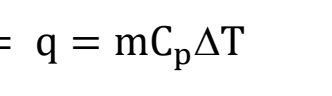
What do all of the variables mean in the heat equation?
q: Heat required to change temperature. Units of J or cal (kcal).
m: mass. Mass is usually in units of g but occasionally moles. Thus, “m” really symbolizes amount.
∆T: temperature change. Using either units of °C or K will result in the same answer because the temperature change is numerically the same for both °C and K.
Cp: specific heat, which depends on the substance. Units of J/g°C or cal/g°C, but occasionally g might be substituted for moles. For the same reason given for ∆T above, J/g°C is equivalent to J/g K. Some example substances are given below.
C = mCp: heat capacity, which is the quantity of heat required to change the temperature of the system by one degree. Units of J/°C or cal/°C
Difference between SPECIFIC HEAT and HEAT CAPACITY

What is the trick for solving heat problems (with the different q values)
You must find all of the q values present, and how they are related (like, what endo and eco, and where their heat is flowing)
In what situations should you use q = n∆H ? (also, it is _____ based)
When….
-a reaction occurs
-a solution is made by mixing
-a phase change occurs
ENTHALPY based