Acids, Bases and Salts
1/17
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
18 Terms
acid
A substance that tastes sour, reacts with metals and carbonates, pH below 7, and turns blue litmus red.

base
A substance that tastes bitter, feels slippery, pH above 7, and turns red litmus paper blue.

salt
An ionic compound made from the neutralization of an acid with a base. pH of 7 (neutral)
neutral
A solution with a pH of 7.

acidic
pH < 7;
Contains more hydrogen ions than hydroxide ions
basic
pH > 7;
Contains more hydroxide ions than hydrogen ions
alkaline
A synonym for basic solutions; pH > 7
pH
A number scale use to identify acids and bases. "Potential/Power of Hydrogen" Above 7 = basic, below 7 = acidic, 7 = neutral

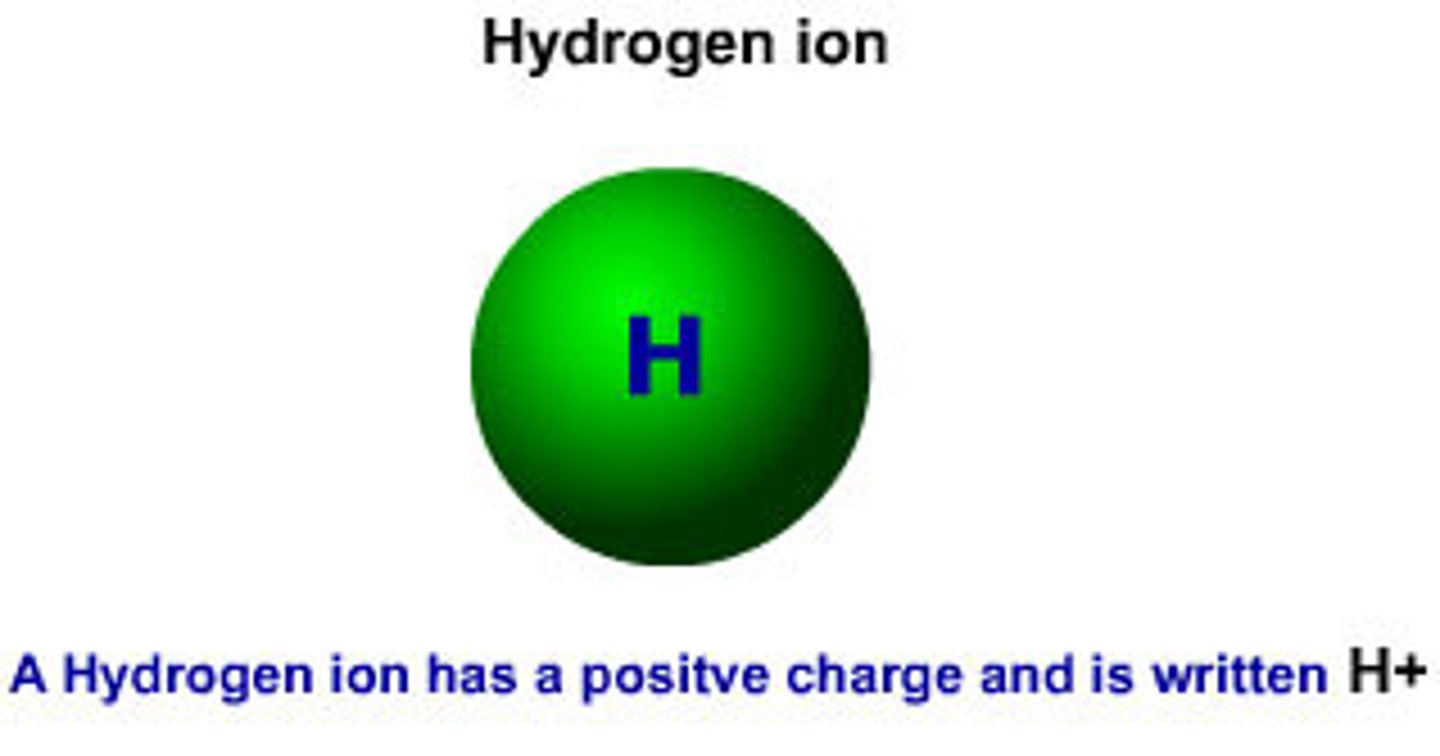
H+
hydrogen ion; when H+ ions outnumber the OH- ions a solution is acidic.
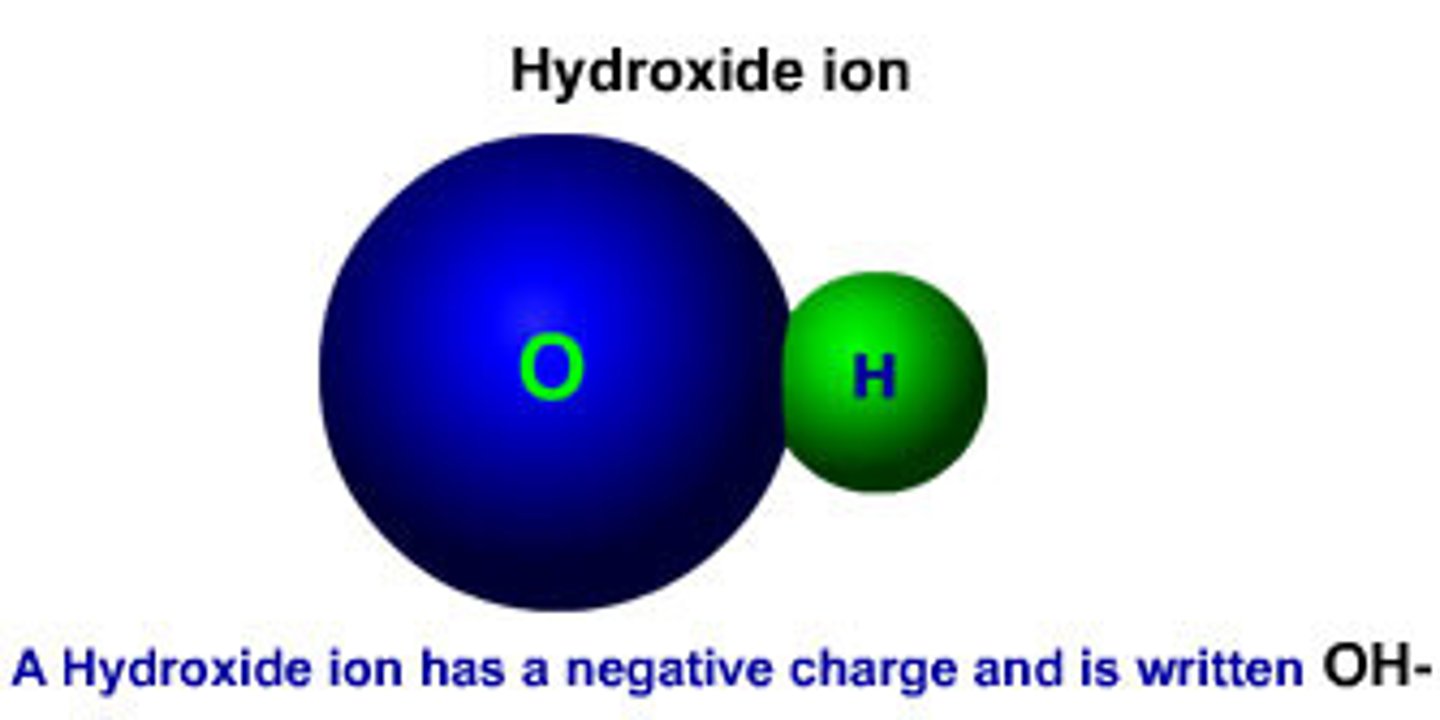
OH-
hydroxide ion; when OH- ions outnumber the H+ ions a solution is basic.
ion
An atom or group of atoms that has a positive or negative charge.
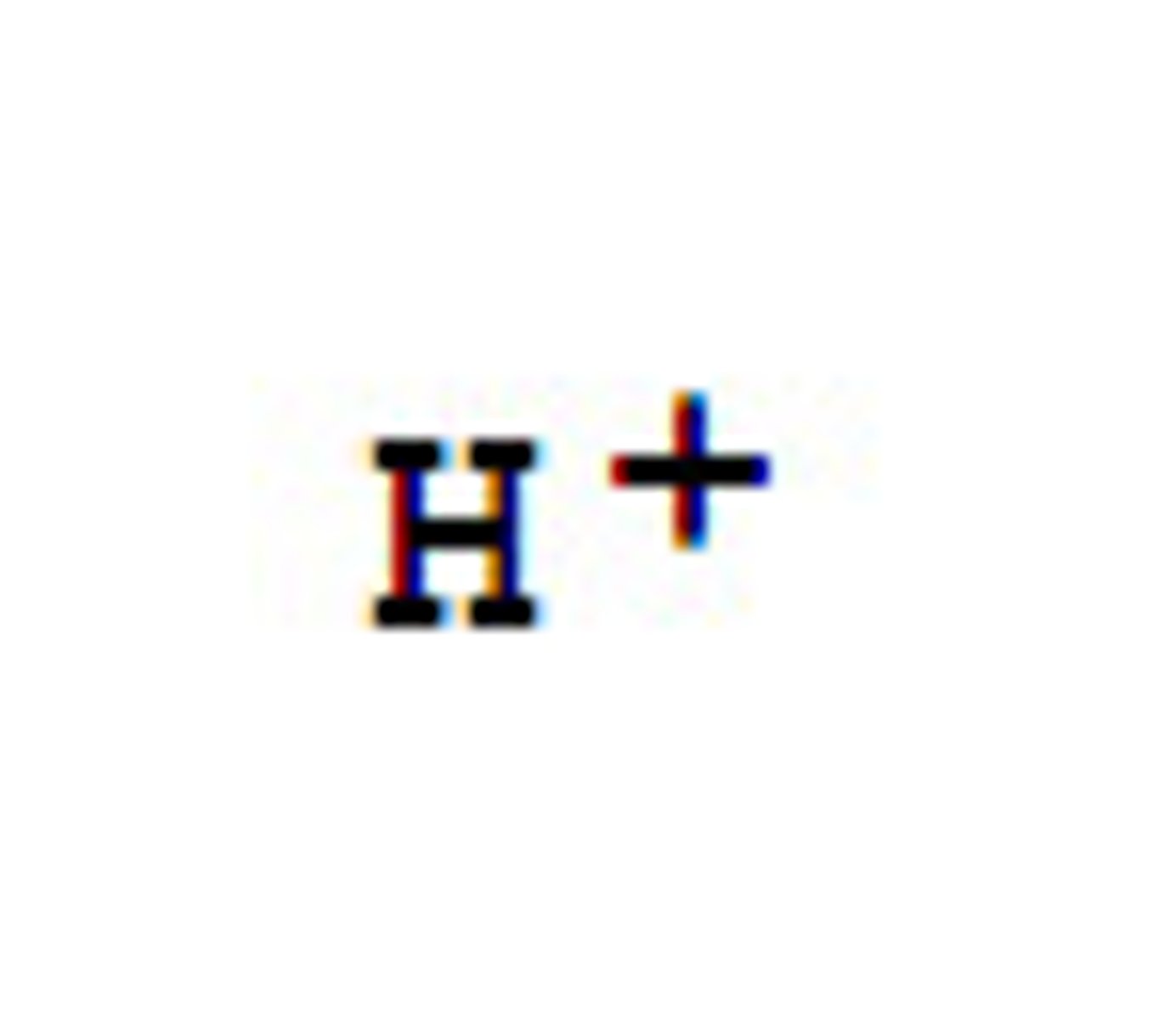
compound
A substance made up of atoms of two or more different elements joined by chemical bonds
The gas given off when a metal reacts with an acid
hydrogen
The way to separate an insoluble solid from a solution
filtering
The way to separate a soluble salt from a solution
evaporation
precipitate
the solid product (insoluble)
soluble
when a chemical dissolves
insoluble
when a chemical does not dissolve