Bio Chem Quiz 6
1/39
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
40 Terms
Nonpolar Amino Acid
Contain one amino group, one
carboxyl group, and a nonpolar side chain
Polar Neutral
Contain polar but neutral side
chains (6 amino acids) (alcohol)
Polar Acidic Amino Acids
Contain a carboxyl group as part of
the side chains (2 amino acids)
Polar Basic Amino Acids
Contain an amino group as part of
the side chain (3 amino acids)
Amino acids are organic compounds that contain a
_____ group and a _____ group and are found in
proteins as _____.
amino; carboxyl; alpha-amino acids
3 multiple choice options
Incomplete dietary proteins contain inadequate
amounts of:
one or more essential amino acids
3 multiple choice options
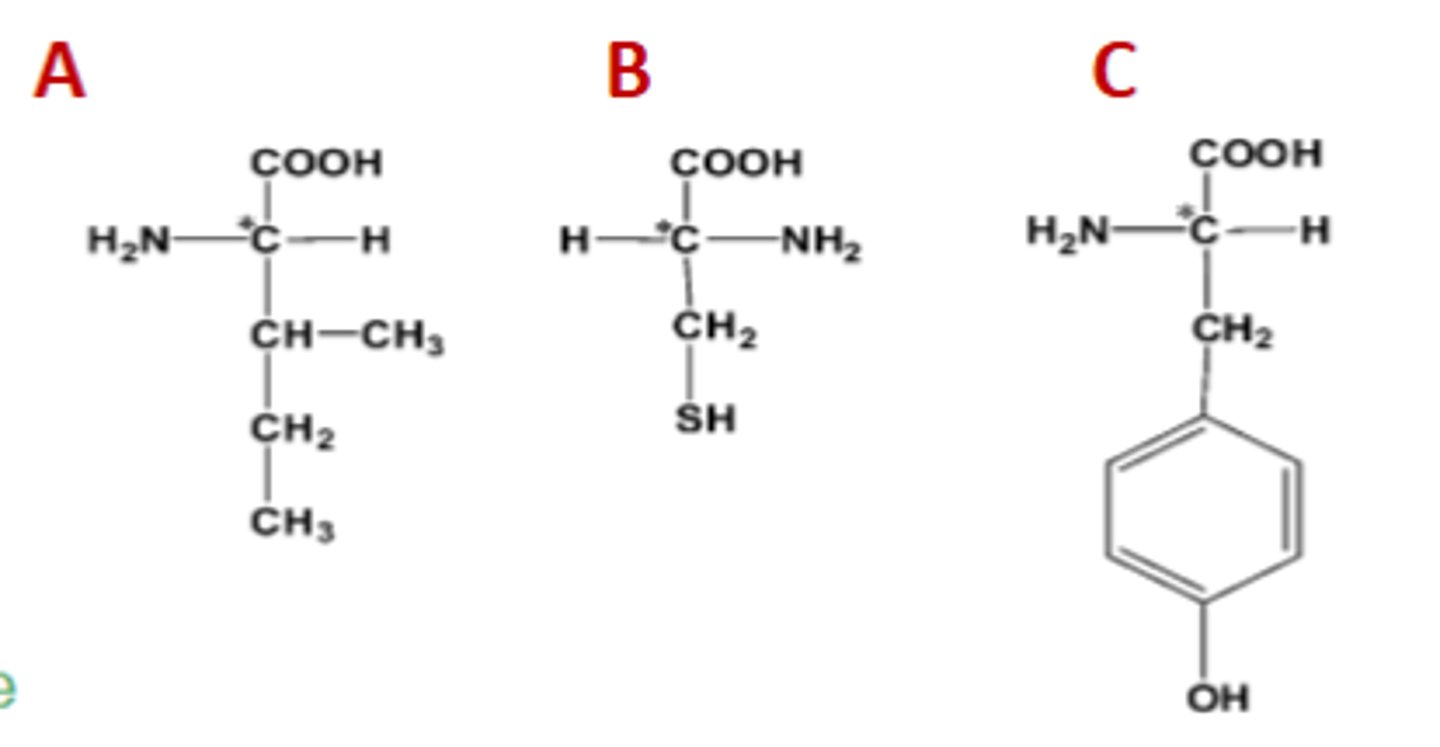
Name the following amino acids with correct designation for the enantiomer (chiral carbon is indicated by *)
A: L-isoleucine; B: D-cysteine; C: L-tyrosine
With few exceptions, the amino acids found in nature and in proteins are ___ isomers
L
3 multiple choice options
An amino acid with a positive charge on one atom
and a negative charge on another atom with an
overall charge of zero is known as a _____.
zwitterion
A _____ protein contains only amino acid residues,
and a _____ protein contains one or more non-amino
acids in the structures.
simple; conjugated
3 multiple choice options
The order in which amino acids are linked in a
protein is known as the _____ structure.
primary
The two most common types of secondary
structures of proteins are the _____ and the _____.
alpha helix; beta pleated sheet
3 multiple choice options
What type of attractive interaction, that contributes
to the tertiary structure of a protein, would be found
buried in a nonaqueous environment within the
protein?
hydrophobic interactions
3 multiple choice options
The structure of hemoglobin, with organization of its
alpha and beta subunits, is an example of what type
of protein structure?
quaternary
3 multiple choice options
Which of the following best describes what happens
to a small peptide when placed in an acid solution
and heated?
The small peptide undergoes hydrolysis to produce free amino acids.
3 multiple choice options
What is the consequence of protein denaturation?
all of the above
3 multiple choice options
What are enzymes?
specialized catalysts of biochemical reactions
2 multiple choice options
What is the difference between a simple enzyme and a
conjugated enzyme?
Simple enzymes are composed only of protein chains while conjugated enzymes have a nonprotein part in addition to a protein part.
A: transferase B. Lyase
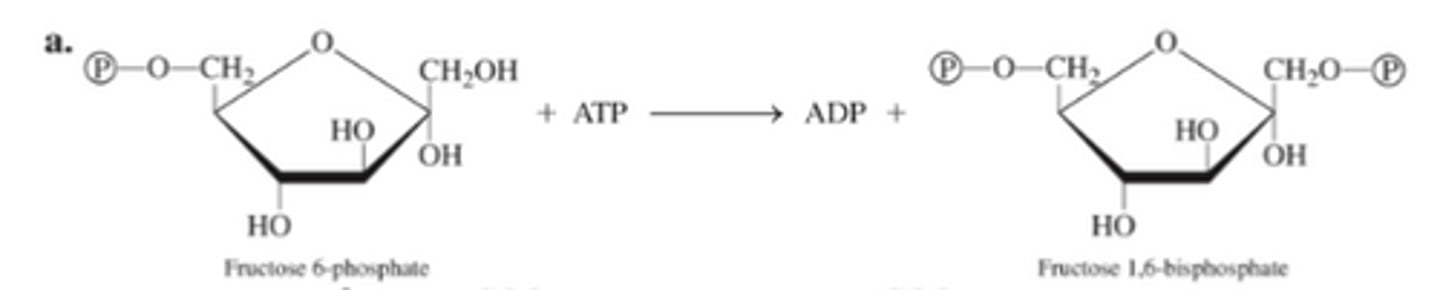
What function does the substrate perform in an
enzyme-catalyzed reaction?
It is the substance upon which the enzyme acts.
3 multiple choice options
What is the area of an enzyme where catalysis of a substrate
occurs called?
active site
Which of the following statements is true about an
enzyme's absolute specificity?
Enzyme will catalyze a particular reaction for only one substrate.
What factors affect enzyme activity?
All of the above
3 multiple choice options
Identify the functions of carbohydrates in the human body.
All the above.
3 multiple choice options
Carbohydrates are classified as:
monosaccharides, disaccharides, oligosaccharides, and polysaccharides.
3 multiple choice options
A- not; B- not; C- chiral; D-not
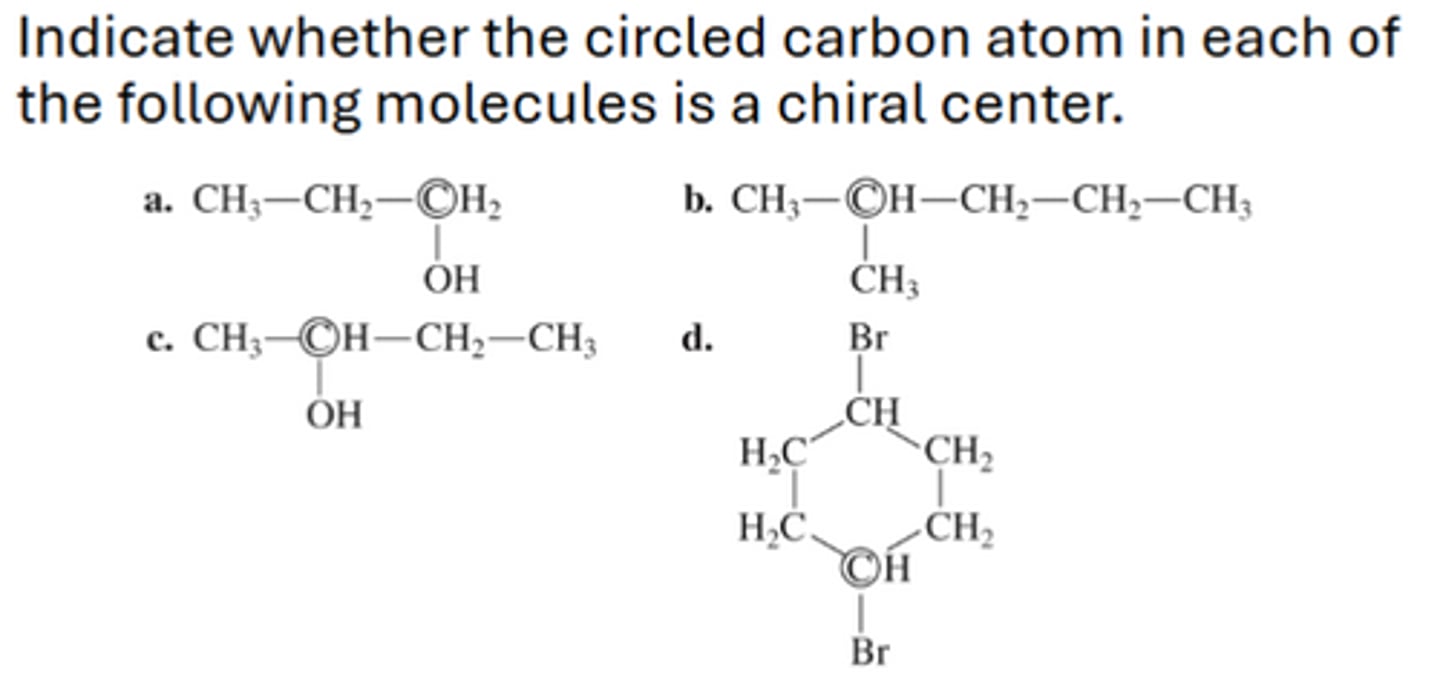
A carbohydrate molecule whose mirror image is not
superimposable is said to be a _____ molecule.
chiral
3 multiple choice options
What feature does a carbohydrate possess that
generates stereoisomerism?
presence of a chiral center
3 multiple choice options
What formula is used to show the two-dimensional structure of groups around chiral centers in a molecule?
Fischer projection
3 multiple choice options
How can you identify the D isomer of a carbohydrate
by looking at the Fischer projection formula?
The -OH group on the chiral carbon farthest from
the carbonyl group is pointing to the right.
3 multiple choice options
What is meant when it is said that carbohydrates are optically active compounds?
A solution of carbohydrate will rotate the plane of
polarized light.
3 multiple choice options
What structural feature distinguishes an aldose and a ketose?
An aldose possesses an aldehyde group, and a
ketose possesses a ketone group.
3 multiple choice options

Which monosaccharide is the most important from a human nutritional standpoint?
D-glucose
Monosaccharides in nature have cyclic forms because they can form:
internal hemiacetals that are stable.
Haworth projection formula for a monosaccharide is a
formula showing:
a two-dimensional notation that specifies the three-
dimensional structure of a cyclic monosaccharide
a. Ribose b. fructose c. galactose, glucose d. Fructose

What is the linkage that is formed when two monosaccharides react to form a disaccharide called?
alcohol group linkage
Which disaccharide is a nonreducing sugar?
sucrose
A _____ is a polysaccharide in which only one type of
monosaccharide monomer is present.
homopolysaccharide