S.3 - Classification of Matter
0.0(0)
Studied by 0 peopleCard Sorting
1/5
There's no tags or description
Looks like no tags are added yet.
Last updated 12:07 PM on 3/27/26
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
6 Terms
1
New cards
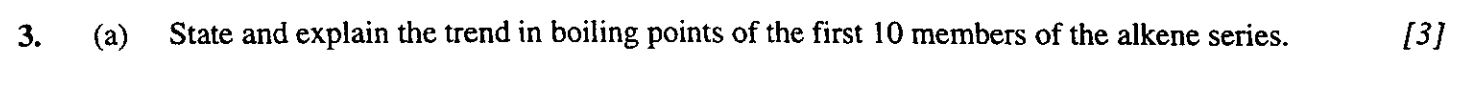
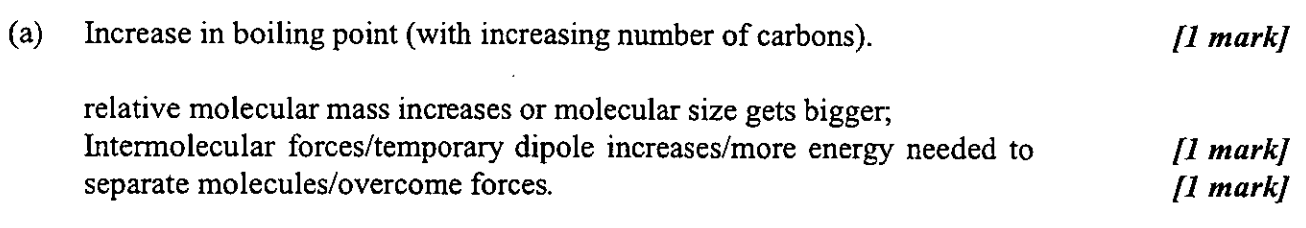
2
New cards
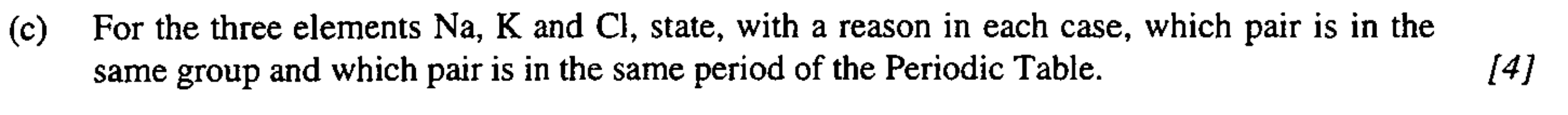

3
New cards


4
New cards
a) Define, in words or with an equation, the first ionization energy of Na
b) State how the first ionization energy varies down group 1
c) Li, Na and K react with water. Which of the three reactions will be the most vigorous? Explain this at an atomic level.
d) State whether the second ionization energy of sodium is less than, the same as, or greater than the first ionization energy. Explain your answer.

5
New cards


6
New cards

