Science Separating Mixtures (Keyword)
1/19
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
20 Terms

Boiling
The change of state from a liquid to a gas.
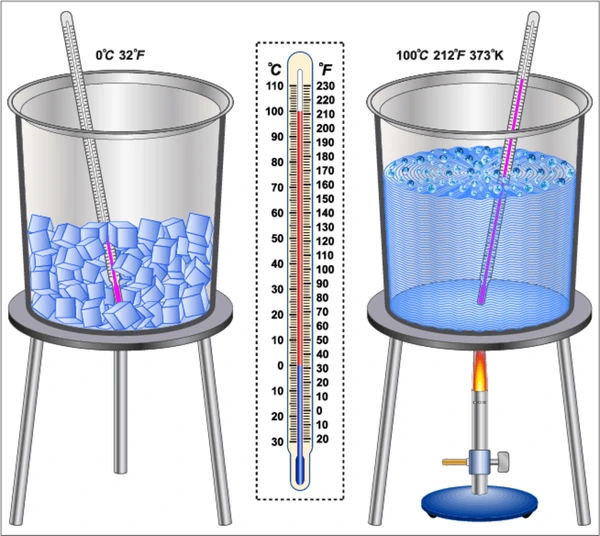
Boiling point
The temperature at which a substance changes state from a liquid to a gas.
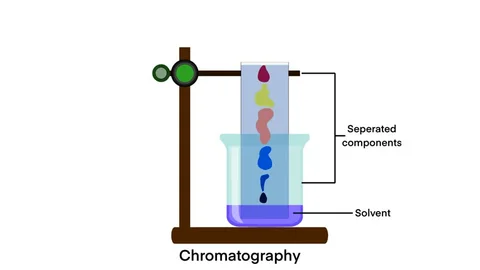
Chromatography
Used to separate different coloured substances.
Condensation
The change of state from a gas to a liquid.
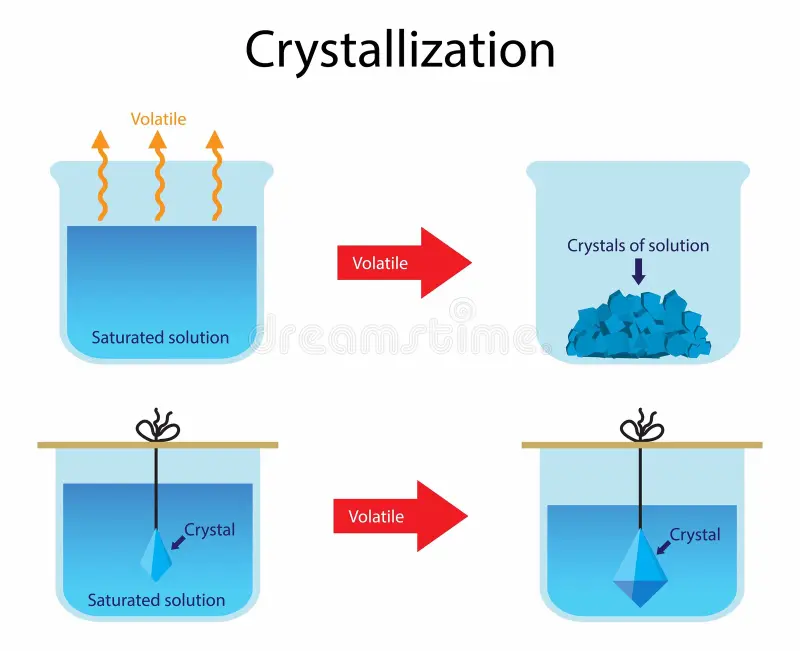
Crystallisation
The process of evaporating a solvent from a solution, leaving crystals of the solute behind.

Dissolve
When a solute mixes completely with a solvent to make a solution.
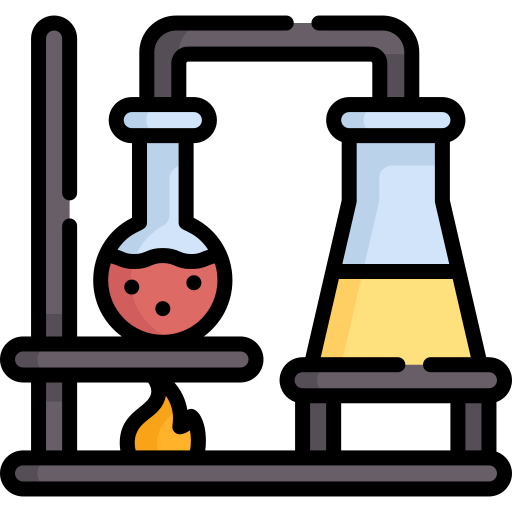
Distillation
A separation technique that separates substances by boiling and condensing.
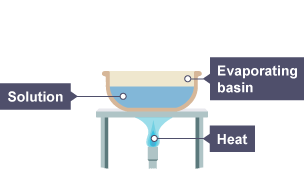
Evaporation
The process of a liquid turning into a gas slowly.

Filtrate
The liquid collected after filtration.
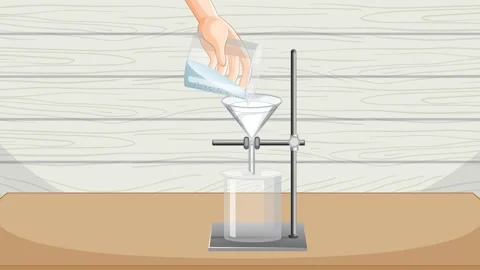
Filtration
A separation technique that uses filter paper to remove an insoluble substance from a liquid.
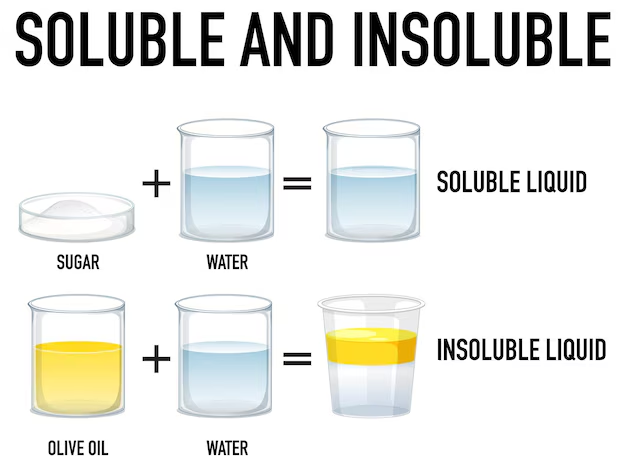
Insoluble
A substance that will not dissolve in a liquid.
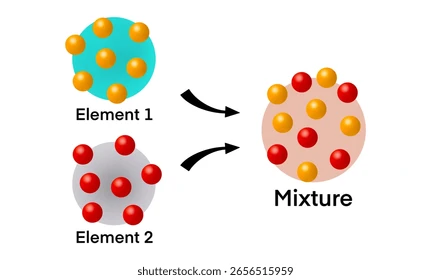
Mixture
Two or more pure substances mixed together, whose properties are different from the individual substances.

Residue
The solid left in the filter paper after filtration.
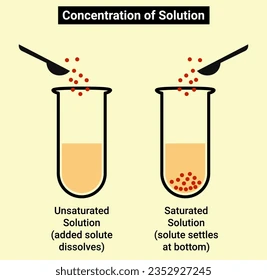
Saturated solution
A solution where no more solute can dissolve.
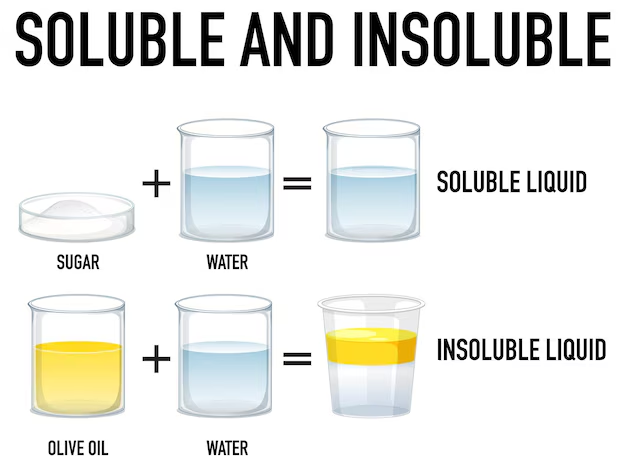
Soluble
A substance that can dissolve in a liquid.

Solute
A substance that dissolves in a liquid.

Solvent
A substance, usually a liquid, that dissolves another substance.

Solution
A mixture formed when a solute dissolves in a solvent.

Solubility
The maximum mass of solute that dissolves in a certain volume of solvent.
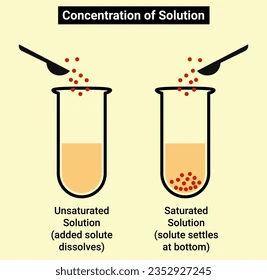
Unsaturated solution
A solution where more solute can still dissolve.