Equilibrium
1/13
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
14 Terms
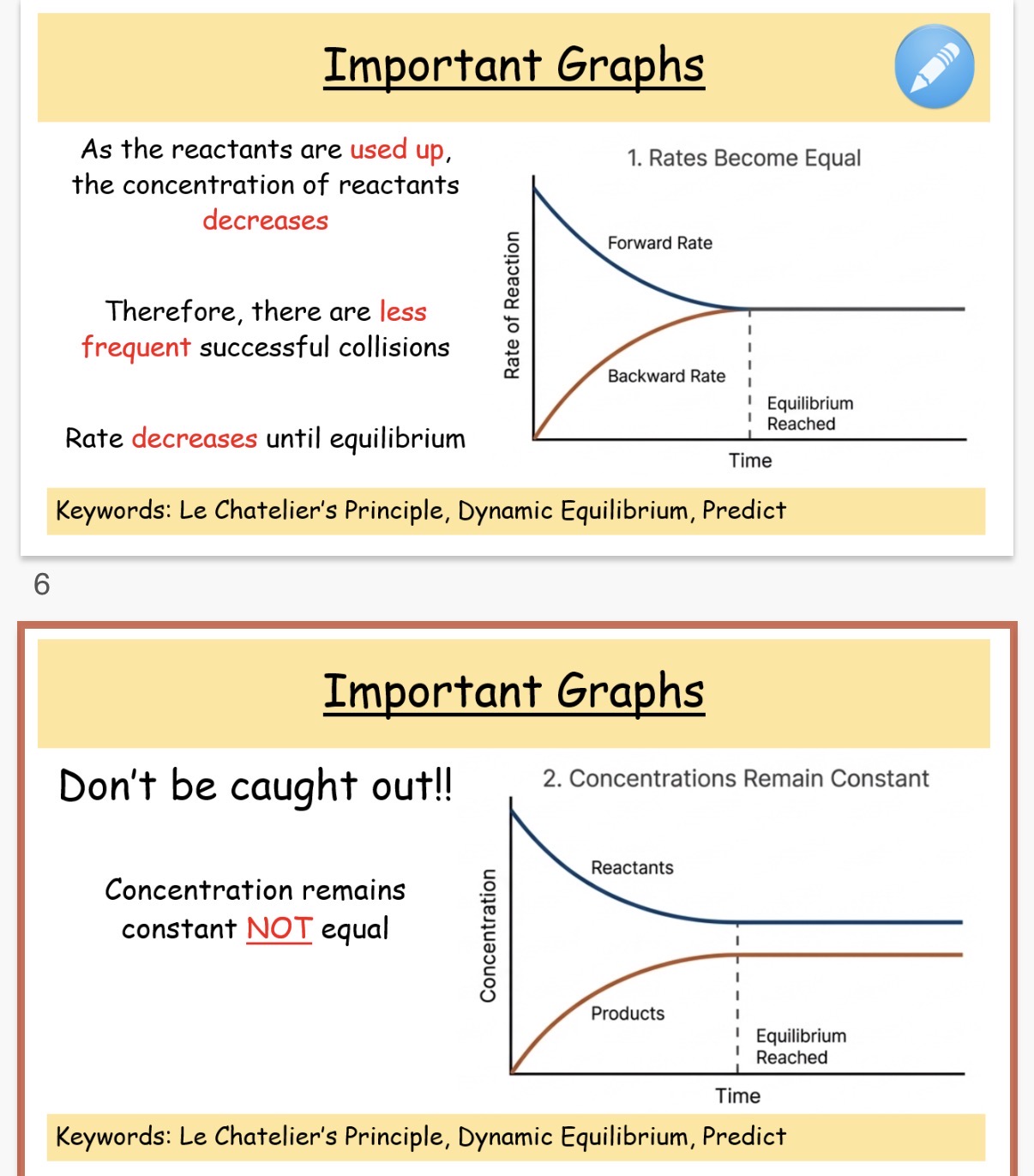
What is dynamic equilibrium?
Which statement is not always correct for a reaction at equilibrium?
A: The concentrations of the reactants and products are equal.
B: The equilibrium can be achieved starting from the reactants.
C: The equilibrium can be achieved starting from the products.
D: The rate of the forward reaction is equal to the rate of the reverse reaction.
What is Le chatelier’s principle?
Note- this shift will either be to the left(backwards reaction) or to the right(forward reaction), depending on which helps to oppose the applied change.
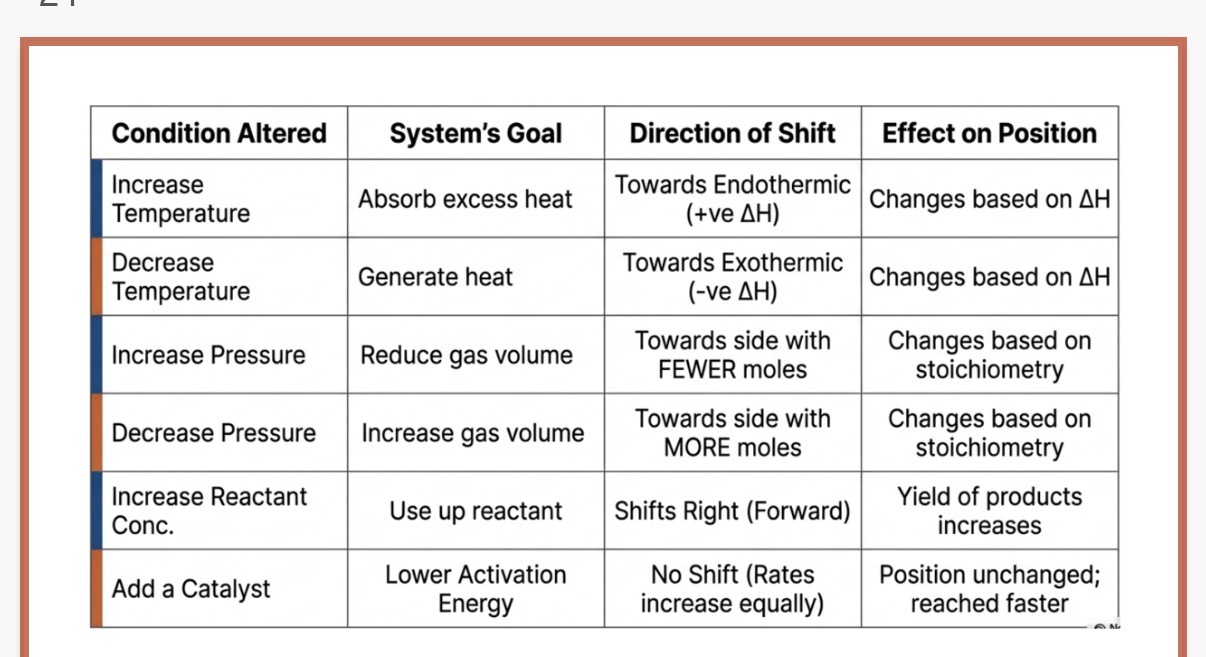
Change in Concentration
•When you add or remove a reactant or product, the system tries to restore the ratio.
What happens when you add reactants?
What happens when you add product?
what happens when you remove product?
Change in Pressure (Gas Systems Only)
•Pressure only affects equilibrium if there are gases involved and a different number of moles on each side of the equation.
What happens when the pressure increases in a system?
What happened when the pressure decreases?
Change in Temperature
•To predict a temperature shift, you must know if the reaction is exothermic (releases heat) or endothermic (absorbs heat). Treat "heat" as a physical reactant or product.
The forward reaction is endothermic. What happens to the position of equilibrium when temperature increases?
What happens when the temperature decreases?
What happens to the position of equilibrium if the temperature increases and the forward reaction is exothermic?
The Role of a Catalyst
Do catalysts affect the position of equilibrium?
A catalyst …13? the activation energy for both the forward and reverse reactions equally. This means the system reaches equilibrium faster, but the actual concentrations of reactants and products at equilibrium remain exactly the same.
Where the rate of the forward and backward reaction are equal in a closed system.
A
When a system is subjected to a change in temperature, pressure or concentration, the position of equilibrium will shift to counteract(oppose) that change. (So dynamic equilibrium is maintained)
The position of equilibrium shifts to the right (toward products) to consume the extra reactant.
The position of equilibrium shifts to the left (toward reactants) to consume the extra product.
The position of equilibrium shifts to the right to replace what was lost (this is often used in industry to maximize yield).
The system shifts toward the side with fewer moles of gas to reduce the pressure.
The system shifts toward the side with more moles of gas to increase the pressure.
The position of equilibrium shifts to the right (increase in temp always favours the endothermic direction)
Equilibrium shifts to the left(temp decreases favours the exothermic direction)
Equilibrium shifts to the left
No. 13. Lowers
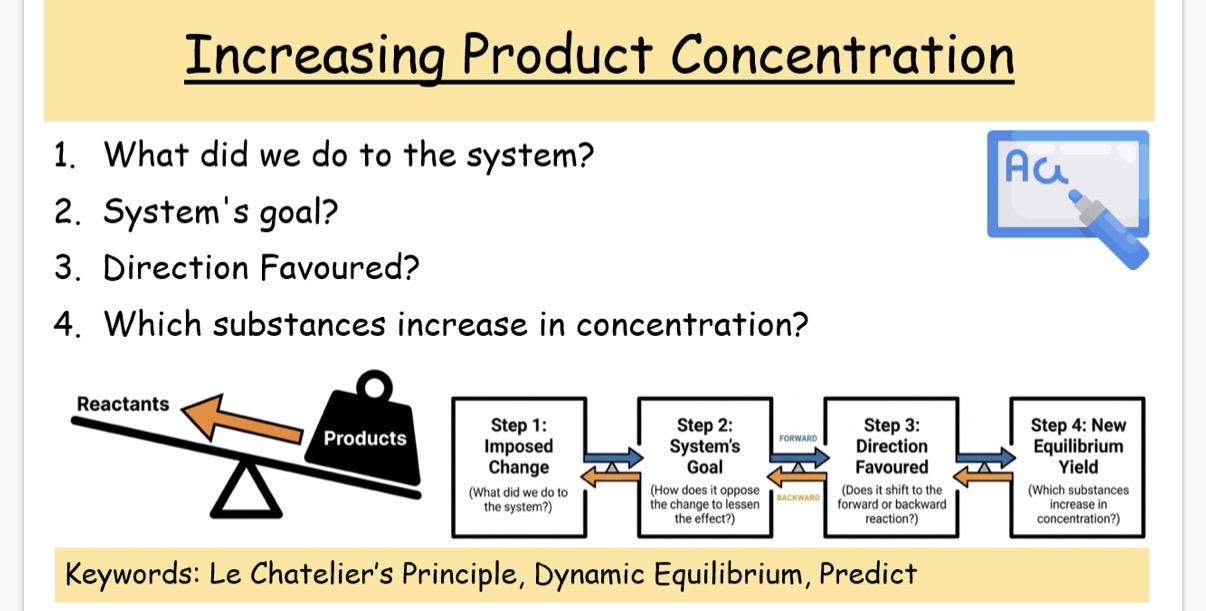
Answer image question?
(1) increase the concentration of products
(2) Reduce the concentration of products
(3)Favours the backward reaction so equilibrium shifts to the left
(4) Reactants concentration increases

Answer image q?


Answer image q?
Explain why using a catalyst has no effect on the percentage yield?
2.A catalyst increases the rate of both the forward and the reverse reactions by the same amount (by providing an alternative pathway with a lower activation energy). Because it speeds up both directions equally, the position of equilibrium does not change, and therefore the percentage yield remains the same.

![<p><strong>Kc equilibrium</strong></p><p>•The exact position of equilibrium can be calculated using the expression in the image(must always be in square brackets):</p><ol><li><p>What is the formula to calculate concentration?</p></li><li><p>What is the units for volume and concentration</p></li><li><p>Write the Kc expression for</p></li></ol><p>2NO(g) + O2(g) → 2NO2 (g)</p><p>•Kc is in the form [products]/[reactants] this means that:</p><p>•If Kc is 1 the equilibrium position is halfway between the products and reactants (equal)</p><ol start="4"><li><p>If Kc is above 1, the equilibrium position shifts to the …4?and so there is a greater concentration of …5?</p><ol start="6"><li><p>what if the Kc value is less than 1?</p><p>info• in Kc you don’t include the molecules/elements that aren’t gases, in the calculations.</p></li></ol></li></ol><p>•The only condition that affects the value of Kc is …7?. If temperature change shifts position of equilibrium to the right then the value of Kc will …8?. For all other conditions seen the positions of equilibrium may change but the value of Kc remains …9?.</p>](https://assets.knowt.com/user-attachments/a09b9232-ef01-4fdf-9372-18c88064ad08.heic)
Kc equilibrium
•The exact position of equilibrium can be calculated using the expression in the image(must always be in square brackets):
What is the formula to calculate concentration?
What is the units for volume and concentration
Write the Kc expression for
2NO(g) + O2(g) → 2NO2 (g)
•Kc is in the form [products]/[reactants] this means that:
•If Kc is 1 the equilibrium position is halfway between the products and reactants (equal)
If Kc is above 1, the equilibrium position shifts to the …4?and so there is a greater concentration of …5?
what if the Kc value is less than 1?
info• in Kc you don’t include the molecules/elements that aren’t gases, in the calculations.
•The only condition that affects the value of Kc is …7?. If temperature change shifts position of equilibrium to the right then the value of Kc will …8?. For all other conditions seen the positions of equilibrium may change but the value of Kc remains …9?.
C= mol/volume
V(Dm*3) C(mol dm*-3)
right 5. Products
the equilibrium position shifts to the left(reactants side) and so there is a greater concentration of reactants.
Temperature 8. Increase. 9.constant


Answer the 4 questions in the image?
Info: For question 2 we know that the units for concentration is mol dm-3. Since the product [NO2] is squared, we have to square the units hence why there is 2 Mol dm-3 units on the numerator. [N2O4] has no indices so there is only unit on the denominator. When we cancel the units out, we get Mol dm-3/ 1. Which can be simplified to Mol dm-3.
Info: The second one is incorrect. it should be 1/ (mol dm-3) (mol dm-3) once the units have been cancelled out. Then to get the units onto the numerator, you need to inverse the indices so u get (mol-1 dm3) (mol-1 dm3) / 1. You can then multiply the units indices to get your answer: Mol-2 dm6
![<p>Info: For question 2 we know that the units for concentration is mol dm-3. Since the product [NO2] is squared, we have to square the units hence why there is 2 Mol dm-3 units on the numerator. [N2O4] has no indices so there is only unit on the denominator. When we cancel the units out, we get Mol dm-3/ 1. Which can be simplified to Mol dm-3.</p><p></p><p>Info: The second one is incorrect. it should be 1/ (mol dm-3) (mol dm-3) once the units have been cancelled out. Then to get the units onto the numerator, you need to inverse the indices so u get (mol-1 dm3) (mol-1 dm3) / 1. You can then multiply the units indices to get your answer: Mol-2 dm6</p>](https://assets.knowt.com/user-attachments/68b6c5b8-c244-4303-85af-98d0396bd668.jpg)

ANSWER INAGE Q?
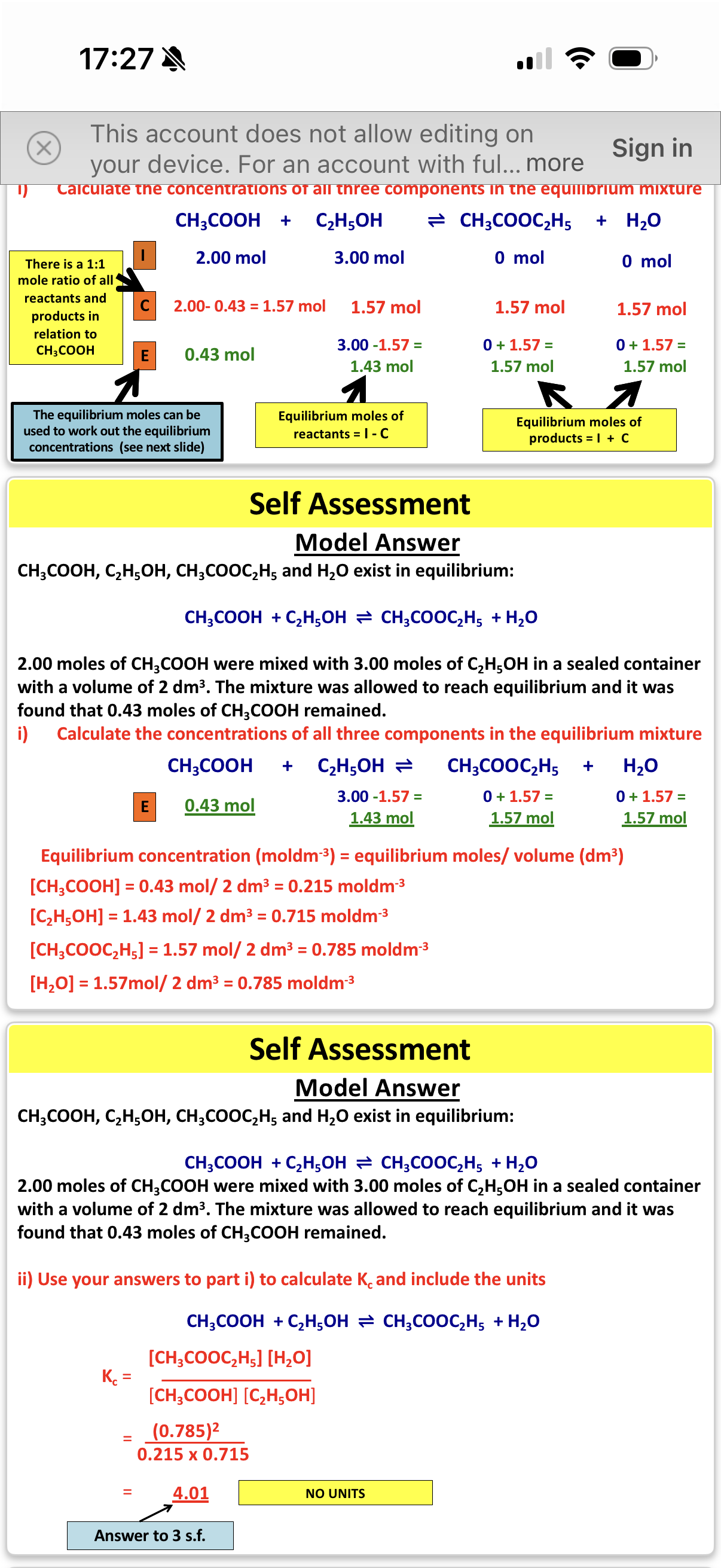
•Calcukating Kc with equilibrium moles being provided


• Calcukating Kc using the initial moles of reactants and products.
Info: This will involve us using the ICE method where:
I = Initial Moles (moles of reactants and products at the start of the reaction)
C = Change in moles (this is linked to stoichiometry of the reaction)
E = Equilibrium moles (moles of reactants and products once equilibrium has been reached)
NOTE:If the intial moles of the product isn’t give in the question, we always assume it’s 0
NOTE: The change in moles will be the same for all reactants. Additionally, all products will have the same change in moles as other products(however The reactants and products won’t have the same change)
NOTE: If the change in moles for the reactants are negative, than the change in moles for the products will always be the opposite (so positive in this case) and vice versa.
NOTE: The change in moles is the same for all reactants however, if one of the reactants have 1mole and the other has 2 moles, the change I moles for the reactant with 2 moles will be doubled.
NOTE: You can use the molar ratio to find the change in moles for the products if u found the change for the reactants.(however you must remember that the sign of the change will be inverses for the products and the value of the change may multiplied or divided depending on the moles of the products.
Answer image q?


Answer image q?
note- The c in Kc stands for concentration, hence why our units are always in mol dm-3
Info: you need to divide the al the moles by 2 to get the concentration. This is because the volume present is 2dm*3

Info:

Answer image q?
Note: When the total moles of reactants = the total moles of products, the volume of each chemical species can cancel out. Therefore, there is no need to calculate the concentration.


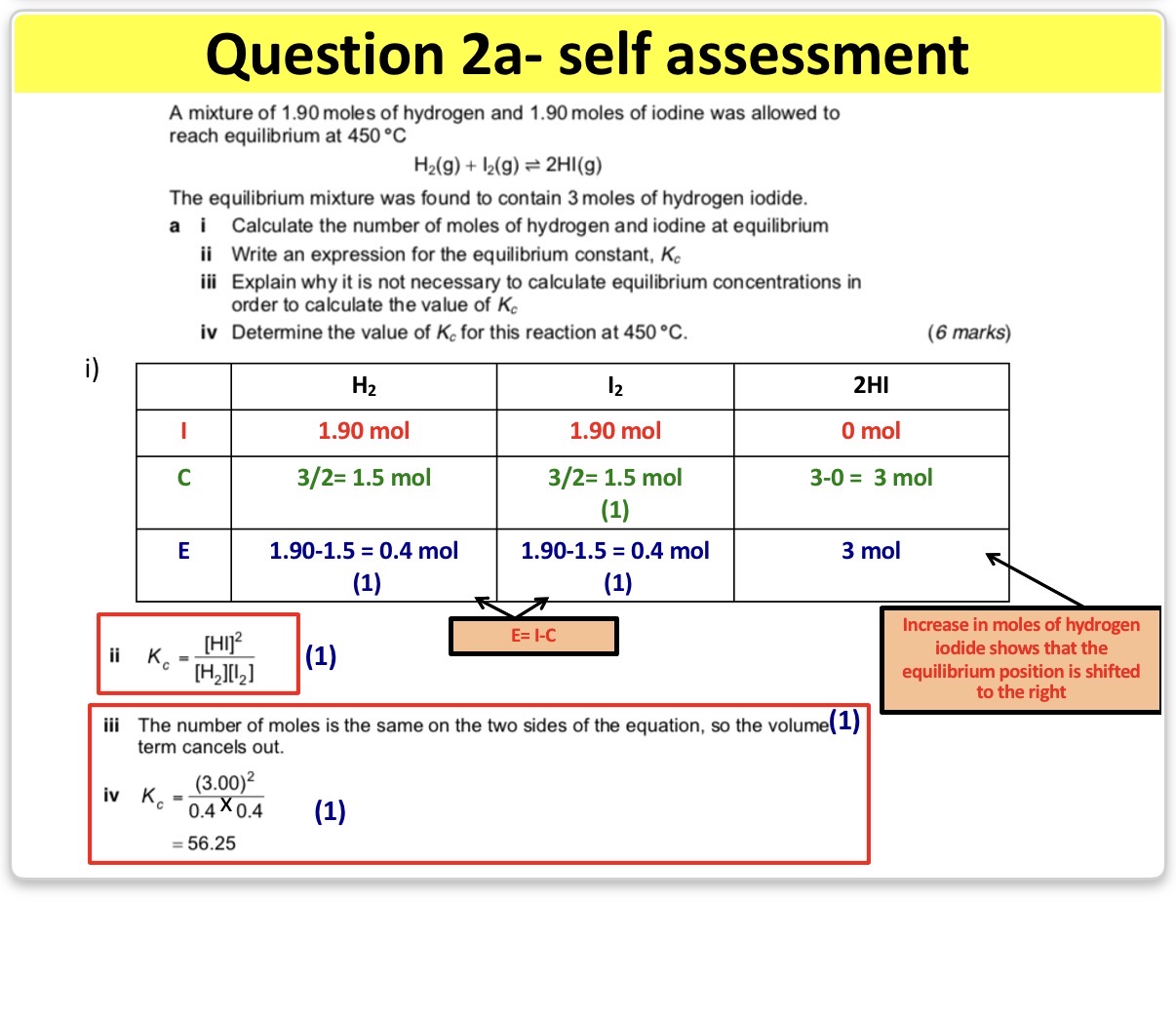

Answer image q?


Answer image q?
