Reactions of alcohols
1/4
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
5 Terms
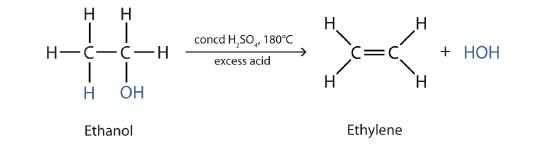
Write the equation for dehydration of alcohols, and what are the conditions needed
Concentrated phosphoric acid and heated under reflux
What type of reaction is reflux
It is an elimination reaction
What are the conditions needed for substitution of alcohols
A halide salt and sulfuric acid e.g.
NaBr(s) + H2SO4(aq) → HBr +NaHSO4
Note:
HBr is what reacts with the alcohol to form the haloalkane and water
What is the overall equation for substitution of an alcohol (butan-2-ol)
NaBr(s) + H2SO4(aq) + CH3CHOHCH2CH3 → CH3CH2CHBrCH3 + H2O + NaHSO4
How would you purify the halo alkane formed
If there is organic layers and aqueous layers you would separate them using a separating funnel. You would then add and anhydrous salt to organic layer. Then distil at its boiling point and collect