GEOL 238 Module 3 & 4 Flashcards
1/171
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
172 Terms
Origin of carbonate sediments
Carbonate sediments are born, not made
Majority of carbonate sediments are precipitates from organisms that produce skeletons and shells
Two types of carbonate sediment factories
Pelagic factory
Benthic factory
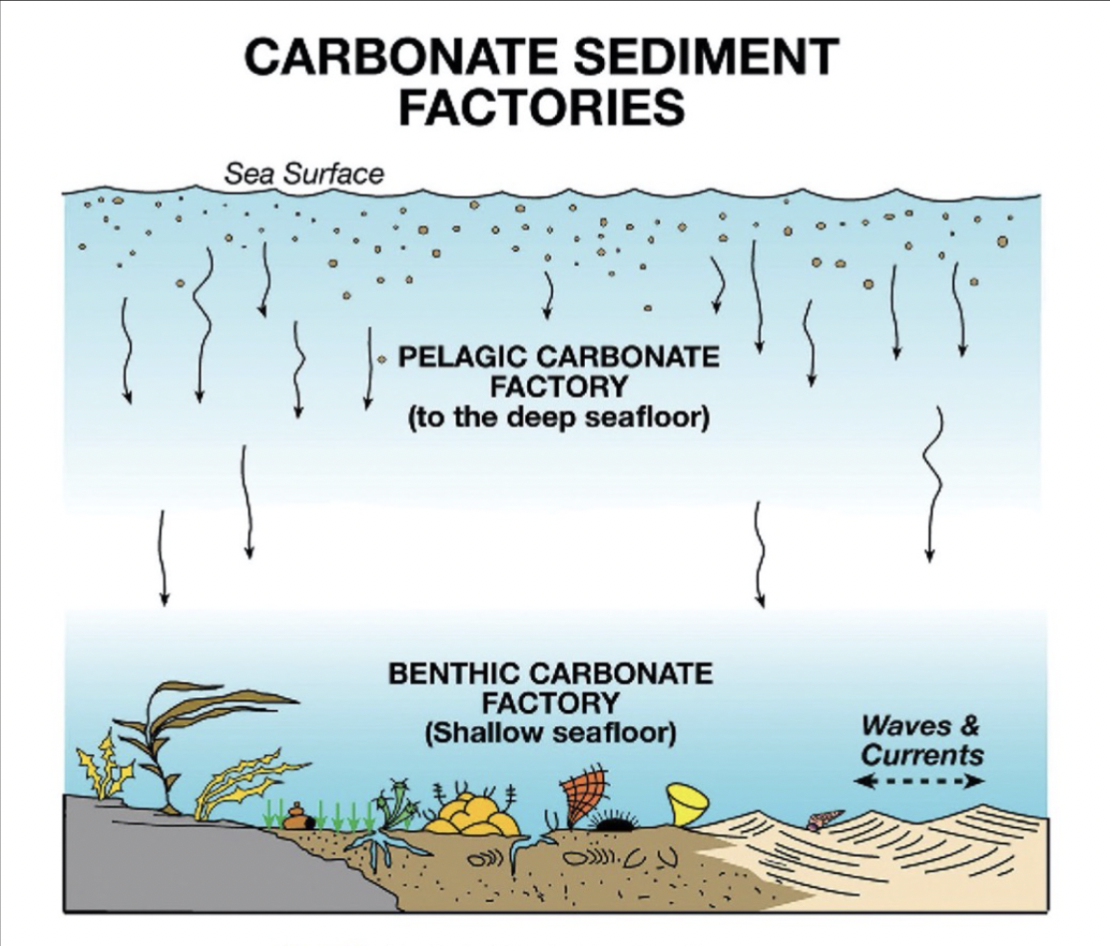
Pelagic factory
The surface ocean; microscopic carbonate producers that fall to seafloor when they die
Benthic factory
The shallow seafloor
4 optimal conditions for a carbonate factory
Shallow
Warm
Nutrient rich
Sunlit
Allochems
Silt to gravel-sized carbonate grains in carbonate rocks
4 types of allochems
Skeletal Particles (can be benthic or planktic)
Coated grains (Ooids, Pisoids, oncolites)
Peloids
Intraclasts
Skeletal fragments
Derived from either:
Benthic organisms (live on seafloor) → ex. gastropod, mollusc, brachiopod, bryozoan, crinoid, coral, green algae
Planktic organisms (live in water column) → ex. gastropod, foraminifera
Peloids
Grains (usually fecal pellets) composed of microcrystalline calcite or aragonite
Smaller than ooids and have no internal structure

Ooids
Coated carbonate grains with a nucleus
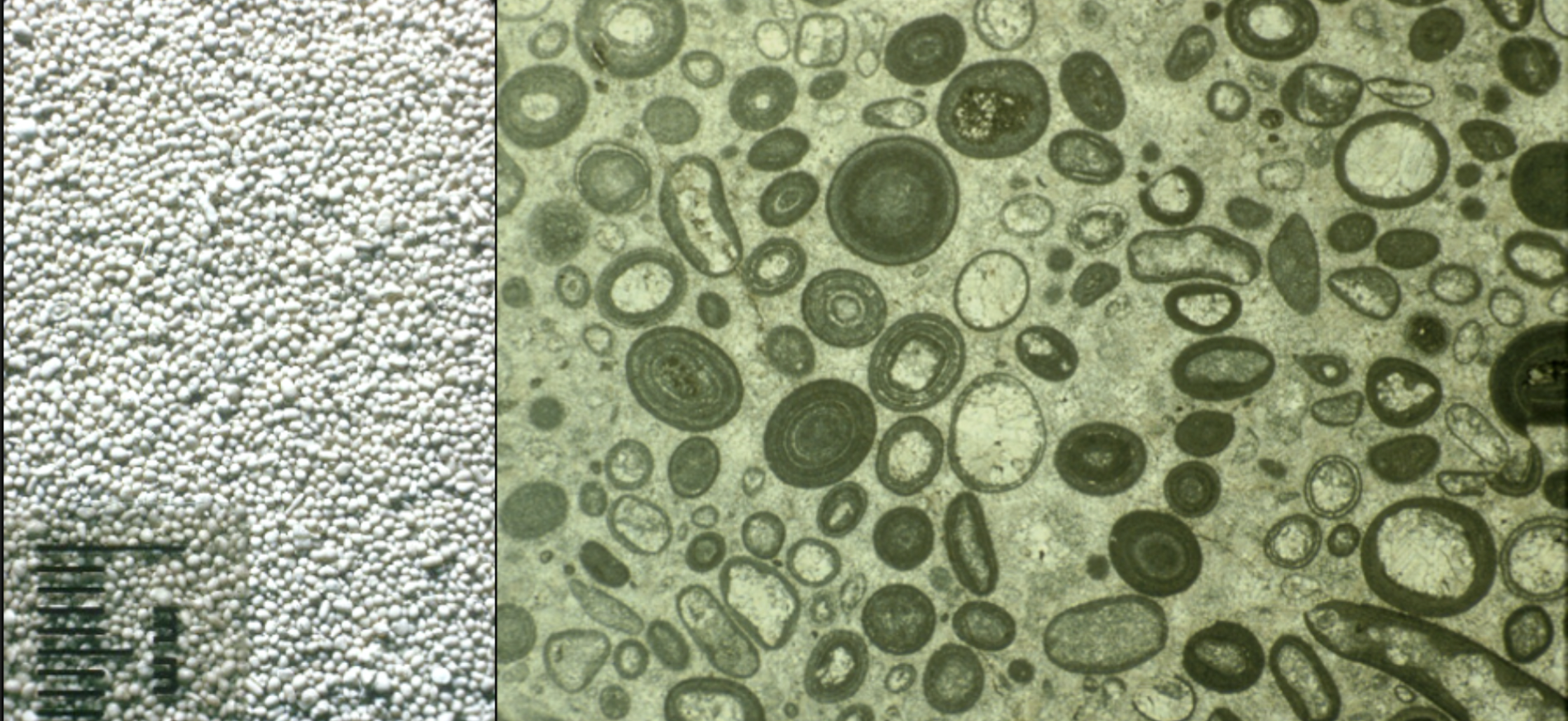
Pisoids
Similar to ooids; but are larger than 2mm and usually more irregular
They may have an algal origin

Intraclasts
Fragments of lithified or partly lithified sediment that was transported only a short distance
Extraclasts
Fragments consisting of lithology not represented in the immediate depositional environment
Carbonate mud
Can form by direct precipitation or by breakdown of skeletal components (e.g. some green algae)

Stromatolites
Formed of microbial mats (e.g. cyanobacteria). The sediment gets trapped in fine filaments in mats, microbes grow up and around sediment to form new mat
Algal laminations are crinkly
Responsible for oxygenating our atmosphere!

Oncoids
Coated irregular layers bound by Cyanobacteria (microbial origin) formed in energetic subtidal environments
Develop in a more energetic environment than stromatolites
2 methods of Dunham classification
Classification based on relative amounts of mud and allochems
Classification based on how organisms bind the rock
Diagenetic change
Chemical processes that occur post deposition
Why are limestones vulnerable to diagenetic change?
Because skeletal fragments are in equilibrium with seawater, but not with freshwater or groundwater
After burial, may start dissolving allochems
and precipitating cement
3 regions of the peritidal environment
Supratidal
Intertidal
Subtidal
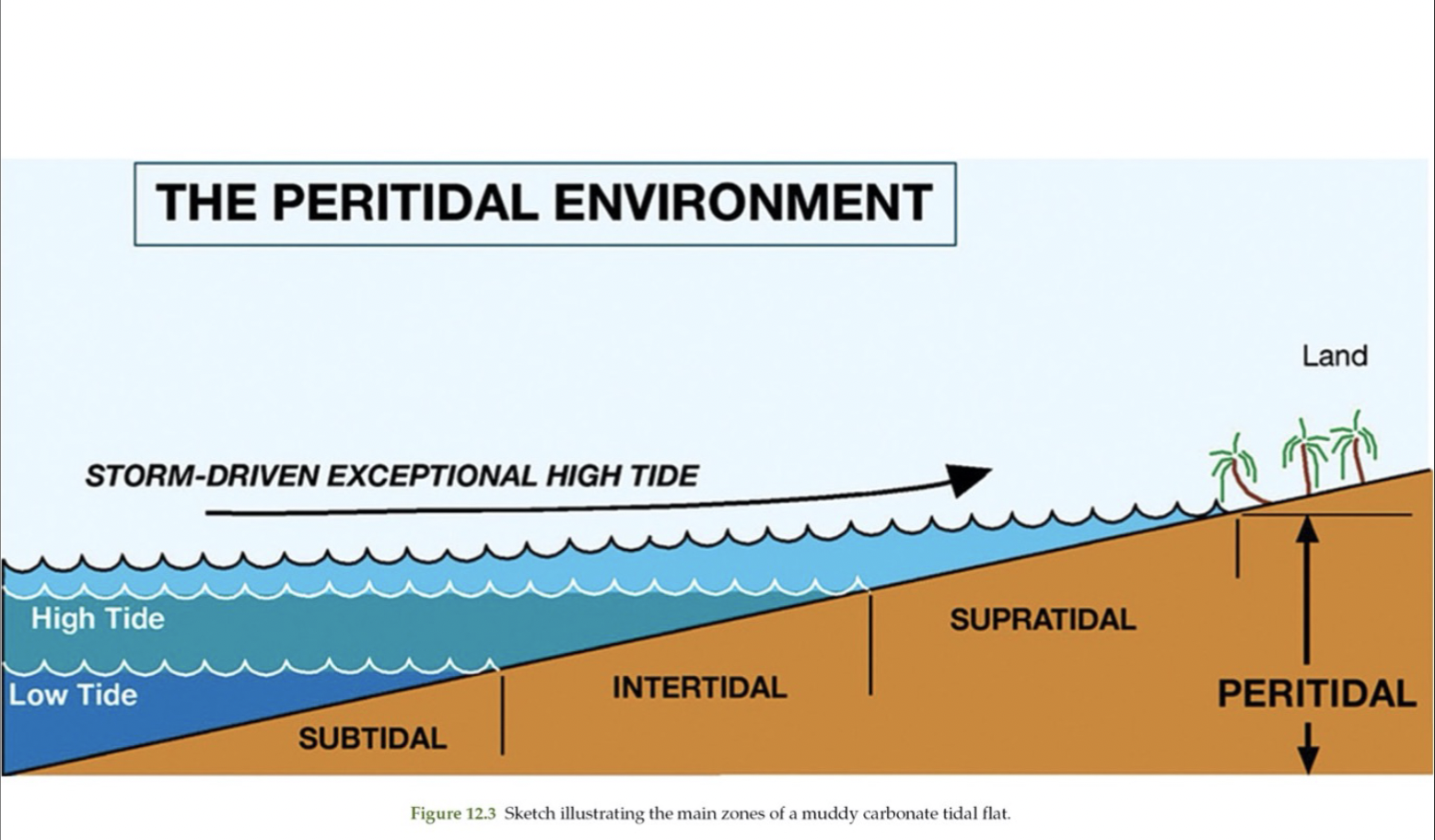
Supratidal
More equivalent to backshore. Flooded during highest tides only
Intertidal
Exposed and flooded during tidal cycles
Subtidal
Below low tide
3 types of supratidal environments
Humid ‘algal marsh’
Well drained and elevated
Sabkha (arid)
Sabkha
An arid supratidal environment with intense evaporation
Supratidal (only flooded during extremely high tides)
Evaporation is so intense it pumps ground water through to continuously precipitate halite

2 types of intertidal environments
Low energy
High energy
4 types of subtidal environments
Lagoonal muds
Lime muds and sands
Lime sand shoals
Stromatolites
Reefs
2 features of humid supratidal environments
Mangrove swamps and algal marshes
Intertidal mud flats
2 features of tidal channels + tidal flats
Algal mats
Mangroves growing on channel levees
Desiccation cracks (even in humid settings)
Cracking of microbial mats

Location of carbonate sand beaches
At the front of the tidal flat in Bahamian tidal flat depositional environments
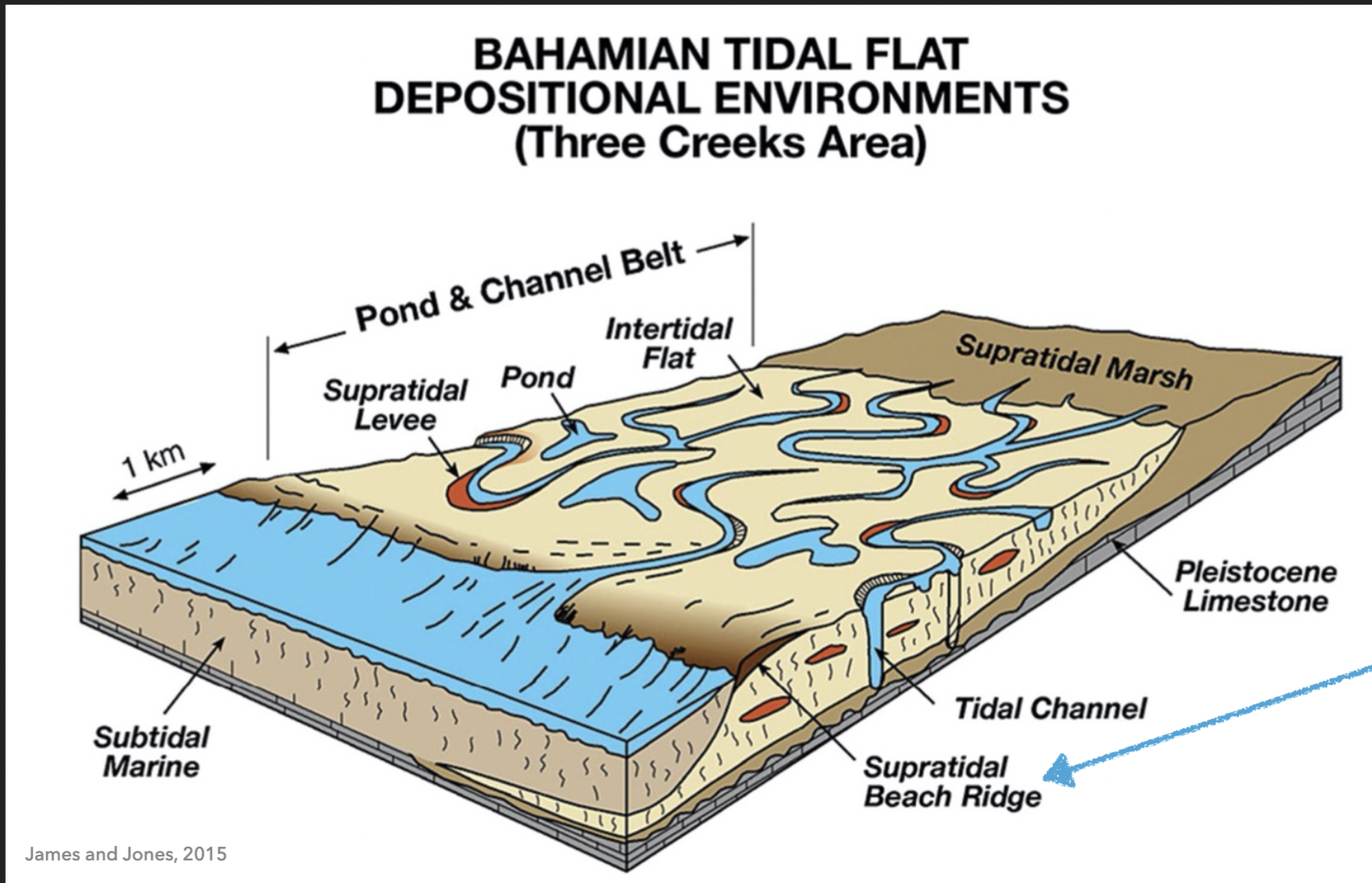
3 features of hypersaline subtidal environments
Stromatolitic reef
Flooded tidal flat
Low stromatolite domes and microbial mats

4 features of semi-arid intertidal environments
Microbial mats
Ponding seawater
Stressed stromatolites
Oolitic sand with wave ripples

Evaporite formation in semi-arid intertidal environments
Evaporation is just enough to precipitate gypsum (CaSO4 . 2H2O) within sediment
4 stages of evaporation of seawater
50% of seawater evaporated → Calcite (CaCo3)
80% of seawater evaporated → Gypsum (CaSO4 . 2H2O)
90% of seawater evaporated → Halite (NaCl)
95% of seawater evaporated → K + Mg salts
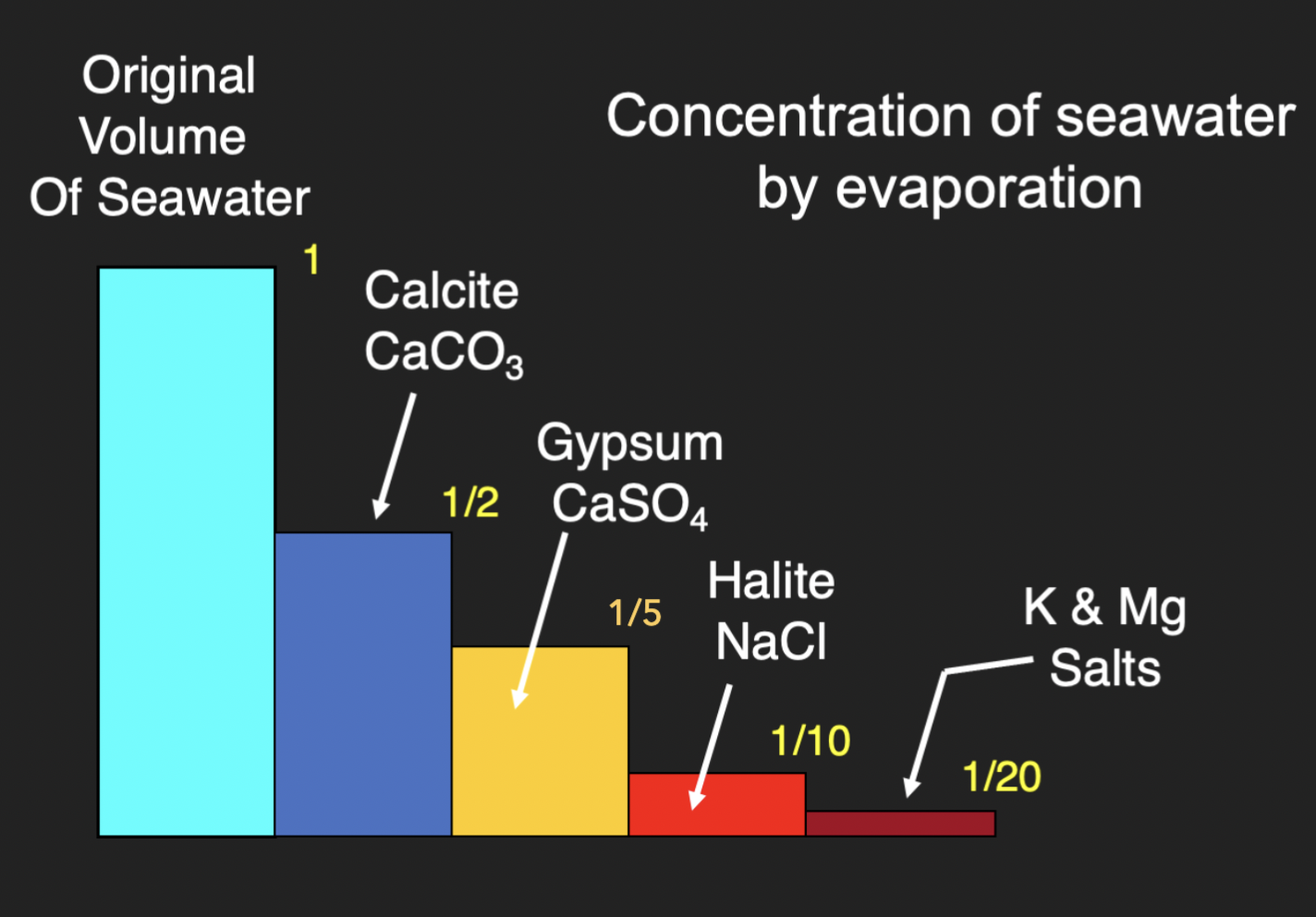
Where do evaporites form?
Arid peritidal systems
4 features of arid intertidal systems
Similar to tidal channels
No mangroves
Expansive microbial mats along channel margins
Gypsum and anhydrite
3 types of modern muddy peritidal cycles
Humid; normal marine salinity (Bahamas)
Semi-arid; hypersaline (western Australia)
Arid; normal marine salinity (Persian Gulf)
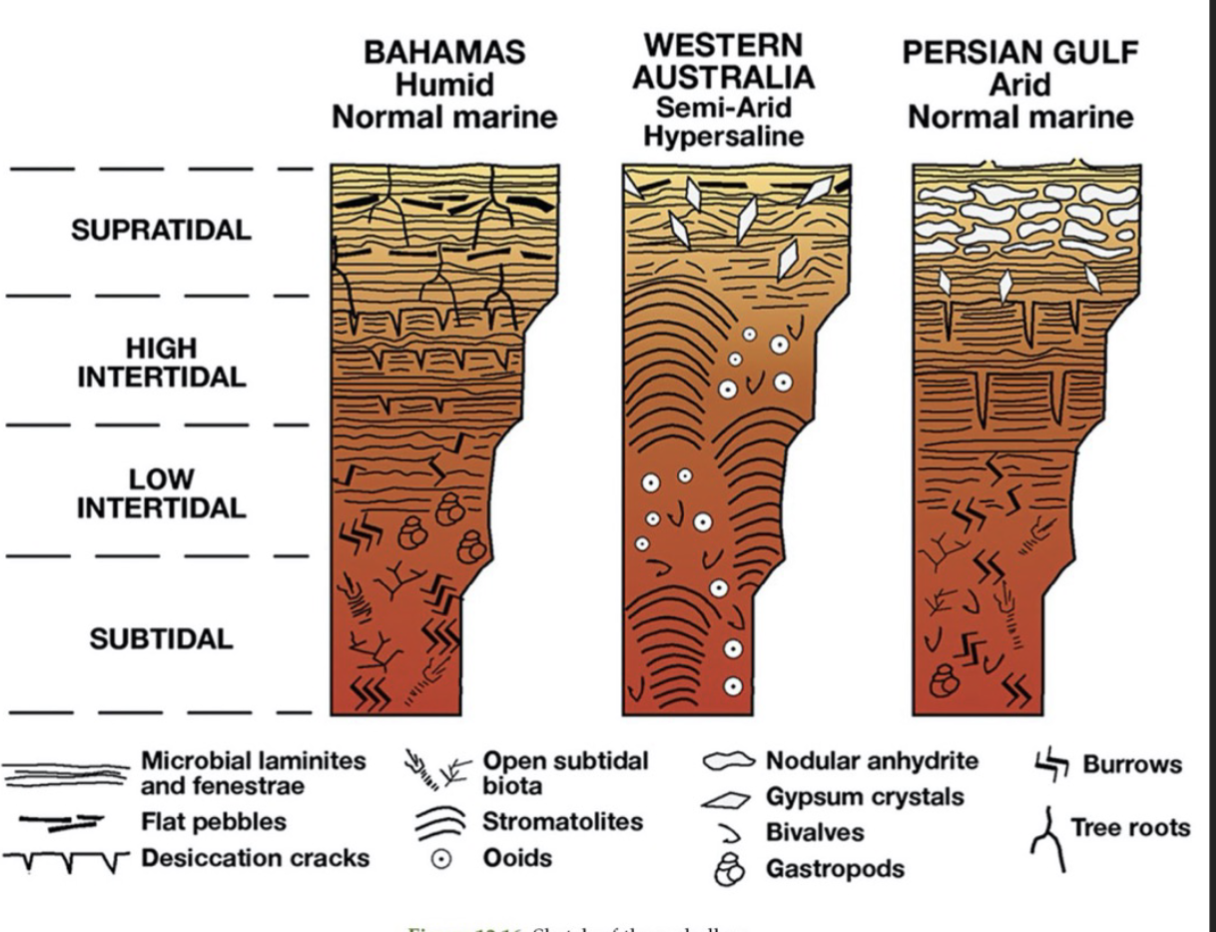
Humid; normal marine salinity (Bahamas) peritidal facies model
BOTTOM TO TOP
Subtidal: bioturbated, muddy
Low intertidal: microbial laminites and fenestrae, burrows
High intertidal: microbial laminites and fenestrae, dessication cracks
Supratidal: flat pebbles, microbial laminites and fenestrae, tree roots
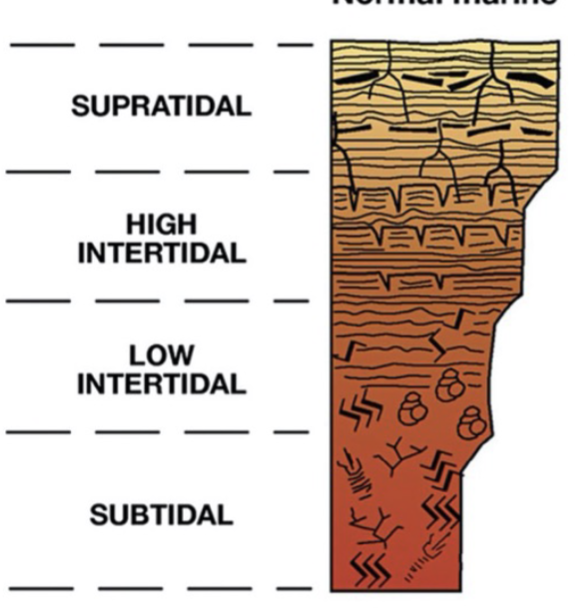
Overall trend of peritidal cycles
Shallowing and coarsening upwards
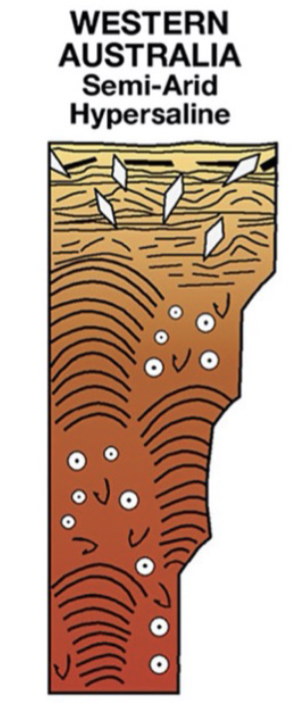
Semi-arid; hypersaline (western Australia) peritidal facies model
BOTTOM TO TOP
Subtidal: stromatolites, bivalves, ooids
Low intertidal: stromatolites, bivalves, ooids
High intertidal: stromatolites, bivalves, ooids
Supratidal: microbial laminites and fenestrae, flat pebbles, gypsum crystals
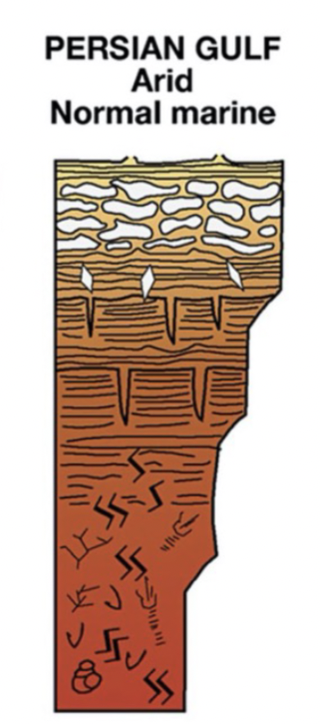
Arid; normal marine salinity (Persian Gulf) peritidal facies model
BOTTOM TO TOP
Subtidal: bioturbated, muddy
Low intertidal: microbial laminites and fenestrae, burrows
High intertidal: microbial laminites and fenestrae, deep desiccation cracks
Supratidal: microbial laminites and fenestrae, anhydrite nodules, gypsum crystals
3 changes to peritidal cycles over time
Appearance of bioclasts (Early Paleozoic)
Decline of stromatolites
Evolution of angiosperms
Evolution of deep-burrowing crustaceans
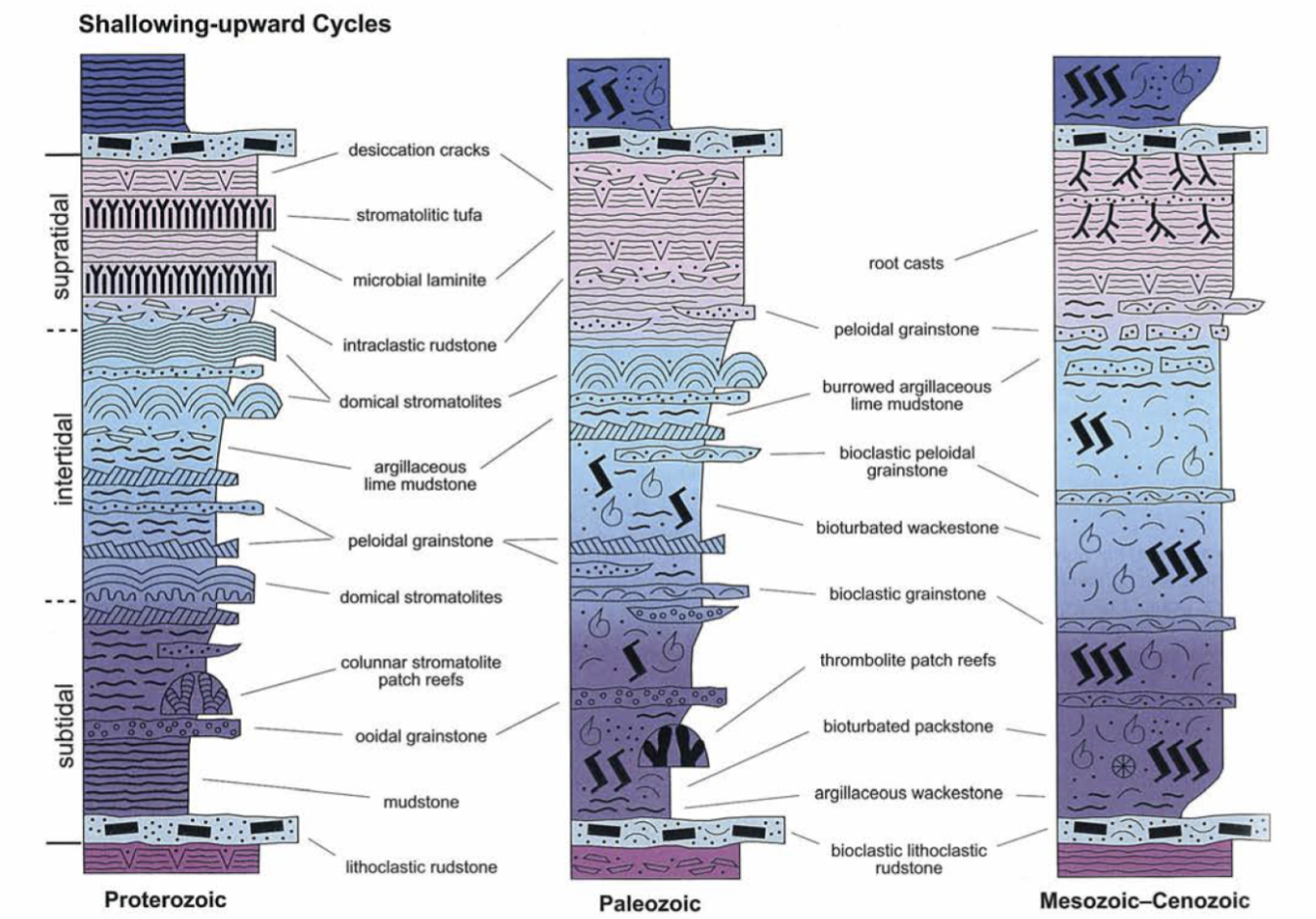
Peritidal cycles
Shallowing upward cycles with stacked subtidal, intertidal, and supratidal deposits
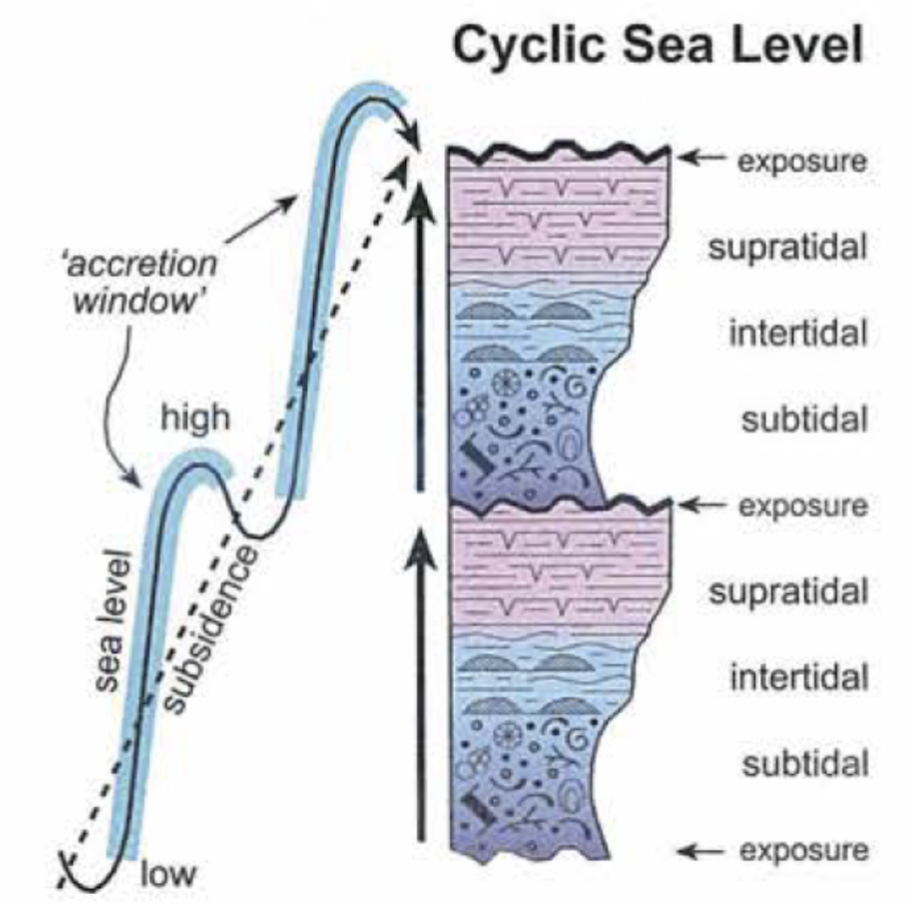
2 sources of peritidal cycles
Autogenic → aggradation or shifting around of environments
Allogenic → driven by sea- level cycles
Location of pelagic carbonate factory
Open ocean; sediments end up in deep ocean basins
Location of benthic carbonate factory
Continental shelves and islands
3 factors determining where carbonates are found in the world’s oceans
Solubility differences between different carbonate minerals
Ocean circulation
Changes in seawater chemistry with depth
The amount of CO2 in seawater drives….
…. precipitation vs dissolution of calcium carbonate
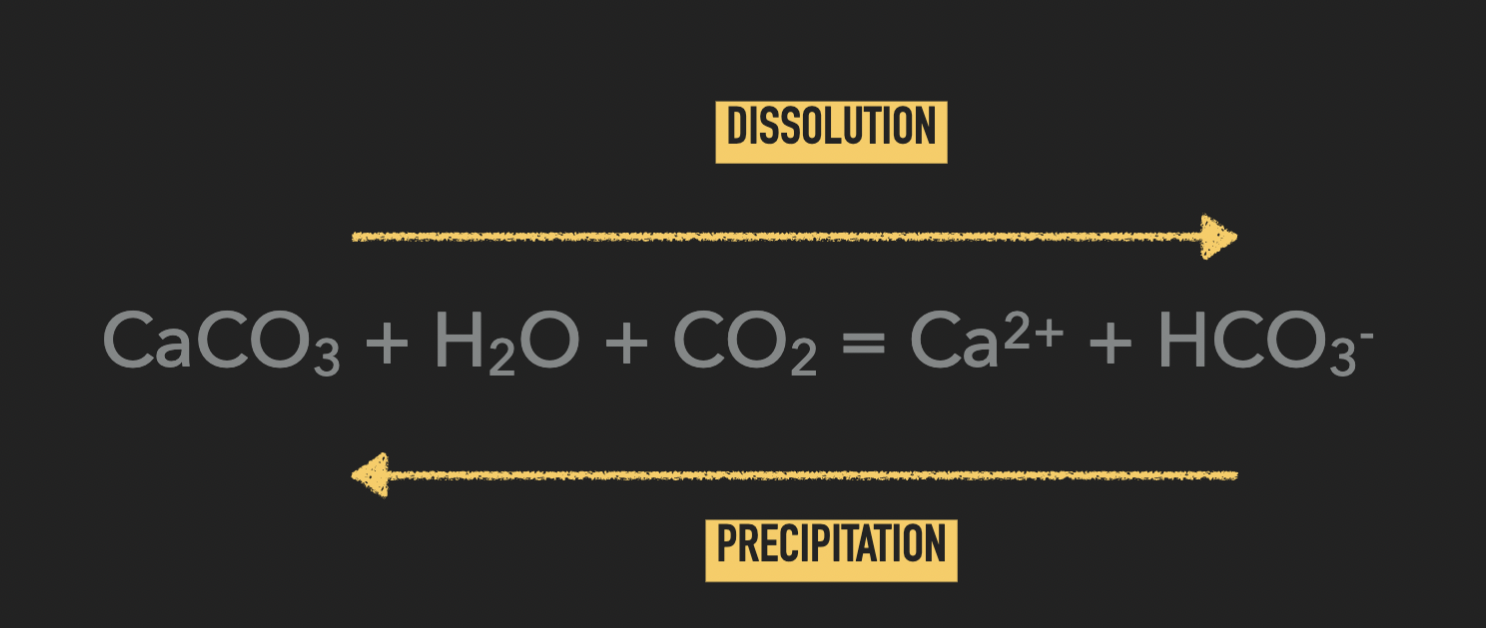
Equation of precipitation vs dissolution of calcium carbonate
CaCO3 + H2 O + CO2 = Ca2+ + HCO3-
→ dissolution
← precipitation
CO2 dissolution and water temperature
Cold water can dissolve more CO2 than warm water
Cold water = dissolution of CaCO3
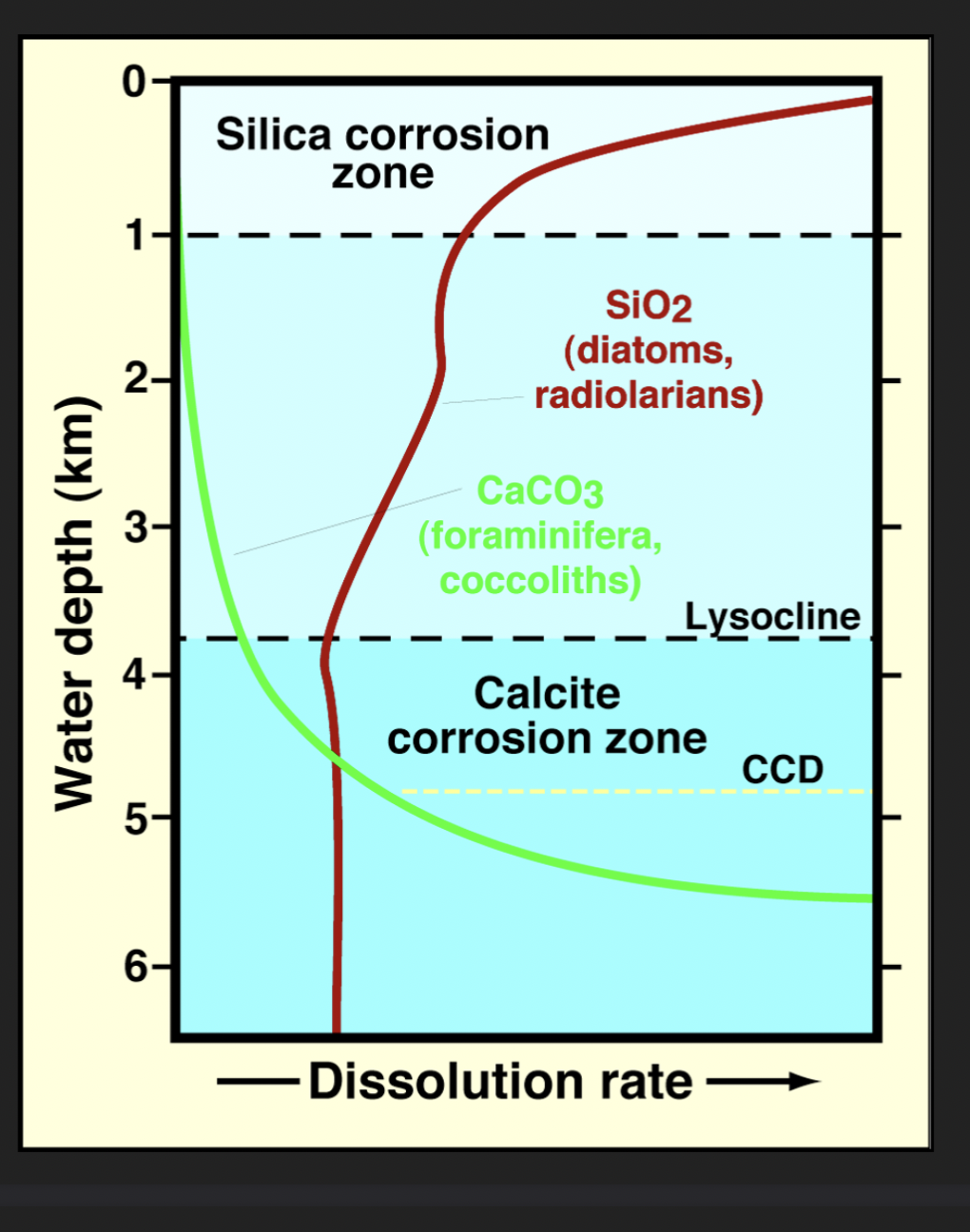
Lysocline
Water depth when we first start to see the corrosion/dissolution in carbonate particles
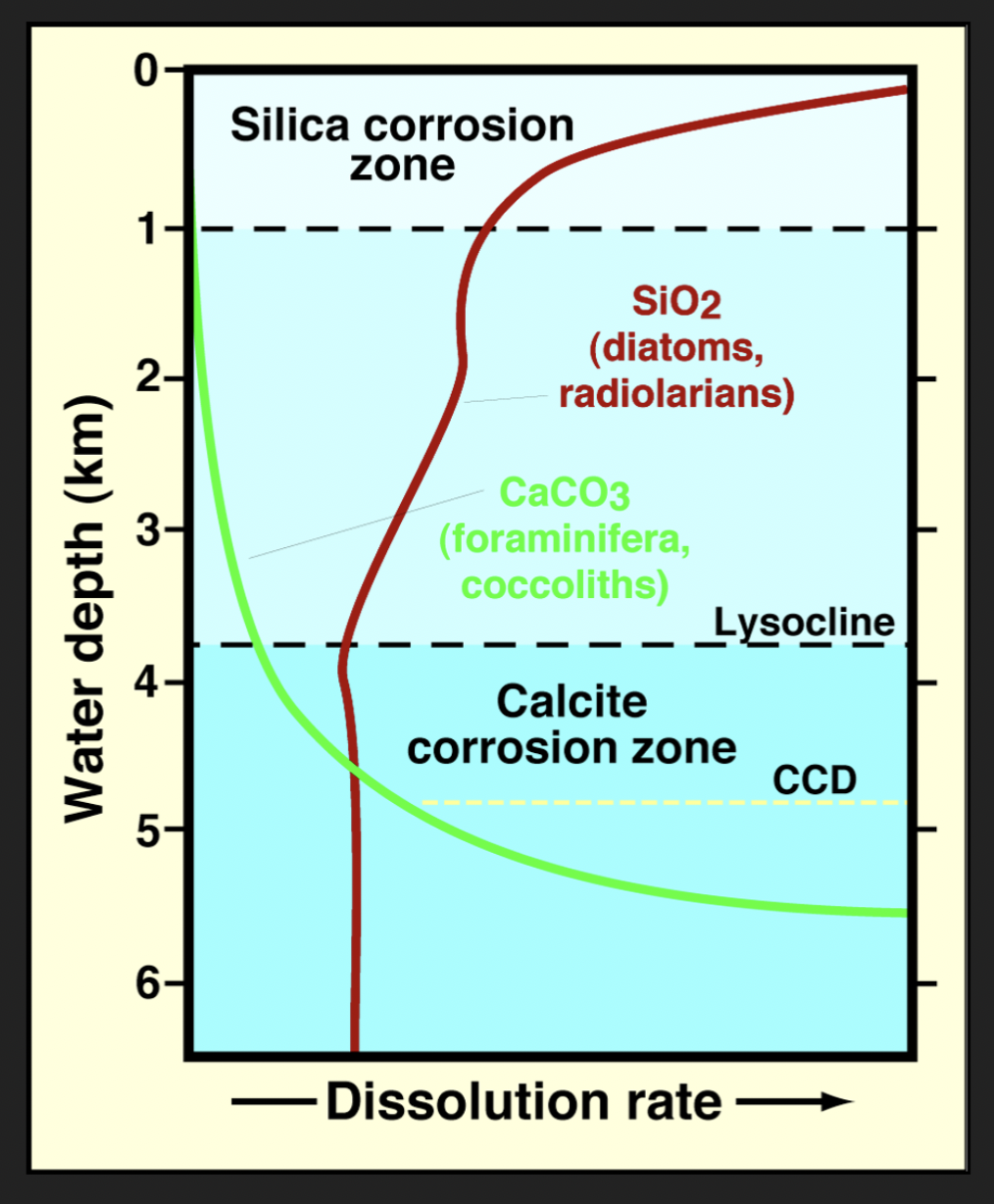
Carbonate Compensation Depth (CCD)
Water depth at which carbonate sediment no longer accumulates
Rate of accumulation = rate of dissolution
CO2 dissolution and thermohaline circulation
Seawater ages with time and older water has higher CO2 content
Water at seafloor in N. Pacific is the oldest water in circulation system
Two sources of deepwater carbonate sediment
Pelagic factory
Periplatform (re-sedimented)
Pelagic factory
Long periods of suspension rain of calcareous phytoplankton and zooplankton
Periplatform (re-sedimented)
Sediment derived from the shallow-water carbonate factory (platform) and redeposited in deeper water
This material is called a re-sedimented deposit
When did the pelagic factory evolve
Didn’t get going in a considerable way until the Jurassic
Carbonate mud in rocks older than Jurassic in deepwater formed on the platform (peri-platform ooze) and been re-sedimented
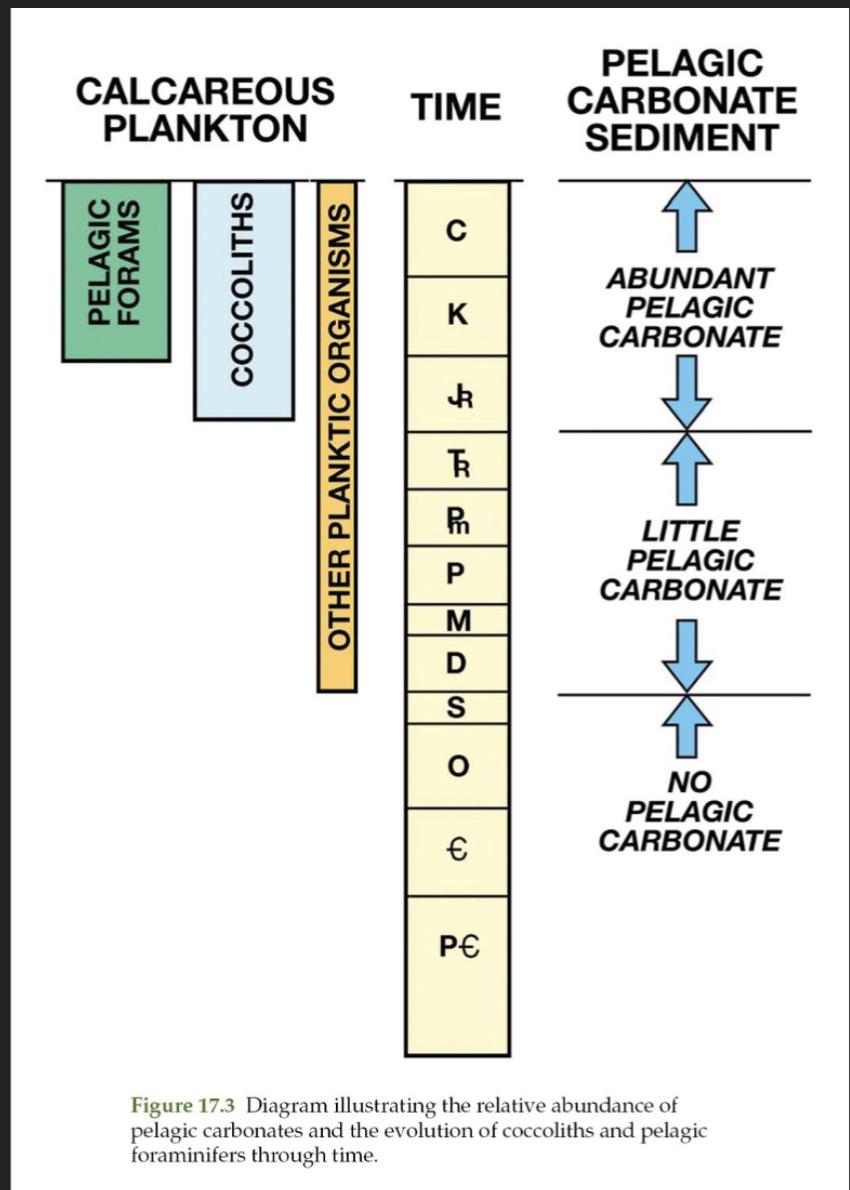
2 organisms which are sources of pelagic carbonates
Foraminifera
Coccolithophores (produce chalk)
Where does resedimented peri-platform ooze accumulate?
More in proximal slopes than distal slopes

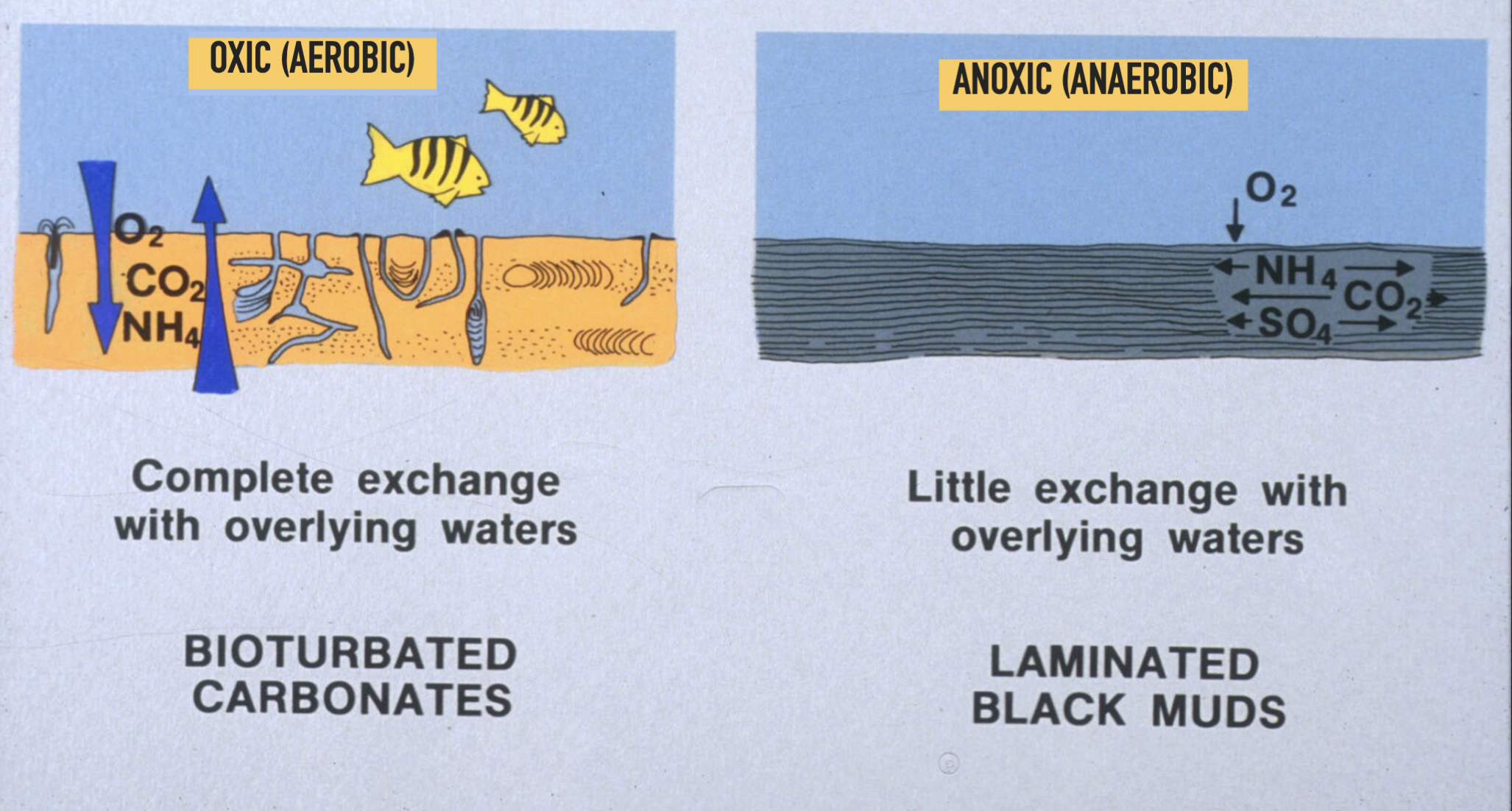
Difference between oxic (aerobic) vs anoxic (anaerobic) environments
Oxic (aerobic) environments → bioturbated carbonates
Complete exchange with overlying waters
Anoxic (anaerobic) environments → laminated black muds
Little exchange with overlying waters
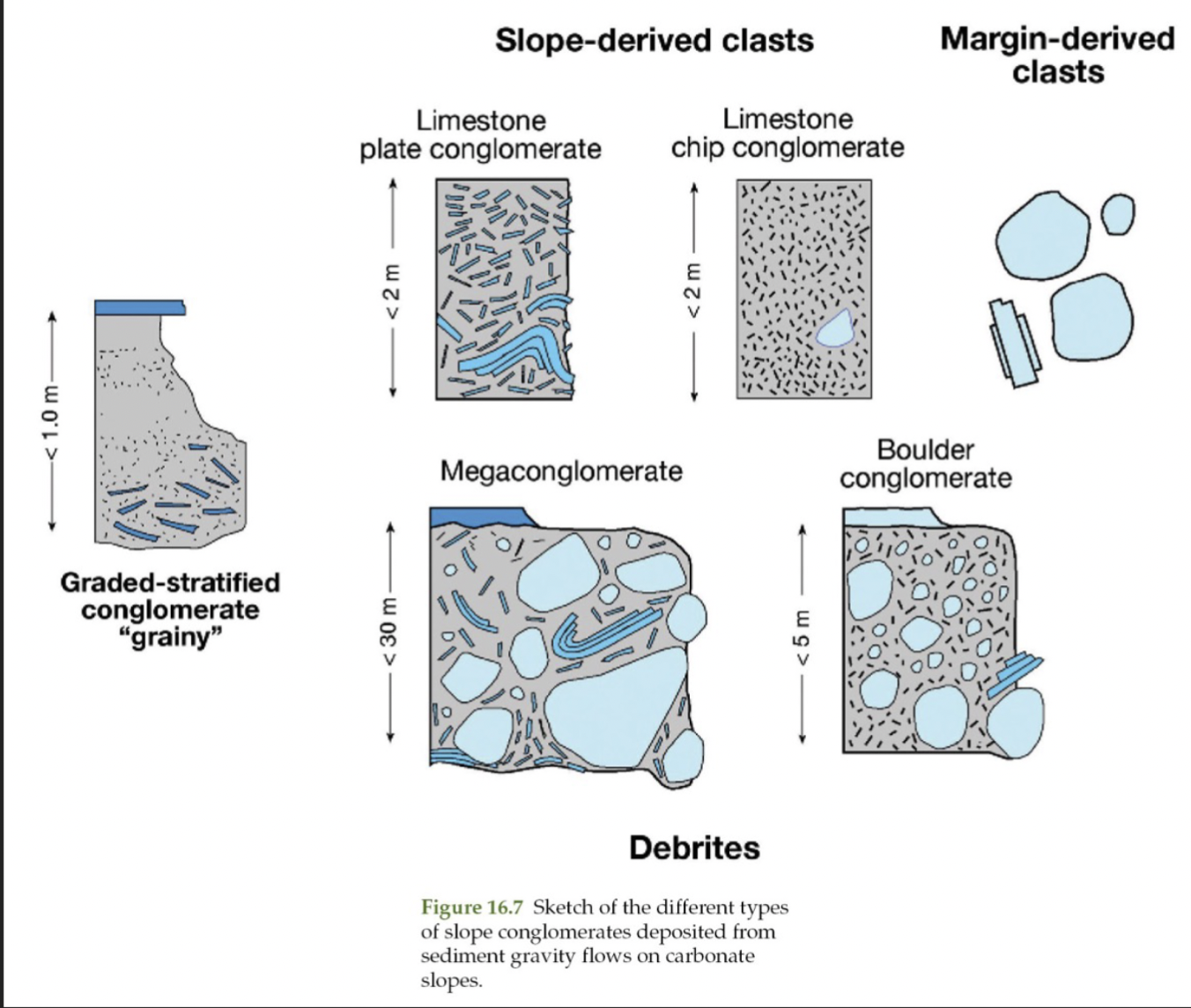
Formation of carbonate slope conglomerates
Resedimented
Coarser deposits found higher on slope
Same transport processes as siliciclastic gravity flows
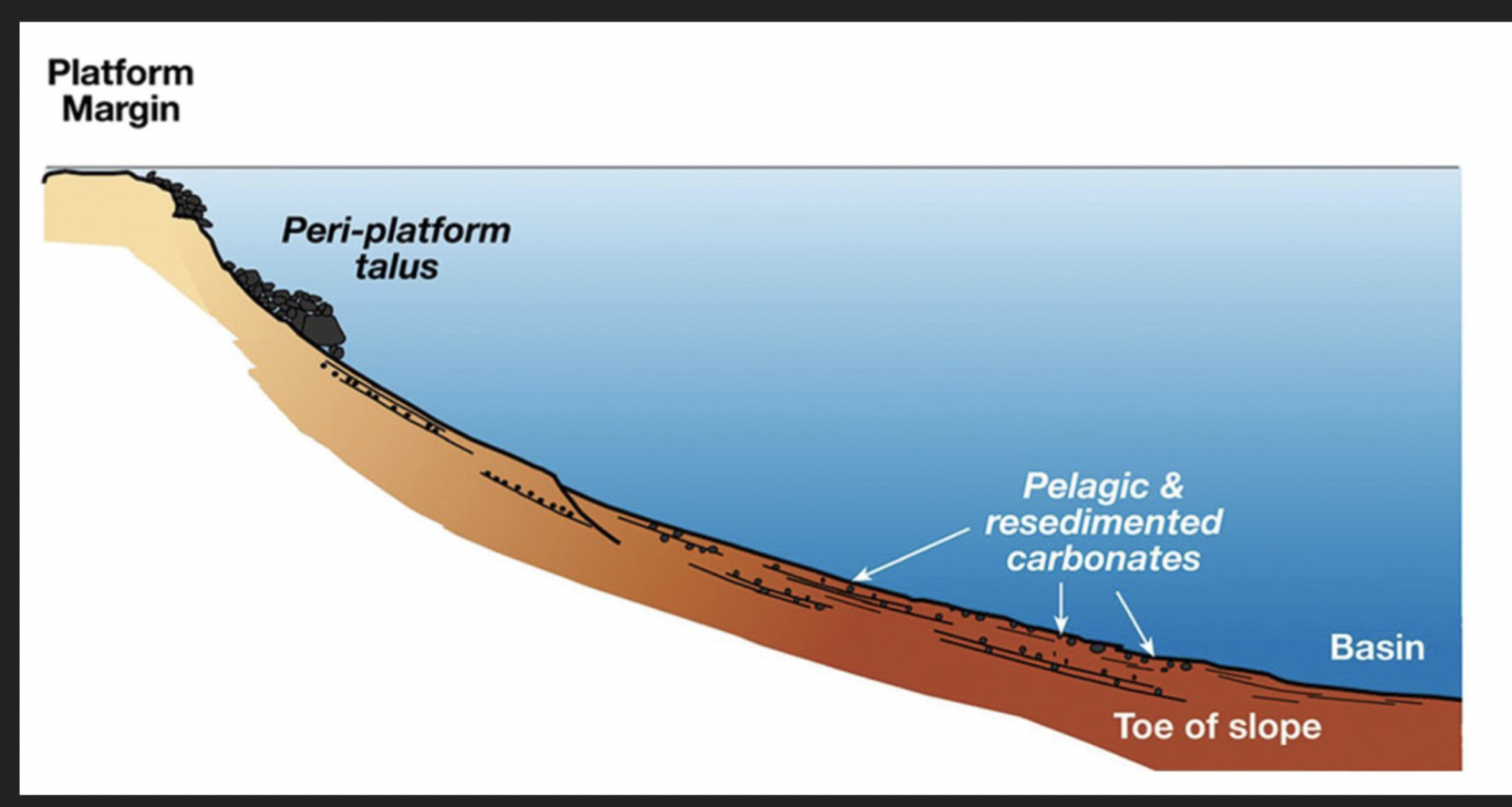
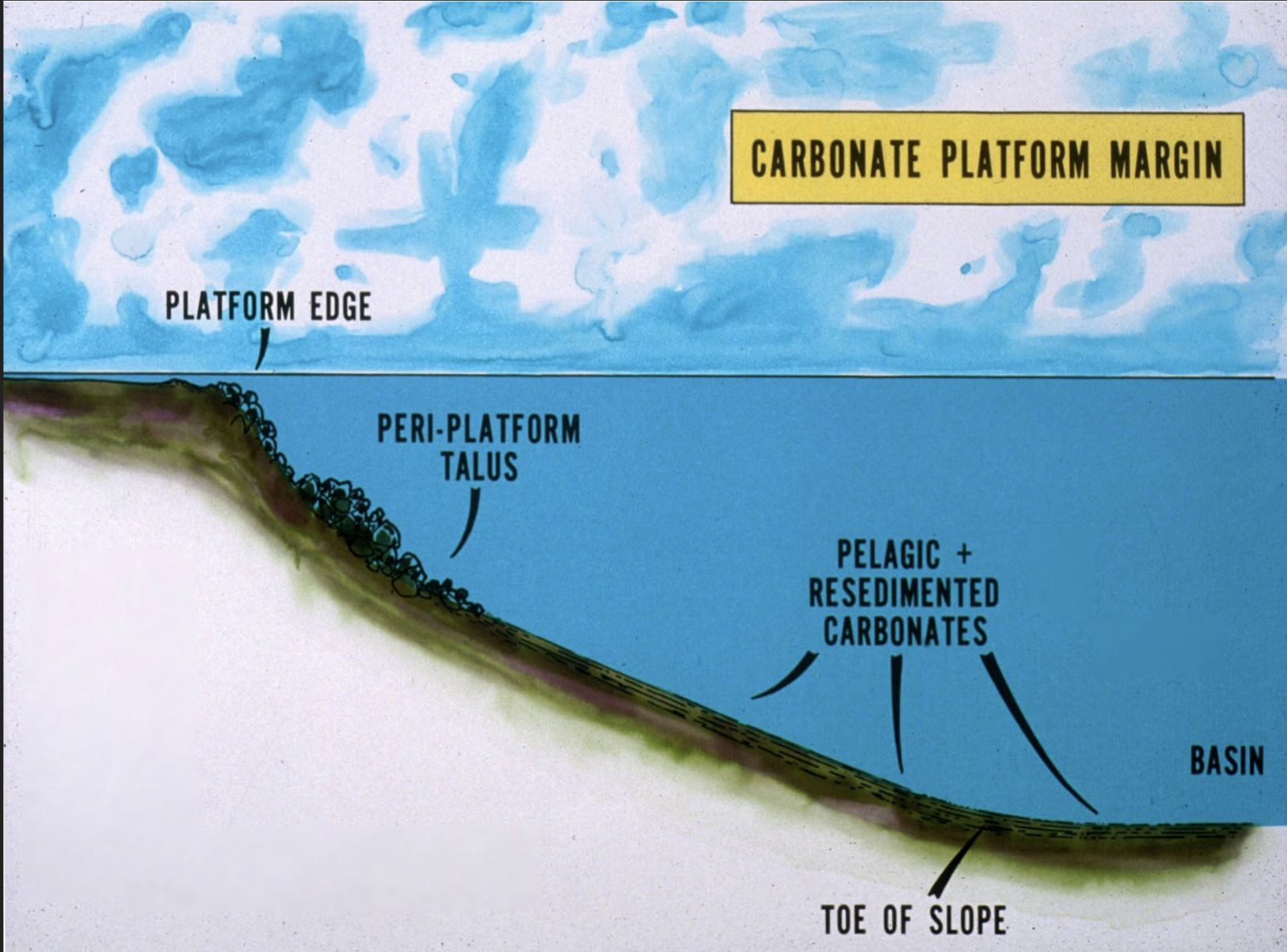
Carbonate platform margin
Platform edge
Peri-platform talus
Pelagic and re-sedimented carbonates (extending to toe of slope)
Basin
Evaporite
Minerals that precipitate from solutions when water evaporates, can form in marine and non-marine settings
Order of precipitation for evaporite compounds
Least soluble compounds are precipitated first
Calcium Carbonate → Calcium sulfate → Sodium Chloride → K and Mg salts
Calcium sulfates (most common evaporites)
Refers to gypsum and anhydrite
Gypsum is hydrous form of calcium sulphate
Anhydrite can form through direct precipitation or through dehydration of gypsum upon burial
Anhydrite may become hydrated to gypsum

4 kinds of gypsum/anhydrite textures
Selenite crystals
Alabaster (fine-grained gypsum)
Fibrous gypsum
Nodular textures (chickenwire and enterolithic structures)
Formation of selenite crystals (gypsum/anhydrite texture)
When gypsum precipitates directly from seawater→ large selenite crystals

Formation of alabaster (gypsum/anhydrite texture)
When gypsum forms through rehydration of anhydrite → alabaster (fine-grained gypsum)

Formation of fibrous gypsum (gypsum/anhydrite texture)
Forms in veins
Formation of nodular textures (gypsum/anhydrite texture)
When gypsum grows within carbonate or clayey sediments → gypsum crystals will alter to anhydrite pseudomorphs
2 sub-textures:
Chickenwire structure
Enterolithic structure
Chickenwire structure (gypsum/anhydrite texture)
When gypsum nodules in carbonate/clayey sediments grow large enough to start coalescing and interfering

Enterolithic structure (gypsum/anhydrite texture)
When gypsum nodules in carbonate/clayey sediments demand more space as nodules grow, creating folded, contorted, ropy layers

3 kinds of halite deposits
Salt crusts (shallow water)
Laminated forms (deeper water; typically also
include anhydrite/gypsum laminae)
Teepee structures (peritidal)
Teepee structures
Expansion of salt crusts due to mineral precipitation can cause buckling into inverted v-structures, common in peritidal environments
Halite teepees → supra tidal setting
Carbonate teepees → junction between water-table and sediment surface

Playa lakes (ephemeral lakes, also called salt pans)
Periodic, usually seasonal, wetting and drying in arid environments leads to alternations of evaporites and clastic sediment layers
Can have smaller continental evaporite settings where water accumulates (e.g. between dunes)
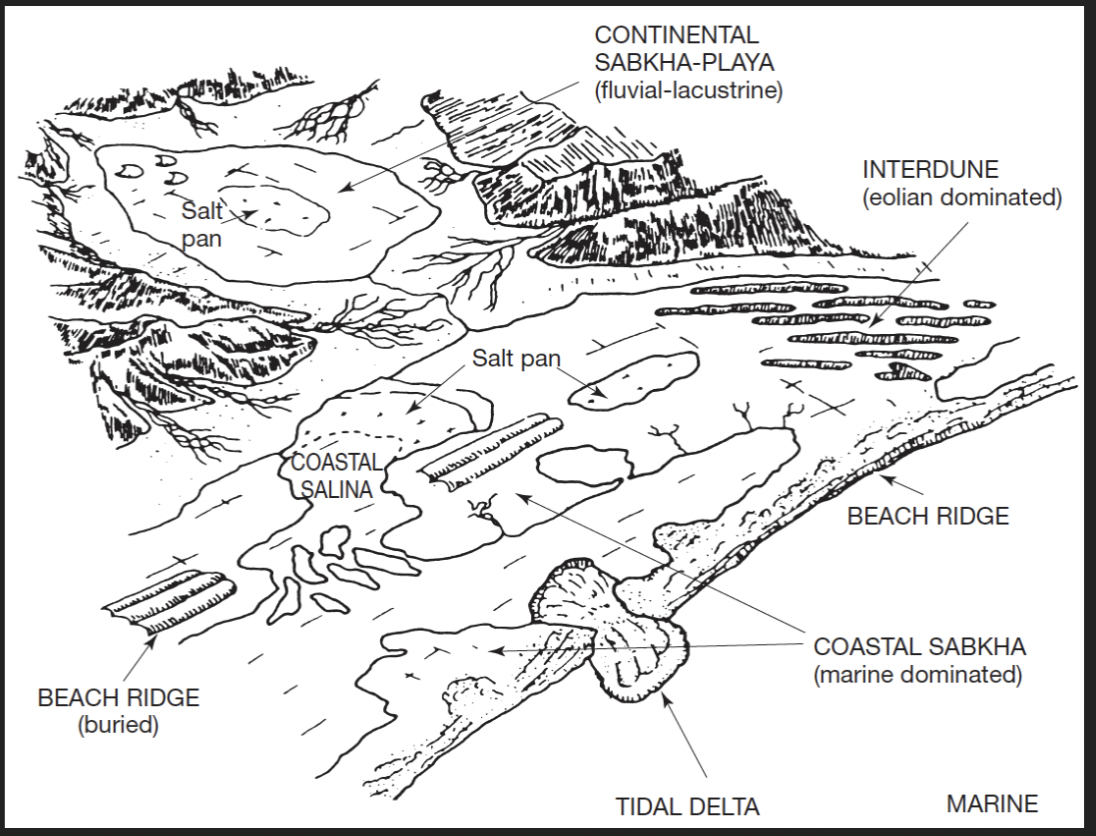
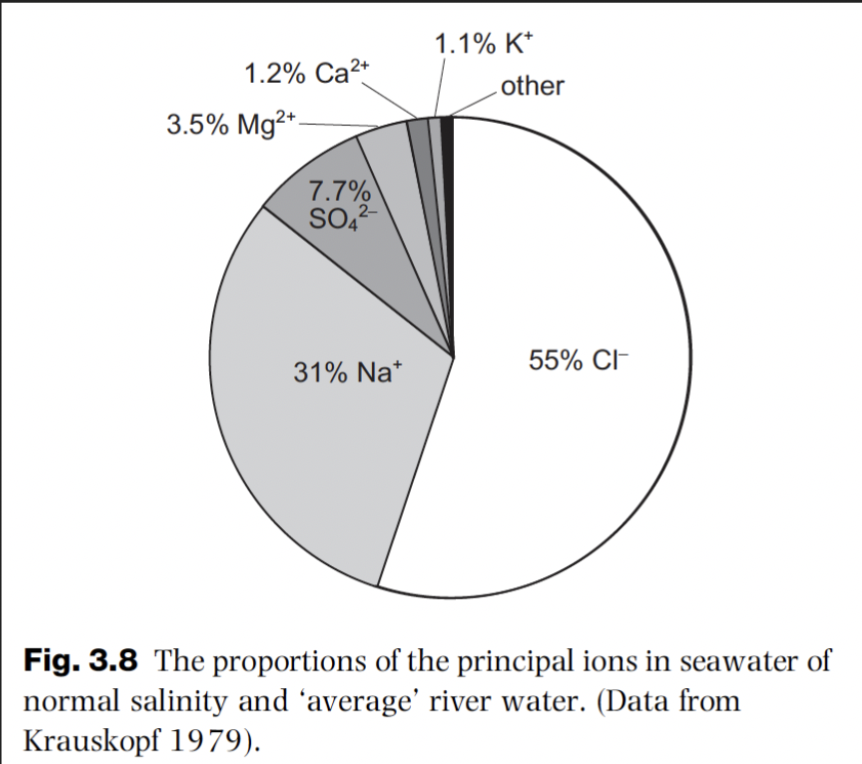
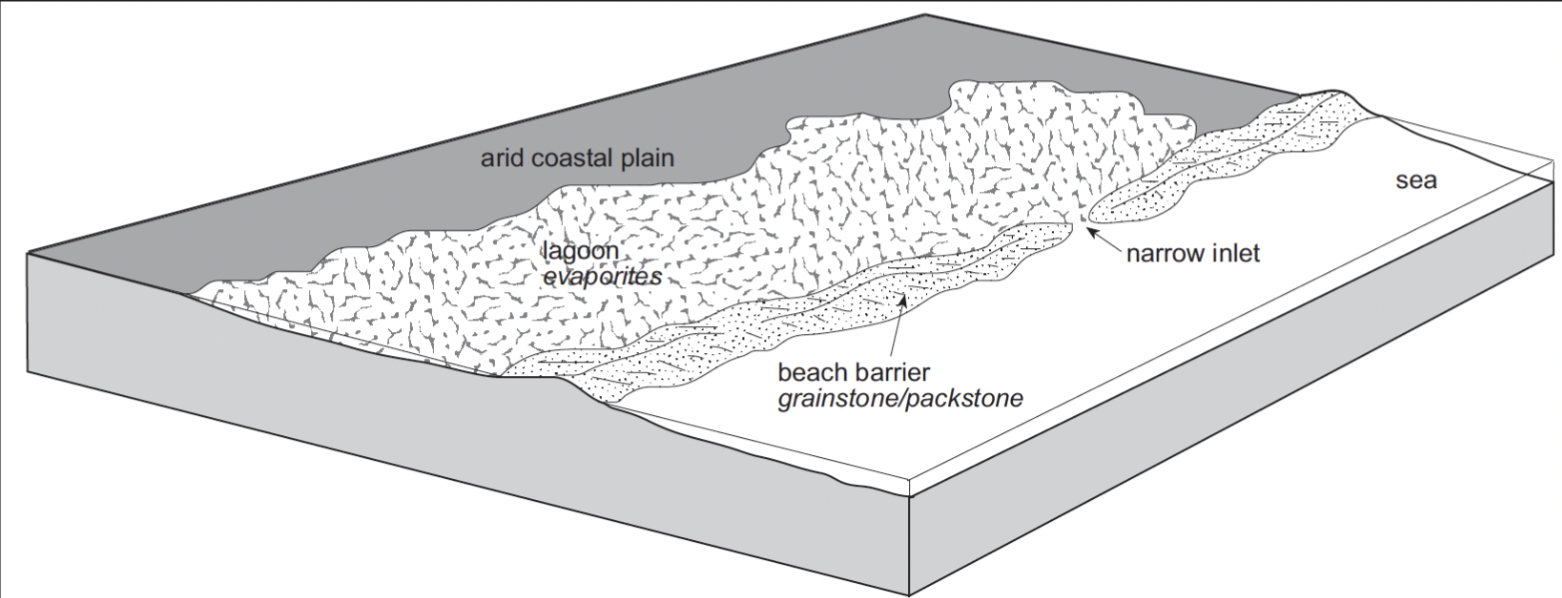
Where do marine evaporites form today?
Restricted coastal regions like salinas and coastal sabkhas
Salinas (arid lagoons and salt pans) may precipitate layered gypsum and/or halite
Fluctuations in salinity will result in variations in precipitated minerals and occasional carbonate layers
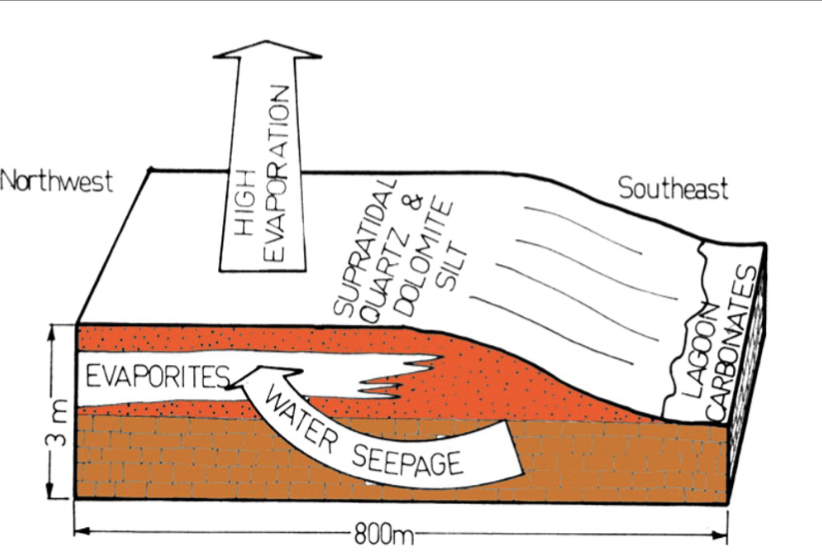
Where do evaporites form within sabkhas?
Within sediment above a saline water table with high evaporation rate
Ancient evaporites
Geologic evidence for thick accumulation of evaporites in deep marine basins in the past
Evaporation of a column of seawater 1,000 m thick will produce ~15 m of evaporites
Need no (or very limited) freshwater supply to basin and restricted opening to ocean so water can be replenished slowly
Formation of deepwater evaporites
Thick evaporite deposits require arid conditions and a silled basin (some kind of constriction (vertical or lateral) that impedes circulation of seawater)
Sills can be tectonic features (folds, etc.) or reefs
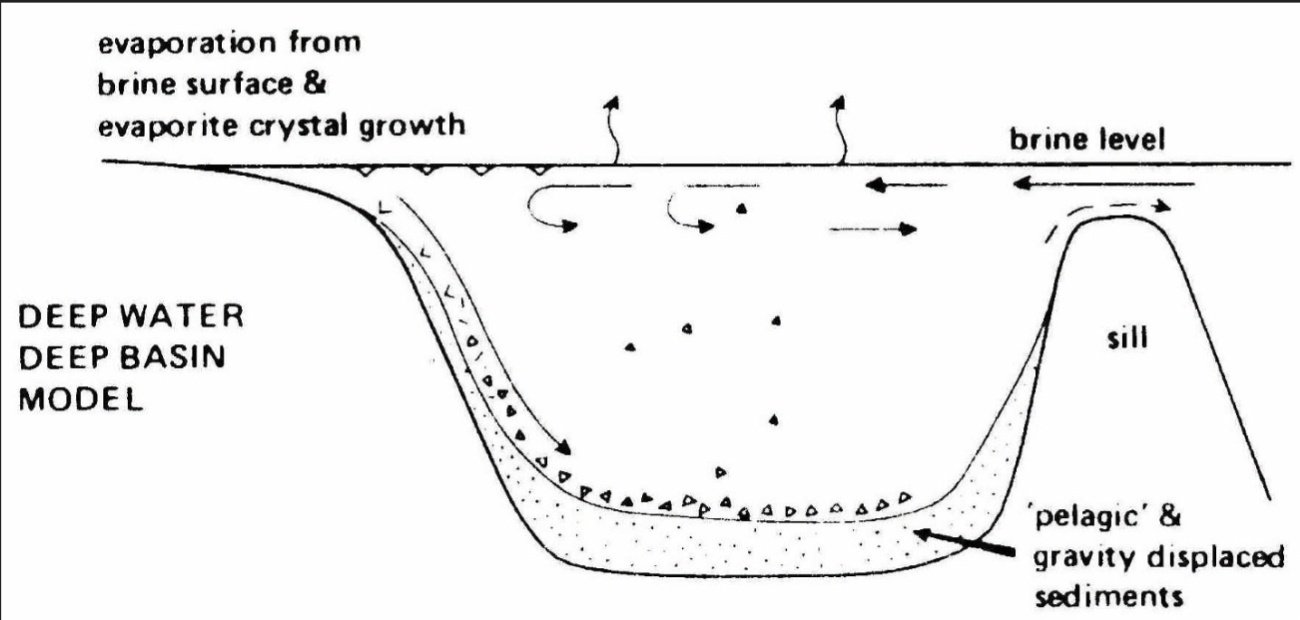
Messinian salinity crisis
Today, more water is evaporating from the Mediterranean than is being delivered by rivers
Causes of evaporation still debated, but the ‘crisis’
lasted from ~ 6 Ma - 5.3 Ma
This water is replenished by a current flowing through Strait of Gibraltar and a deeper current brings warm, salty water back out to Atlantic
Stratigraphy
The study of larger successions of sedimentary rocks and how time is recorded
3 principles defined by Nicholas Steno
Principle of Superposition
Principle of Original Horizontality
Principle of Lateral Continuity
Formation
A fundamental lithostratigraphic unit; a body of rock, identified by lithologic characteristics and stratigraphic position, that is mappable either at the surface or in the subsurface
Member
A lithostratigraphic unit next in rank below a formation (formations can be broken into one or more members)
Group
Consists of assemblages of formations
4 types of stratigraphic surfaces
Conformity
Disconformity
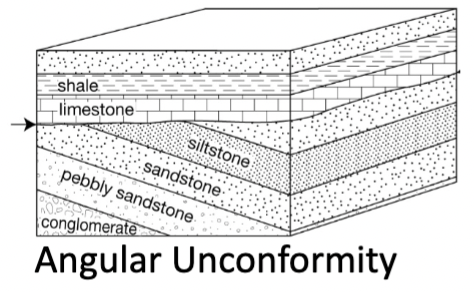
Angular unconformity
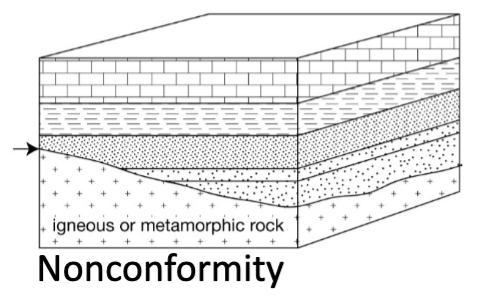
Nonconformity
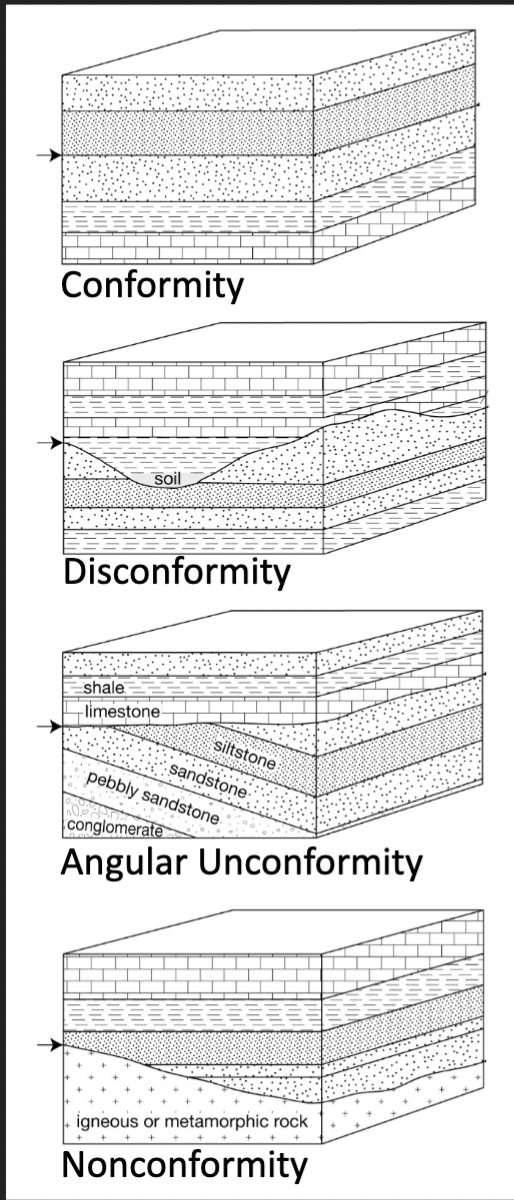
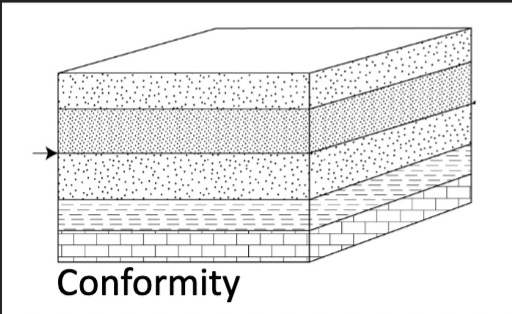
Conformity
A surface that separates younger and older strata but along which there is no physical evidence of non-deposition
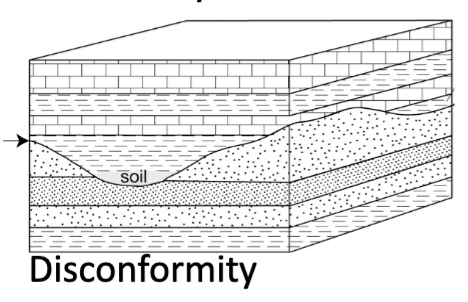
Disconformity
Strata concordant, but evidence of erosion with significant missing time
Angular unconformity
Older strata erosionally overlain by younger strata deposited at a different angle
Nonconformity
Younger strata overlying igneous or metamorphic rock
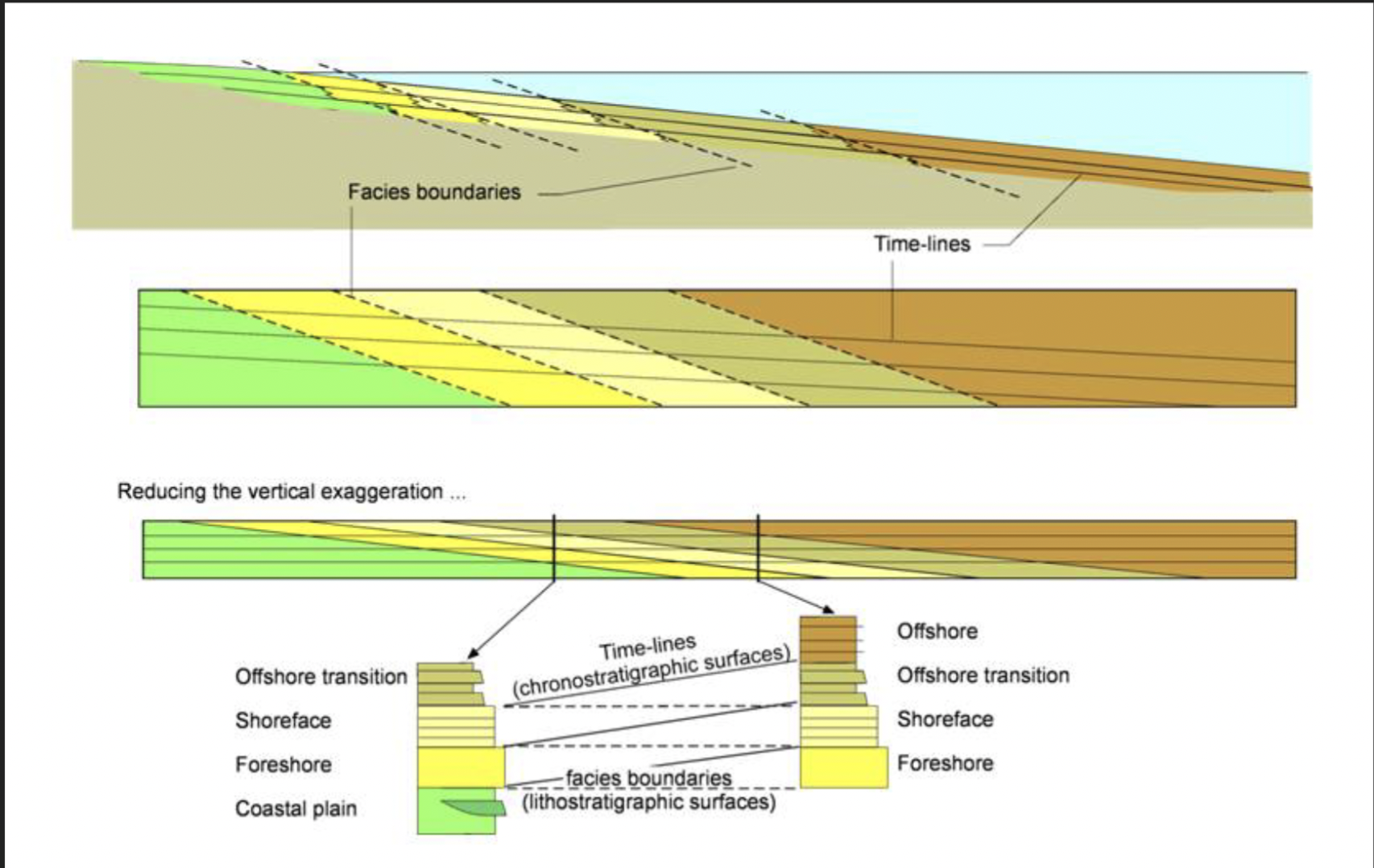
Facies
A package of sedimentary rock distinguishable by lithologic, structural, and biogenic aspects detectable in the field
Interactions between facies and stratigraphy
We want to recognize important stratigraphic surfaces (e.g., unconformities) and characterize the facies between those surfaces
Lithostratigraphy
Linking units of similar lithology and stratigraphic position
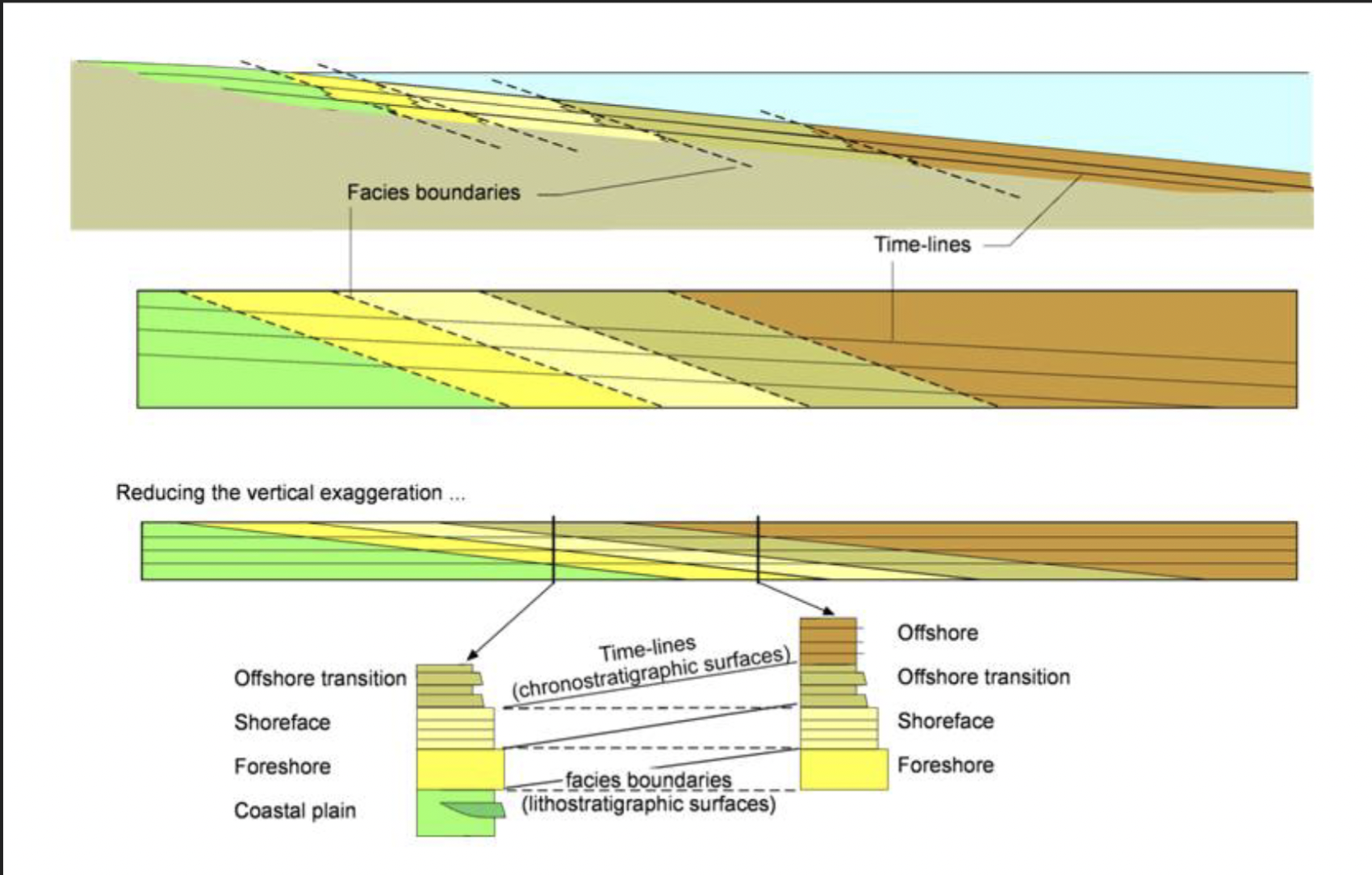
Chronostratigraphy
Linking units that were deposited at the same time (regardless of lithology)
Foundation of sequence stratigraphy
Dunvegan formation
A clastic wedge (i.e. a delta complex) that built eastward into a shallow marine sea as a result of a phase of mountain building in the Rockies
Reefs
Shallow water carbonate deposit in the subtidal carbonate factory
2 kinds of platforms
Rimmed platform (barrier reef system)
Unrimmed platform (no barrier reef system; can be open shelf or ramp)
Patch reef
Small reefs that develop in a restricted lagoon environment
5 dating/correlation techniques in stratigraphy
Biostratigraphy
Absolute dating
Magnetostratigraphy
Chemostratigraphy
Sequence stratigraphy
Biostratigraphy
The use of fossil occurrences within the rock record to establish correlations between time-equivalent rock strata as determined by the presence of a particular fossil species