GW BGZ 2024 Case 8 - A glass of (soy) milk before sleep
1/41
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
42 Terms
What is sarcopenia?
Sarcopenia is the progressive and generalized loss of skeletal muscle mass, strength, and function that occurs with aging. It affects both muscle quantity and quality, meaning that remaining muscle fibers often generate less force. It is considered a geriatric syndrome with multifactorial origins including metabolic, hormonal, nutritional, and neuromuscular factors.
Key consequences of sarcopenia include:
Frailty
Reduced mobility and walking speed
Increased risk of falls and fractures
Loss of independence and reduced quality of life
When does age-related muscle loss begin, and at what rate?
Muscle loss starts gradually after age 30, with noticeable decline accelerating during the fourth or fifth decade.
After age 50, muscle mass declines at an estimated rate of 0.5–1% per year, while strength declines faster at 1–2% per year.
After age 60, both mass and strength losses accelerate.
Mechanism:
Loss results from a chronic imbalance between muscle protein synthesis (MPS) and muscle protein breakdown (MPB), with synthesis insufficient to counteract breakdown.
Contributing factors:
Hormonal changes (decline in testosterone, growth hormone, IGF-1)
Reduced physical activity
Chronic inflammation
Inadequate protein intake
Neuromuscular changes
How does sarcopenia affect muscle strength and physical performance?
Strength declines faster than mass, reflecting both reduced muscle fiber quality and neuromuscular impairments.
Functional consequences include:
Reduced walking speed and balance
Difficulty performing daily activities (e.g., climbing stairs, rising from chairs, carrying objects)
Increased susceptibility to injury
Severe sarcopenia contributes to frailty, hospitalization, and higher mortality risk.
What is the physiological basis of sarcopenia?
Sarcopenia develops due to multiple interacting mechanisms:
Chronic imbalance in muscle protein turnover:
Muscle is in constant turnover (~1–2% per day).
Aging shifts balance toward net protein breakdown, causing gradual muscle loss.
Anabolic resistance:
Aging muscles respond less efficiently to anabolic stimuli such as protein intake or resistance exercise.
Requires higher protein doses to achieve comparable muscle protein synthesis (MPS) to younger adults.
Reduced physical activity:
Less mechanical loading → decreased MPS, smaller fiber size, fat infiltration.
Immobilization accelerates muscle loss.
Hormonal changes:
Declines in testosterone, growth hormone, and IGF-1 reduce muscle maintenance and growth capacity.
Neuromuscular degeneration:
Loss of motor neurons → fiber denervation → atrophy or altered fiber composition.
Chronic low-grade inflammation (“inflammaging”):
Impairs anabolic signaling and protein synthesis, reduces muscle regenerative capacity.
Reduced dietary protein intake:
Due to lower appetite, dental issues, digestive problems, or socioeconomic factors, limiting amino acids for MPS.
What are the main mechanisms of muscle loss with aging?
Inadequate protein intake: Older adults often consume less protein than required.
Anabolic resistance: Reduced efficiency of muscles to use ingested protein for MPS.
Increased protein needs: Illness, chronic disease, and inflammation raise protein requirements.
Negative energy balance: Calorie deficits exacerbate protein breakdown.
Secondary contributors: Sedentary behavior, hormonal changes, chronic inflammation.
Bottom line: Aging and lifestyle factors tip the balance toward net muscle loss.
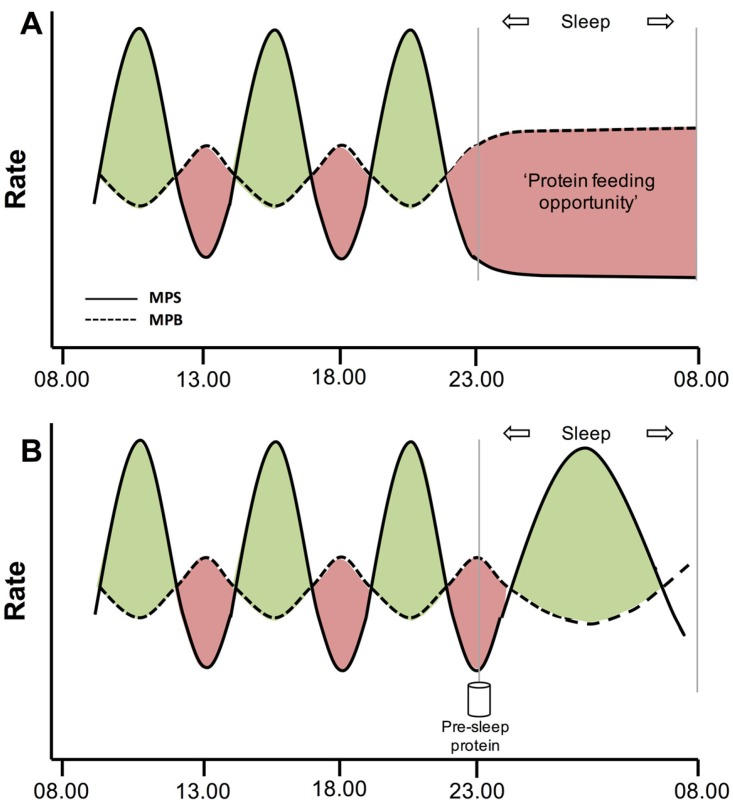
How does nutrition regulate muscle protein metabolism?
Muscle mass is regulated by balance between MPS and MPB.
Protein and amino acids:
Stimulate MPS (especially essential amino acids, notably leucine)
Leucine activates the mTOR pathway, a master regulator of protein synthesis
Insulin from meals suppresses MPB
Protein dose and distribution:
Older adults require ~30–40 g protein per meal (vs. ~20 g for young adults)
4–6 evenly distributed protein-rich meals per day maximize daily MPS
Pre-sleep protein intake supports overnight MPS
Protein type:
Fast-digesting, leucine-rich proteins (whey) → highest MPS
Slow-digesting proteins (casein, soy) less effective unless fortified with leucine
How does exercise influence sarcopenia?
Resistance exercise is the most effective intervention to stimulate MPS.
Effects of resistance training:
Increases muscle fiber size and strength
Improves neuromuscular coordination
Enhances insulin sensitivity
Primes muscle to better respond to dietary protein (overcoming anabolic resistance)
Post-exercise MPS remains elevated for 16–48 hours depending on training status.
Synergistic effect when combined with protein ingestion results in greater net muscle protein accretion.
How can sarcopenia be reduced or prevented?
Primary strategies:
Adequate dietary protein intake:
1.2–1.5 g protein/kg body weight/day for older adults
Spread evenly across 4–6 meals
Include leucine-rich proteins and consider pre-sleep protein
Resistance exercise:
Stimulates MPS and improves muscle quality and strength
Counteracts anabolic resistance
Maintain daily physical activity:
Avoid prolonged inactivity
Improves metabolic health and preserves muscle mass
Secondary strategies:
Address chronic inflammation, hormonal imbalances, and underlying diseases
Optimize nutrition (calories, micronutrients, hydration)
Key takeaway: Sarcopenia is multifactorial but can be slowed or partially reversed with combined nutritional and exercise interventions.
How does protein type and timing influence MPS in older adults?
Protein type:
Fast-digesting, leucine-rich proteins (whey) maximize MPS
Slow-digesting proteins less effective unless fortified with leucine
Protein timing:
Post-exercise protein intake enhances recovery and MPS
Pre-sleep protein provides amino acids during overnight fasting
Total daily protein distribution is more important than a single “anabolic window”
Optimal strategy: 4–6 evenly spaced protein-rich meals with 30–40 g protein per meal for older adults.
What is the role of leucine and mTOR in muscle protein synthesis?
Leucine: Essential amino acid that acts as a signal and substrate for MPS.
Mechanism:
Leucine activates the mTOR (mechanistic target of rapamycin) pathway, which regulates cell growth and protein synthesis.
mTOR activation leads to ribosomal activation and increased translation of muscle proteins.
Other signals: Insulin and resistance exercise also activate mTOR.
Practical implication: Leucine-rich proteins are essential to counteract anabolic resistance in older adults.
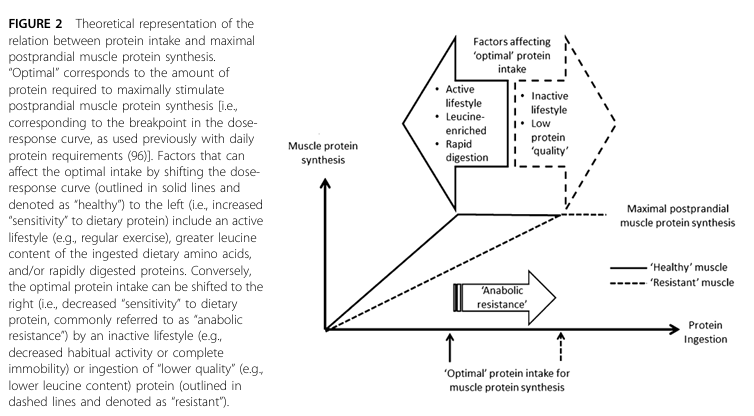
What is anabolic resistance?
Anabolic resistance is the reduced ability of skeletal muscle, particularly in older adults, to increase muscle protein synthesis (MPS) in response to anabolic stimuli, such as dietary protein intake, essential amino acids, and resistance exercise.
In younger individuals:
Protein ingestion leads to a robust increase in MPS.
In older adults:
The same protein intake produces a blunted MPS response.
Higher protein doses are required to achieve maximal stimulation
What is the primary physiological manifestation of anabolic resistance?
The primary manifestation is a blunted stimulation of muscle protein synthesis (MPS) after protein or amino acid ingestion.
Key characteristics:
Reduced postprandial (after-meal) increase in MPS
Possibly reduced suppression of muscle protein breakdown (MPB)
Higher amino acid threshold required to activate anabolic pathways
Importantly:
The maximum capacity for MPS is often preserved if sufficient protein is consumed.
The issue lies in reduced sensitivity, not total inability.
Thus, anabolic resistance is best described as a rightward shift in the dose-response curve of protein intake versus MPS.
How does anabolic resistance contribute to sarcopenia?
Anabolic resistance contributes to sarcopenia by promoting a chronic negative muscle protein balance over time.
Mechanism:
Each meal stimulates less MPS than in younger individuals
Daily protein intake becomes insufficient to maintain muscle mass
Small deficits accumulate over years
Consequences:
Progressive loss of muscle mass
Decline in strength and physical function
Increased risk of frailty and disability
Thus, anabolic resistance is a key physiological barrier to maintaining muscle mass with aging.
What are the main mechanisms underlying anabolic resistance?
Anabolic resistance is multifactorial, involving impairments at several levels:
Digestive and absorptive limitations
Reduced amino acid availability
Impaired muscle perfusion and delivery
Reduced insulin sensitivity
Defective intracellular signaling (mTOR pathway)
Physical inactivity
Chronic low-grade inflammation
Neuromuscular changes
These mechanisms interact across the entire pathway:
Digestion → absorption → circulation → delivery → uptake → intracellular signaling → protein synthesis
How do digestion and absorption contribute to anabolic resistance?
With aging, there may be impairments in protein digestion and amino acid absorption, leading to reduced systemic availability of amino acids.
Key points:
Slower or less efficient breakdown of dietary protein
Reduced absorption efficiency in the gut
Increased splanchnic extraction, meaning more amino acids are retained in the gut and liver
Consequences:
Fewer amino acids enter systemic circulation
Reduced availability of amino acids for skeletal muscle
Blunted stimulation of MPS
This is often described as amino acids “remaining in the gut” rather than reaching muscle tissue.
How does impaired muscle perfusion contribute to anabolic resistance?
Muscle perfusion refers to blood flow to skeletal muscle, which is essential for delivering amino acids and insulin.
In aging:
Insulin-mediated vasodilation is reduced
Capillary recruitment is impaired
Blood flow to muscle after meals is diminished
Possible causes:
Endothelial dysfunction
Increased vasoconstrictors (e.g., endothelin-1)
Consequences:
Reduced amino acid delivery to muscle
Impaired nutrient uptake
Blunted anabolic signaling
Thus, even if amino acids are present in circulation, they may not effectively reach muscle tissue.
What is the role of insulin resistance in anabolic resistance?
Insulin plays a permissive role in muscle protein metabolism by:
Suppressing muscle protein breakdown (MPB)
Facilitating nutrient uptake and blood flow
In aging:
Insulin resistance develops
Reduced ability to suppress MPB
Impaired nutrient delivery and utilization
Consequences:
Less favorable net protein balance
Reduced efficiency of post-meal anabolic response
Although insulin does not strongly stimulate MPS directly, it is essential for maintaining overall protein balance.
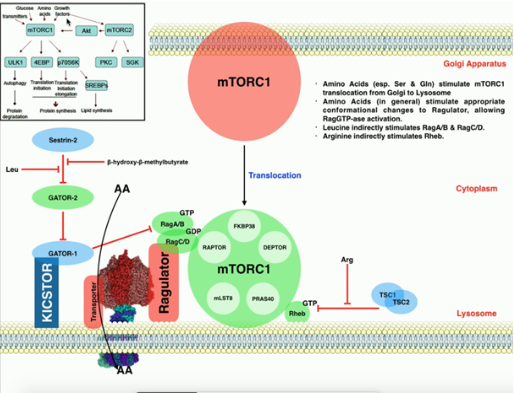
How does impaired intracellular signaling contribute to anabolic resistance?
The most critical intracellular pathway is the mTOR (mechanistic target of rapamycin) signaling pathway, which regulates protein synthesis.
In young muscle:
Amino acids (especially leucine) activate mTOR
mTOR stimulates translation and MPS
In aging muscle:
Reduced sensitivity of mTOR to amino acids
Impaired activation of downstream targets (e.g., S6K)
Possible reduction in ribosomal content
Consequences:
Higher amino acid threshold needed to activate MPS
Reduced efficiency of protein synthesis
This represents a central cellular defect in anabolic resistance.
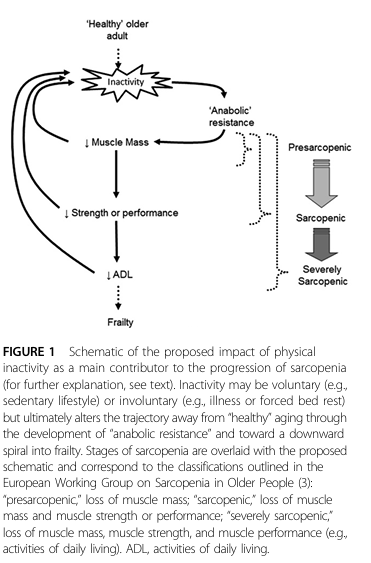
What is the role of physical inactivity in anabolic resistance?
Physical inactivity is one of the most important and modifiable contributors to anabolic resistance.
Effects of inactivity:
Reduced basal and postprandial MPS
Impaired mTOR signaling
Decreased amino acid transport into muscle
Reduced insulin sensitivity
Diminished muscle blood flow
Even short-term inactivity (e.g., reduced daily steps, bed rest) can:
Rapidly induce anabolic resistance
Accelerate muscle loss
Key insight:
Anabolic resistance is not solely due to aging, but strongly influenced by inactivity
Inactivity can mimic or accelerate the aging process in muscle
How does chronic inflammation contribute to anabolic resistance?
Aging is associated with chronic low-grade inflammation (“inflammaging”), characterized by elevated cytokines.
Effects on muscle:
Impairs mTOR signaling
Reduces muscle protein synthesis
Promotes protein breakdown
Inhibits muscle regeneration and satellite cell activity
Consequences:
Reduced anabolic responsiveness
Impaired recovery and adaptation
Inflammation creates a catabolic environment that opposes muscle maintenance.
Can anabolic resistance be reversed?
Anabolic resistance cannot be completely reversed, as it is partly driven by intrinsic aging processes.
However, it can be significantly attenuated or partially overcome through targeted interventions.
Key concept:
Aging reduces sensitivity, but capacity for muscle growth remains under optimal conditions
Thus, appropriate lifestyle and nutritional strategies can restore much of the anabolic response.
What nutritional strategies help reduce anabolic resistance?
Higher protein intake per meal:
~0.4 g/kg body weight per meal
Ensures sufficient amino acid stimulus
Higher total daily protein intake:
≥1.2 g/kg/day
Leucine-rich proteins:
Leucine activates mTOR and stimulates MPS
Whey protein is particularly effective
Protein distribution:
Evenly spaced meals maximize repeated MPS stimulation
Pre-sleep protein ingestion:
~40 g protein supports overnight MPS
Use of rapidly digestible proteins:
Improves amino acid availability and peak MPS response
How does resistance exercise reduce anabolic resistance?
Resistance exercise is the most effective intervention to improve anabolic sensitivity.
Mechanisms:
Activates mTOR signaling
Increases muscle blood flow
Enhances amino acid transport into muscle
Improves insulin sensitivity
Increases ribosomal capacity
Effects:
Restores responsiveness to protein intake
Enhances MPS for up to 24 hours post-exercise
Key concept:
Exercise primes muscle to respond more effectively to amino acids
Why is the combination of exercise and protein intake important?
Exercise and protein intake have a synergistic effect:
Exercise:
Increases sensitivity of muscle to amino acids
Protein intake:
Provides the building blocks for protein synthesis
Together:
Maximize MPS
Improve net protein balance
Promote muscle maintenance and growth
Exercise alone is insufficient without amino acids, and protein alone is less effective without the stimulus provided by exercise.
What are the main physiological effects of protein supplementation in older adults?
Stimulates muscle protein synthesis (MPS)
Improves net protein balance (↑ MPS vs MPB)
Helps overcome anabolic resistance
Maintains or increases lean body mass
Improves muscle strength and function
Reduces risk/progression of sarcopenia
Supports recovery and muscle remodeling
Useful when dietary protein intake is insufficient
Why is whey protein particularly effective for older adults?
Rapid digestion → quick rise in blood amino acids
High in essential amino acids (EAAs)
Very rich in leucine → strong mTOR activation
Produces greater MPS response vs other proteins
Helps overcome anabolic resistance
Convenient and easy to consume (important with low appetite)
How does casein protein differ from whey protein?
Slow digestion → gradual amino acid release
Lower peak MPS compared to whey
Better for sustained amino acid availability
Useful for pre-sleep protein intake
Less effective alone for overcoming anabolic resistance
What is the role of leucine in protein supplementation?
Key essential amino acid for MPS
Activates mTOR pathway
Acts as a signal + substrate
Higher leucine needed in older adults
Helps overcome anabolic resistance
Found in high amounts in whey protein
What is the recommended daily protein intake for older adults?
General recommendation:
≥1.2 g/kg body weight/day
Higher needs in:
Illness, inflammation, or injury → up to 1.5 g/kg/day
Higher than young adults (~0.8 g/kg/day)
Needed to compensate for anabolic resistance
Why is protein distribution across the day important?
~0.4 g/kg per meal
Typically 30–40 g protein per meal
Higher than young adults (~20 g)
Required to maximize MPS
Helps overcome anabolic resistance thresholdMPS is stimulated per meal, not continuously
Even distribution → multiple MPS peaks
Prevents long periods of low amino acid availability
Example: 3–5 protein-rich meals/day
More effective than skewed intake (e.g., protein only at dinner)
What is the role of pre-sleep protein intake?
Provides amino acids during overnight fasting
Supports overnight MPS
Reduces muscle breakdown at night
Typical recommendation: ~40 g protein before sleep
Often uses casein (slow digestion)
What types of proteins are there (amino acid classification)?
Essential amino acids (EAAs):
Cannot be synthesized → must come from diet
9 EAAs: leucine, isoleucine, valine, lysine, methionine, phenylalanine, threonine, tryptophan, histidine
Leucine = key trigger of MPS (via mTOR)
Required for muscle protein synthesis (MPS)
Non-essential amino acids (NEAAs):
Can be synthesized by body
Examples: alanine, glycine, glutamine, serine
Important for metabolism but do not stimulate MPS alone
What is protein quality?
Based on:
EAA content (especially leucine)
Digestibility
High-quality protein:
High EAAs
Easily digestible
Strong MPS stimulation
Animal proteins:
Higher quality
85–95% digestible
Plant proteins:
Lower quality (on average)
50–75% digestible
Due to fiber & anti-nutritional factors
What is the difference between animal and plant proteins?
Animal proteins:
High EAAs (complete proteins)
High leucine
High digestibility (85–95%)
Strong MPS response
Plant proteins:
Lower EAA content (often incomplete)
Limiting amino acids:
Lysine (grains)
Methionine (legumes)
Lower digestibility (50–75%)
Weaker MPS per gram
Why do plant proteins stimulate less MPS?
Lower leucine content
Lower total EAAs
Presence of anti-nutritional factors:
Fiber
Phytates
Tannins
Slower digestion & absorption
Higher splanchnic extraction
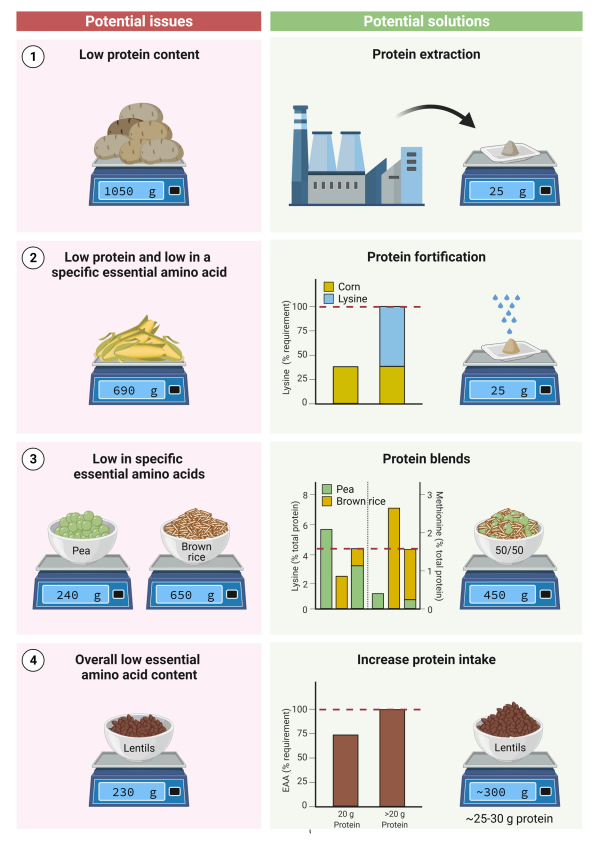
How can plant protein quality be improved?
Increase total protein intake
~10–20% more needed
Combine proteins (complementary):
Grains (low lysine) + legumes (high lysine)
Choose high-quality plant proteins (soy, pea)
Use protein isolates/concentrates:
↑ digestibility (~90%)
↓ anti-nutritional factors
Fortify with amino acids:
Leucine
Lysine
Methionine
Processing methods (to increase digestibility):
Fermentation
Hydrolysis
Heating
What is the difference between vegan, vegetarian, and omnivorous diets?
Vegan diet:
No animal products
Protein: legumes, soy, grains, nuts
Requires careful planning (EAAs)
Vegetarian diet:
No meat/fish
May include:
Dairy (lacto)
Eggs (ovo)
Better protein quality than vegan
Omnivorous diet:
Includes plant + animal foods
High protein quality
Usually no planning needed
What is muscle reconditioning and how does muscle exhibit plasticity?
Muscle reconditioning refers to the ability of skeletal muscle to adapt to environmental, lifestyle, or physiological conditions, either positively (hypertrophy, increased function) or negatively (atrophy, decreased function).
Muscle is highly plastic due to constant protein turnover, which involves breakdown and synthesis of proteins at a daily rate of 1–2% of total muscle protein.
This turnover allows adaptation to stimuli such as:
Resistance or endurance exercise
Nutritional intake (amino acids, protein)
Hormonal changes
Plasticity is critical for maintaining functional capacity, metabolic health, and recovery after illness or immobilization.
What is muscle deconditioning and which conditions contribute to it?
Muscle deconditioning occurs when muscle adapts negatively due to inactivity, disease, or aging.
Causes include:
Immobilization: casting, bed rest
Sarcopenia: age-related loss of muscle mass and strength
Cancer cachexia: rapid muscle loss due to cancer treatment or tumor metabolism
Chronic diseases: COPD, cardiovascular disease, type 2 diabetes (affects muscle quality, mass, and glucose uptake)
Muscle deconditioning results in decreased functional capacity, insulin resistance, and reduced quality of life.
Do carbohydrates or fats affect muscle protein synthesis?
Carbohydrates: increase insulin but do not stimulate muscle protein synthesis directly.
Fat: may delay gastric emptying and amino acid absorption, but no direct effect on synthesis itself.
Protein is the primary driver of anabolic response; timing relative to exercise is crucial.
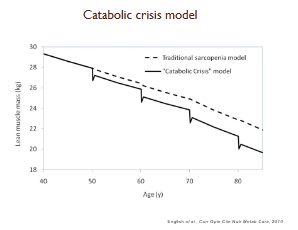
What is the catabolic crisis model?
Episodes of reduced physical activity (illness, hospitalization) cause rapid muscle loss.
Post-crisis, the muscle may not fully recover to pre-event levels → progressive decline in functional capacity over time.
Highlights the importance of prevention, early mobilization, and rehabilitation.
Take home messages
You are what you ate and if you are physically active you are more of what you just ate (athletes)
Opposite in care
Protein ingestion and muscle contraction stimulate muscle tissue protein synthesis
Physical (in)activity (de)sentisizes skeletal muscle tissue to the anabolic properties of dietary protein ingestion
Protein is required to support muscle conditioning in both health and disease