Orgo Lab Quiz + Practicum
1/38
Earn XP
Description and Tags
Some basic chem about Orgo 2 Labs 1-5 and basic experimental set ups
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
39 Terms
Reaction: Secondary alcohol → ketone
Oxidation of Cyclohexanol
Reagent: Sodium hypochlorite (NaOCl, bleach)
Oxidation of Cyclohexanol
Why is the Oxidation of Cyclohexanol green chemistry?
Green chemistry (replaces toxic chromium reagents)
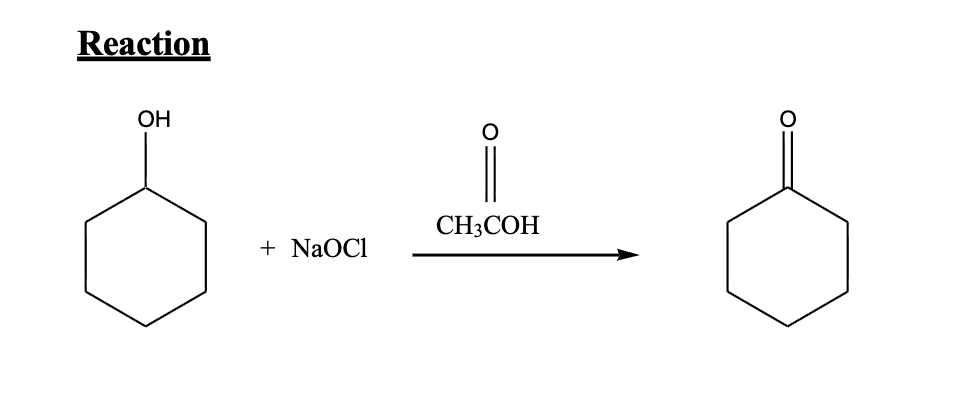
Product: Cyclohexanone
Oxidation of Cyclohexanol
→ If they ask mechanism: involves HOCl formation + oxidation via hypochlorite
Important concept:
Oxidation = increase in C–O bonds / decrease in C–H
Byproducts = water + salt (much safer)
Oxidation of Cyclohexanol
Reaction type: Electrophilic Aromatic Substitution (EAS)
Bromination of Acetanilide (EAS)
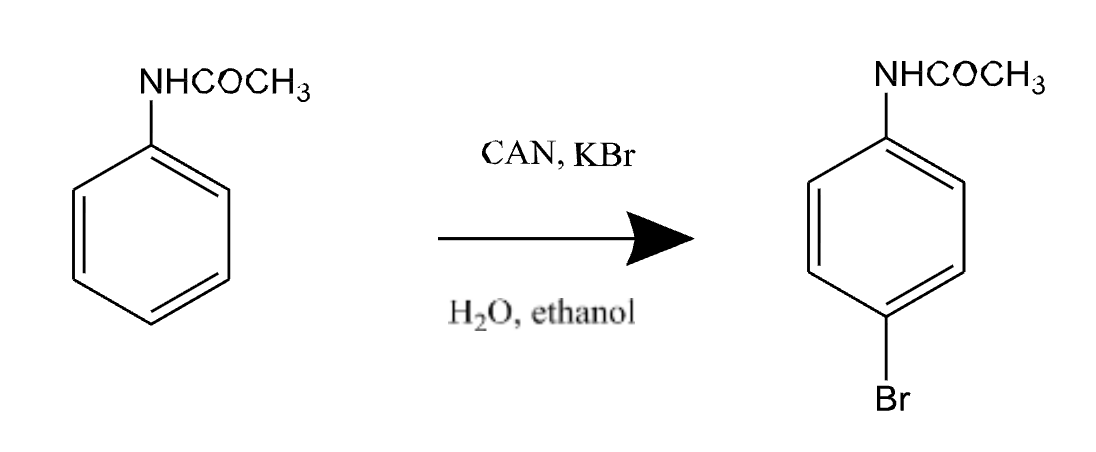
Goal: Add Br to benzene ring
Bromination of Acetanilide (EAS)
Reagents: KBr + CAN (ceric ammonium nitrate) → makes brominating species in situ
Bromination of Acetanilide (EAS)
Why is Bromination of Acetanilide (EAS) green chemistry?
Avoids using Br₂ directly (toxic, dangerous)
Mechanism of Bromination of Acetanilide
Key mechanism steps:
Generate electrophile (Br⁺ or equivalent)
Benzene ring attacks → sigma complex
Deprotonation → restores aromaticity
Is Bromination of Acetanilide ortho or para directing?
Acetanilide is ortho/para directing, but:
Para product dominates (less steric hindrance)
Fluorescence in Fluorescence vs Chemiluminescence lab
1. Fluorescence
Light absorbed → excited state → emits light
Equation:
E=hcλE = \frac{hc}{\lambda}E=λhc
Fluorescein
Emits green light (~525 nm)
pH-dependent structure → changes intensity/color
Chemiluminescence in Chemi and fluorescence Lab
2. Chemiluminescence (Luminol)
Light produced from a chemical reaction, not light absorption
Example:
Luminol + oxidizer → excited product → emits light
Key requirements:
Enough energy from reaction
Pathway to excited state
Light emission or energy transfer
👉 Classic example: forensic blood detection
this lab involved:
NaBH₄ (sodium borohydride)
Possibly comparing with LiAlH₄
Reduction of Camphor
Reductions in Reduction of Camphor
Core Concept:
Reduction of carbonyls → alcohols
Aldehyde → 1° alcohol
Ketone → 2° alcohol
What does NABH4 do In reduction of camphor?
What NaBH₄ does:
Donates hydride (H⁻) → attacks C=O
Then protonation → alcohol
Reagent Strength Differences in reduction of camphor
Reagent | Strength | What it reduces |
|---|---|---|
NaBH₄ | mild | aldehydes + ketones |
LiAlH₄ | strong | esters, carboxylic acids, amides (everything basically) |
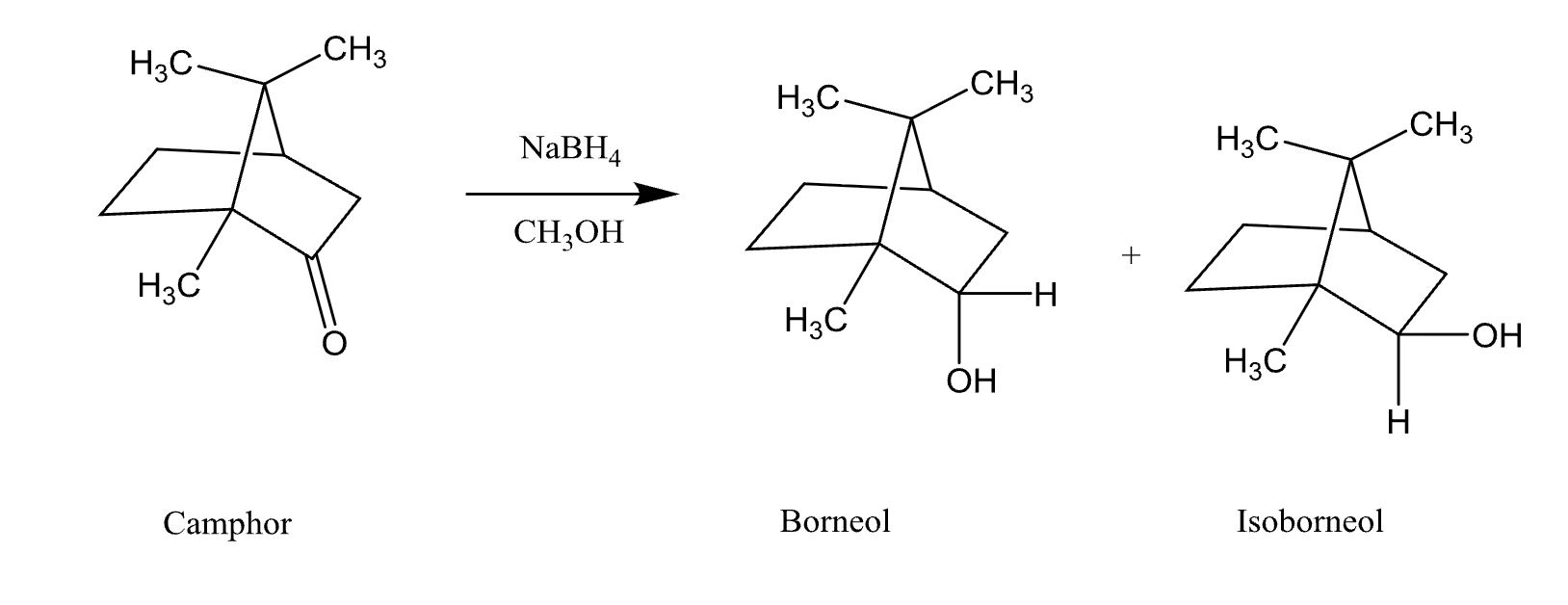
Products in Reduction of Camphor
Borneol and Isoborneal (major product)
Reaction Mechanism of Synthesis of Salicylic Acid
Ester + Base → Carboxylate salt(soap) + Alcohol
Products of Synthesis of Salicylic Acid
Product is carboxylate salt, NOT acid
You need acid workup (H₃O⁺) to get the acid
Why NaOH vs KOH in Synthesis of Salicylic Acid
Both work the same (both strong bases)
Difference is just the cation (Na⁺ vs K⁺)
Q: Difference: extraction vs washing
Extraction = separates compounds
Washing = removes impurities
Organic layer vs aqueous layer—how to tell?
Depends on solvent density (chloroform = bottom, ether = top)
Lab 1 Spam Questions
LAB 1: Oxidation (Cyclohexanol)
Q: What functional group change occurs?
A: Alcohol → ketoneQ: What type of alcohol can be oxidized to ketone?
A: Secondary alcoholQ: Why is NaOCl “green”?
A: Less toxic, produces safer byproducts (water + salt)Q: Oxidation definition (orgo)
A: Increase C–O or decrease C–H bonds
Lab 2 spam Questions
LAB 2: EAS Bromination
Q: What is the electrophile in bromination?
A: Br⁺ (or equivalent electrophilic bromine)Q: What is a sigma complex?
A: Non-aromatic intermediate formed after attackQ: Why is acetanilide ortho/para directing?
A: Electron-donating amide groupQ: Why is para product major?
A: Less steric hindranceQ: Why generate bromine in situ?
A: Safer, avoids toxic Br₂ buildup`
Lab 3 Spam Questions
LAB 3: Fluorescence & Luminol
Q: Fluorescence vs chemiluminescence
A:Fluorescence = light from absorbed energy
Chemiluminescence = light from chemical reaction
Q: What does wavelength relate to?
A: Energy (shorter λ = higher energy)Q: Why does fluorescein depend on pH?
A: Structure changes with protonation → changes emissionQ: What must happen for chemiluminescence?
A: Reaction produces excited-state molecule
Lab 4 Spam Questions
LAB 4: Reduction (NaBH₄)
Q: What does NaBH₄ provide?
A: Hydride (H⁻)Q: Aldehyde → ?
A: 1° alcoholQ: Ketone → ?
A: 2° alcoholQ: Why is NaBH₄ safer than LiAlH₄?
A: Less reactive, works in protic solventsQ: What bond is broken in reduction?
A: C=O π bond
Lab 5 Spam Questions
LAB 5: Saponification
Q: What is saponification?
A: Base hydrolysis of esterQ: Products of saponification
A: Carboxylate salt + alcoholQ: Why is reaction irreversible?
A: Carboxylate is stabilized (won’t reform ester)Q: What step gives carboxylic acid?
A: Acid workup (H₃O⁺)Q: Real-world example
A: Soap formation
Final Overall Spam Questions:
Q: Why does EAS preserve aromaticity?
A: Final step restores conjugated π systemQ: Why is NaBH₄ selective?
A: Only reacts with highly polarized C=OQ: Why do oxidation reactions often use acids?
A: Activate oxidizing agent (like HOCl formation)Q: Why does chemiluminescence often involve oxidation?
A: Releases enough energy to excite electrons
Demonstrate everything you need to know about liquid-liquid extraction!
Vent separatory funnel (VERY tested)
Mix gently to avoid emulsions
Q: What happens if you don’t vent?
A: Pressure buildup → stopper can pop off
Steps:
Add mixture to separatory funnel
Add second solvent
Shake + VENT
Let layers separate
Drain bottom layer
Repeat if needed
⚠ VERY TESTED:
Why vent?
→ Release pressure (prevent explosion)How identify layers?
→ Density (chloroform = bottom)What if emulsion forms?
→ Wait or add saltExtraction vs washing:
Extraction = move compound
Washing = remove impurities
How to Wash An Organic Layer
2. Washing an Organic Layer
Remove impurities (acid, base, salts)
Common washes:
Water → removes polar stuff
NaHCO₃ → removes acids
HCl → removes bases
👉 Key idea:
Washing ≠ extracting (professors test this a lot)
How to dry an organic layer
Use drying agents (CaCl₂, MgSO₄)
Know:
Add until free-flowing powder
Swirl + wait
Steps:
Add drying agent (MgSO₄ or CaCl₂)
Swirl → wait
Look for free-flowing powder
Remove drying agent by:
Gravity filtration OR
Decanting
⚠ Must-know:
What does drying agent remove?
→ WaterHow do you know it's done?
→ No clumpingBIG TRICK:
Drying agent is a solid impurity → must be removed before evaporation
How to do gravity filtration
Remove solid impurities
Used when product is in solution
👉 NOT for collecting crystals
How to perform Vacuum (Büchner) Filtration
What you should know how to do:
Set up: Büchner funnel + filter paper + vacuum flask
Turn on vacuum → wet filter paper → pour mixture
Wash crystals with cold solvent
Let air pull through to dry
High-yield questions:
Why use vacuum filtration?
→ To collect solid quicklyWhy use cold solvent?
→ Prevent dissolving your productWhat happens if vacuum isn’t tight?
→ Poor suction → slow filtration
How to do Recrystallization
Purify solid compounds
Steps:
Dissolve in hot solvent
Cool slowly → crystals form
Filter + dry
👉 Key concept:
Solvent must dissolve compound hot, not cold
How to measure melting point
Determines:
Purity (sharp vs broad range)
Identity
👉 Flashcard:
Impure sample → lower + broader melting point
HNMR sample preparation
Steps:
Dissolve small amount of sample
Use deuterated solvent (e.g., CDCl₃)
Transfer to NMR tube (clean + clear)
Fill to proper height (~5 cm)
⚠ High-yield:
Why deuterated solvent?
→ Doesn’t show up in ¹H NMRWhy small amount?
→ Too concentrated = messy peaksWhat happens if dirty tube?
→ Contaminated spectrum
Reflux Set Up
✅ What’s happening:
Heating reaction without losing solvent
✅ Setup:
Round-bottom flask
Condenser on top
Water flowing (in bottom → out top)
Heat source below
⚠ MUST KNOW:
Why reflux?
→ Speeds reaction while preventing evaporationWhy water flows bottom → top?
→ Keeps condenser fullWhat happens without condenser?
→ Solvent evaporates → reaction fails
Each Technique Final Practice
For each technique, be ready to answer:
What is the purpose?
What could go wrong?
Why is each step necessary?