Chem/Physics MCAT
1/141
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
142 Terms
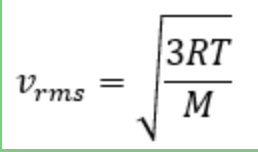
what is the equation for the root mean square velocity of gas particles in a substance
isothermal
word that signifies no temperature change
Adiabatic
a word that signifies no heat exchange (Q=0)
Isobaric
a word that signifies constant pressure
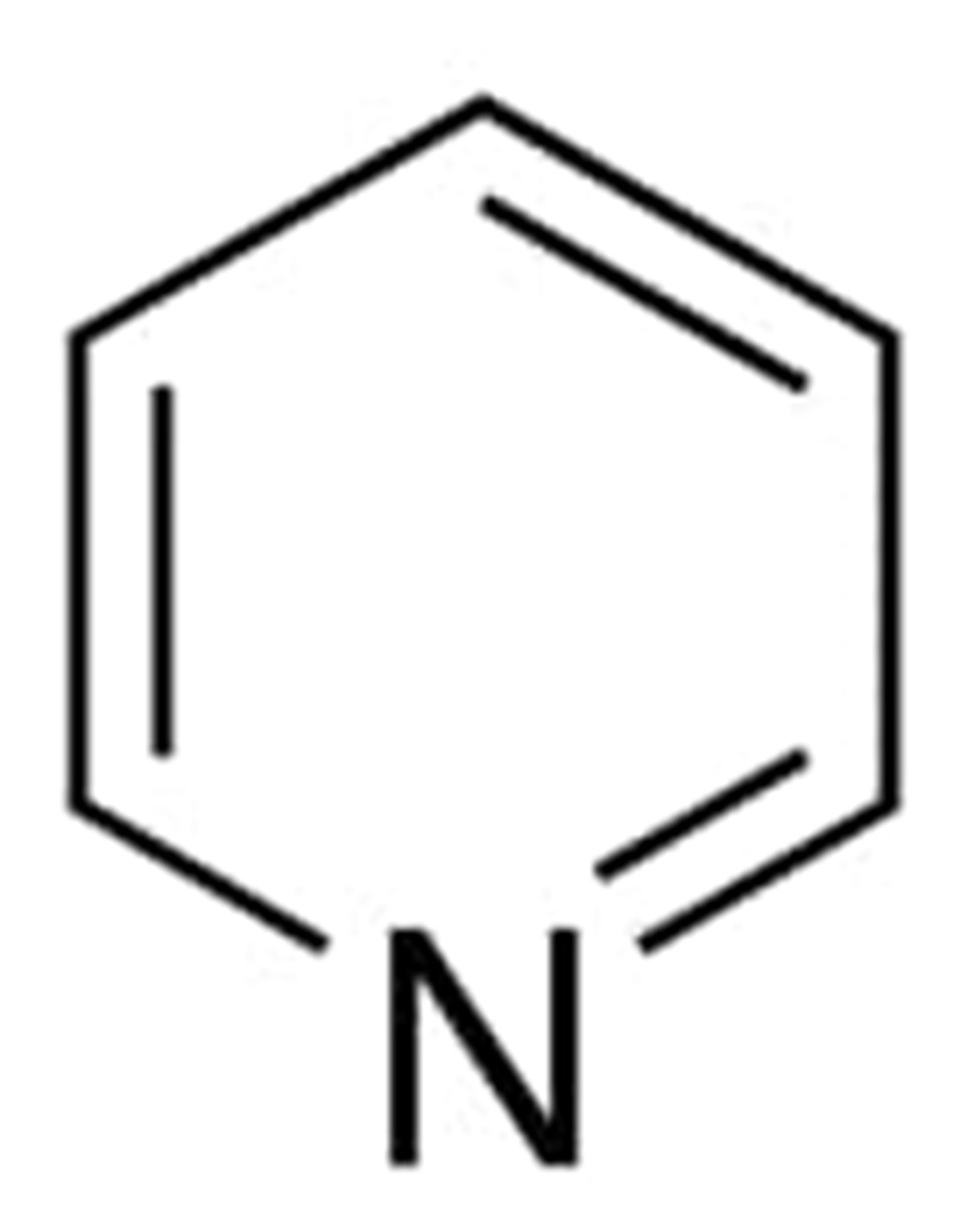
pyridine
the presence of a hemiketal
How can you determine if a cyclic sugar was a ketone?
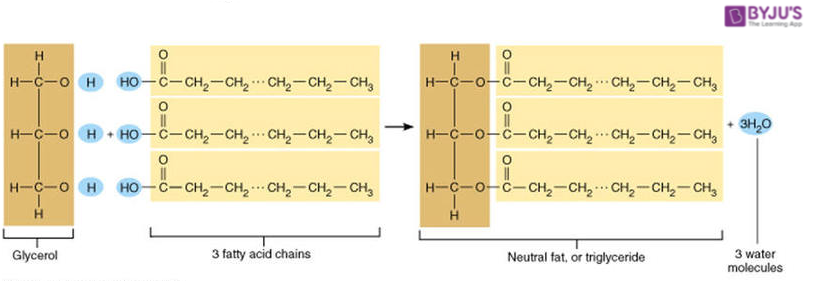
structure of triglyceride

Archimedes’ Principle equation
homogenous catalysts
catalysts that are in the same phase as the reactants
Acid chlorides > Acid anhydrides > Esters > Carboxylic acids > Amides
What is the order of relative reactivities of carboxylic acids and their derivatives?
Alcohol (OH) IR Peak
Deep U shape around 3000-3600 cm -1
Carboxylic Acid (OH) IR Peak
broad peak overlapping with C-H bonds, giving it a jagged appearance at 2800-3200 cm-1
Carbonyl (C=O) IR Peak
sharp and deep peak at 1700-1750 cm-1
Amine or Amide (N-H) IR Peak
one or two sharp peaks at 3300-3500 cm-1
triglycerides
One glycerol molecule bonded to three fatty acid chains through ester linkages
gas-liquid chromatography
an analytical technique used to separate a complex mixture into its individual components based on two main factors:
boiling point- lower BP moves through the system faster
affinity to the stationary phase
Both of these affect the Retention Time
E=hf
equation relating the energy of a photon to its frequency where h is Planck’s Constant
f = v/λ
equation relating frequency, wavelength, and speed of a photon
avogadro’s number
6 × 10^23
E˚
Electrochemical potential calculated as the difference between the reduction potentials of the cathode and anode reaction
if E˚ is positive, the reaction is spontaneous
Reaction quotient (Q)
tells you where the reaction is right now.
When Q < Keq, the reaction favors the products
When Q > Keq, the reaction favors the reactants
When Q=Keq, the reaction is in equilibrirm
Spectator Ion
Ions that do not participate in the chemical reaction and remain unchanged in solution
Arrhenius base
earliest description of a base that is describe as increasing the concentration of hydroxide (OH-) ions when in solution
Bronsted-Lowry base
type of base that is described to act as a proton acceptor
Lewis Base
a type of base that donates a pair of electrons
Bernoulli’s Principle (Velocity vs. Static Pressure)
In a moving fluid, as velocity increases, static pressure decreases but dynamic pressure increases
dyanamic pressure
The kinetic energy per unit volume of a fluid in motion that increases with velocity
Formula: ½ pv²
Hydrostatic Pressure
The pressure exerted by a fluid at equilibrium at a specific point, caused by the weight of the fluid above it due to gravity.
Formula: P = pgh
Infrared (IR) spectroscopy
a method used to identify specific functional groups present on an unknown molecule. Group frequencies are vibrations that are associated with certain functional groups. A molecule can be identified when its vibrational frequencies on an IR spectrum are compared to known spectra values.
UV-Vis spectroscopy
This technique detects conjugated pi systems and metal-to-ligand charge transfer species, both of which exist here
the field lines get denser
how does the appearance of electric field lines change as the field gets stronger
lewis acid
a molecule that donates an electron pair
lewis base
a molecule that accepts an electron pair
Bronste- lowry acid
a molecule that donates a hydrogen atom
Pd/C
a catalyst used in hydrogenation reactions:
The Pd surface physically pulls the H-H bond apart, making the hydrogens reactive.
Because the alkene "docks" onto a flat metal landing pad, both hydrogens are delivered to the same face of the double bond simultaneously. —> Syn addition
In a DC circuit, a fully charged capacitor acts as an open circuit (a broken wire) and there is no current flowing through —> the bulb is off
A DC battery is connected in a single series loop to a resistor (light bulb) and a capacitor. What is the state of the bulb in the Steady State, and what is the physical reasoning?
Keq
refers to the ratio of products to reactants at equilibrium of the reaction
diamagnetic materials
materials with only paired electrons that are slightly repelled by a magnetic field
paramagnetic materials
materials that have unpaired electrons with random spins, and thus, are weakly attracted to magnetic fields
ferromagnetic materials
materials in which the spins of the electrons in adjacent atoms tend to align parallel to each other. This alignment creates regions called domains, where the magnetic moments are all aligned. When the material is magnetized, these domains line up, resulting in a strong magnetic effect
first ionization energy
how much work an external force must do to steal an electron away from the nucleus's positive pull.
This energy increases as atomic radii decreases and effective nuclear charge decreases
principle quantum number
Describes the size and energy level of the orbital (also called the shell) written as positive integers (n = 1, 2, 3 …).
As n increases, the orbital becomes larger and the electrons spends more time away from the nucleus, increasing its energy
angular momentum quantum number (azimuthal)
Describes the shape of the orbital (the subshell)
l = 0 —> s orbital
l = 1 —> p orbital
l = 2 —> d orbital
l = 3 —> f orbital
Because electronegativity is defined as an atom's ability to attract electrons within a chemical bond.
Since Noble Gases have a full octet, they are chemically inert and rarely form bonds, making their "attraction" to shared electrons effectively zero or undefined
Why are Noble Gases (He, Ne, Ar) typically excluded from the Pauling Electronegativity Scale?
Stokes shift
is the difference in wavelength (or frequency) between the light that a molecule absorbs and the light that it eventually emits.
When a fluorescent molecule (a fluorophore) absorbs a photon, it gains energy. When it later releases that energy as light, the emitted photon almost always has less energy than the one that was absorbed
inverse - as energy increases, wavelength decreases (shorter)
what is the relationship between energy and wavelength?
Increasing length increases resolution because SEC relies strictly on the physical path through porous beads
In Size-Exclusion Chromatography (SEC), how does increasing the column length affect the separation of proteins
thin layer chromotography
a lab method of separating molecules by their polarity
TLC stationary phase
a thin layer of polar absorbent like silica gel
TLC mobile phase
a liquid solvent that moves up the stationary phase via capillary action to separate components
Retention Factor
a numerical value used in chromatography to describe how far a specific compound travels compared to how far the solvent (the mobile phase) travels.
A high retention value indicates a fast sample that troubles far up the stationary phase
-ate
suffix for the standard oxyanion
-ite
suffix for an oxyanion with one fewer oxygen that the -ate form
per-
prefix used when an oxyanion has one more oxygen that the -ate form
hypo-
prefix when an oxyanion has one fewer oxygen than the -ite form
-ic acid
what happens to -ate when the molecule becomes an acid?
-ous acid
when happens to -ite when the molecule becomes an acid
N = n x M
what is the equation for Normality?
Galvanic cells
a cell set up to house spontaneous redox reactions in a way that generates or uses electrical energy to create a current
Anode: negative
Cathode: positive
E˚ = positive
Daniell cell
a system in which two half-cells are connected by a salt bridge, which allows spectator ions to travel in between half-cells → serves to balance the charges as the reaction progresses
electrolytic cells
a system in which electrical energy is used to drive a nonspontaneous redox reaction
anode: positive
cathode: negative
E˚ is negative
charges are attractive
what does a negative electrostatic force indicate?
bohr effect
It explains how certain conditions in the body—specifically, high levels of CO2 and acidity (low pH) —cause hemoglobin to release oxygen more readily to the tissues that need it most
venturi effect
is a principle in fluid mechanics where a fluid's velocity increases and its static pressure decreases as it flows through a constricted section of a pipe.
Pascal’s law
states that when pressure is applied to a confined, incompressible fluid, that pressure change is transmitted undiminished to every part of the fluid and to the walls of its container.
a more spontaneous process
What does a low/negative ∆H value signify?
a more spontaneous process
What does a higher/more positive ∆S value signify?
Hammond’s Postulate
states that the structure of a transition state resembles the species (reactant, intermediate, or product) to which it is closest in energy.
In Endothermic Reactions, the transition state resembles the products
In Exothermic Reactions, the transition state resembles the reactants
Vitamins B and C
Which vitamins are water-soluable?
Vitamins A, D, E, and K
Which vitamins are lipid-soluble?
Vitamin K
The vitamin associated with coagulation — functions to modify clotting factors in the liver
Vitamin A
the vitamin that functions as a precursor for retinal
Vitamin D
The vitamin that functions to regulate calcium and phosphate absorption in the GI tract
Vitamin E
this vitamin functions to Eliminate radicals and protect the cells from oxidative damage
Vitamin C
this vitamin is an antioxidant and is known for collagen synthesis
Vitamin B
This vitamin acts as a coenzyme or a precursor to coenzymes (NAD+, FADH+, etc)
Dyenin
a motor protein that walks towards the minus end of the microtubules, which is oriented towards the center of the cell. Movement toward the center of the cell is described as retrograde. (Dying in the middle of the ring)
Kinesin
a motor protein that is responsible for anterograde transport in cells (Kicking things out)
˚incidence > ˚critical
this results in total internal reflection
The value of an equilibrium constant is ONLY changed by temperature, not concentration.
What is the “golden rule” of equilibrium constants?
Downfield
in what direction do proton signals move if they experience deshielding effects?
geometric isomers
These have the same connectivity (the atoms are attached to the same neighbors) but differ in their spatial arrangement around a rigid bond (cis and trans)
constitutional isomers
These have the same connectivity (the atoms are attached to the same neighbors) but differ in their spatial arrangement around a rigid bond (like a double bond or a ring).
gamma particles
high energy photons that have 0 charge
alpha particles
a helium nucleus with a charge of +2
beta particles
these particles are electrons with a charge of -1
positrons
these particles have a charge of +1
half-equivalence point
the point during titrations at which the pH is equal to the pKa of the solution being titrated because the concentrations of the acid and base are equal (log(1)=0)
inverse: high Ka = low pKa = strong acid
relationship between Ka and pKa?
isolated system
a system that exchanges neither energy nor mass with its surroundings
closed system
a system in which there is energy exchange but not mass exchange with the surroundings
open system
a system that exchanges both mass and energy with its surroundings
latent heat
heat released or absorbed during a phase change
strong nuclear force
the force that holds the protons together in the nuclear > the electromagnetic force that repels the positive charges
binding energy
the energy input needed to break the nucleus apart into its components
mass defect
the difference in expected mass of the nucleus and the actual mass due to some of the mass being transformed into nuclear binding energy
photoelectric effect
when protons hit the surface of a metal, they excite metallic electrons to a higher energy state, causing them to move farther away from the atomic nucleus and be emitted
work function
the minimum amount of energy required to eject an electron from the metallic atomic nucleus = the energy of the incident photon
alpha decay
results in the emission of 2 protons and 2 neutrons, causing the atomic number to decrease by 2 and the atomic weight by 4
beta minus decay
the decay that occurs when a neutron is converted into a proton an electron is emitted