CHE 334 Biochemistry Unit 3 Quiz
1/29
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
30 Terms
Enzymes…
lower activation energy, lower transition state
Enzymes help _____ bonds making them easier to rearragne
weaken
Activation energy
energy needed for the reaction to begin
Gibbs Free energy
how spontaneous a rxn is
oxidoreductase
the transfer of electrons, can catalyze both foward and reverse rxn
Å + B ← → Bº + A
transferase
transfer of a chemical group to create a new substance
AX+ B ← → BX + A
Hydrolase
addition of water to cleave a molecule into two
A + H2O → B + C
Lyase
Breaking a molecule down into two W/O H2O, No ATP used
Generates either a double bond or a ring in order to work
A → B + C
Isomerase
molecule being converted to one of its isomers by rearranging atoms w/in the molecule
A → B
Ligase
A and B are being catalyzed to combine the two
A + B -→ AB
Cofactors
small inorganic chemicals
tightly binds to enzymes
added to enzyme to help with activation
Coenzymes
organic
binds loosely to enzymes
added to enzyme to help with activation
Competitive inhibition
similar structure yet not the exact same shape
Blocks enzyme from working
Binds directly to the active site, competing with the substrate
Non Competitive Inhibition
Binds to the allosteric site
changes enzyme shape to not bind to its specific substrate, does not work
What is the Michaelis-Menten Equation
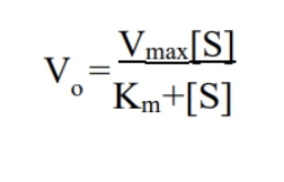
How do you calculate kCat?
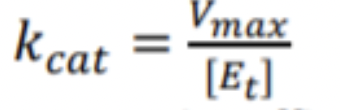
How to calculate specificity constant?

Which of these produces the most product per unit time (Kcat)?
1) 9.7 x 10 4
2) 1.4 × 10 3
3) 3.8 × 10 3
4) 4.0 × 10 5
4) 4.0 × 10 5
Which enzyme likely binds most tightly to its substrate (Km)?
1) 1.5 × 10 -4
2) 2.6 × 10 -1
3) 9.5 × 10 -3
4) 2.5 × 10 -2
1
Which enzyme is the most catalytically efficient (specificity constant)?
1) 9.3 million
2) 1.5 million
3) 10 million
4) 152k
3
How do you know if an enzyme is catalytically efficient?
If it has a high product per unit per time and a loose binding to a substrate
High Kcat and low Km
Where does chymotrypsin cleave and where?
Cleaves on carboxyl
Specific for aromatic acids: Tyr, Phe, Trp
Where does trypsin cleave and where?
Cleave on carboxyl
Specific for amino acids with positive side chains: Lys, Arg
Where does elastase cleave and where?
Cleaves on carboxyl
Specific for Gly, Val, Ala, Leu, & Ile small hydrophobic amino acids
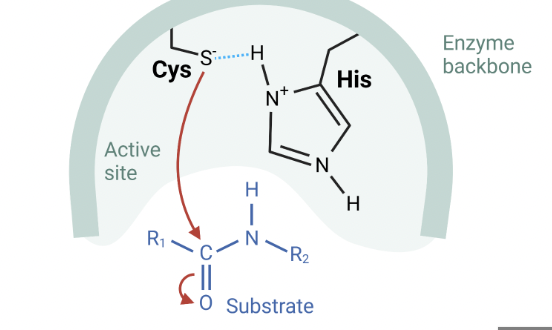
Covalent catalysis
involves transient covalent bonds btw enzyme and substrate
Acid-base catalysis
reaction is accelerated by addition of acid or base that will not be consumed by the rnx
Metal-ion catalysis
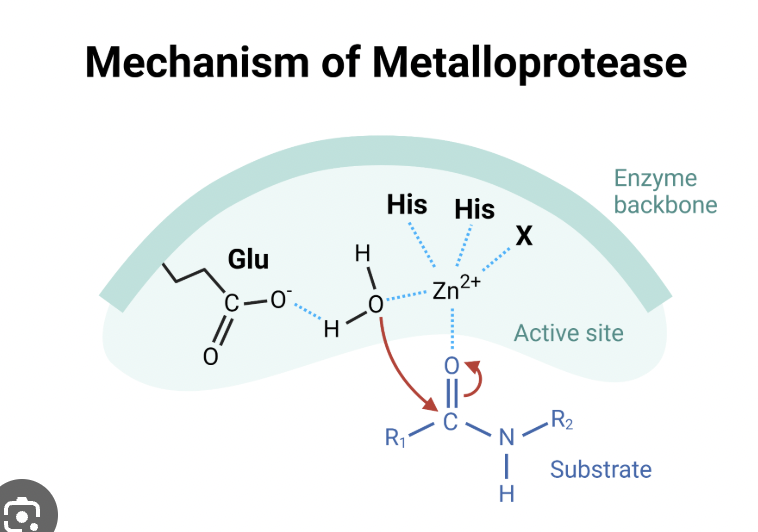
metal atom binds to active site, metal atom interacts with H2O, and deprotonates H2O to make it a nucleophile
Cysteine protease
cleaves at carbonyl group
specific for cysteine
role: immunity and apoptosis
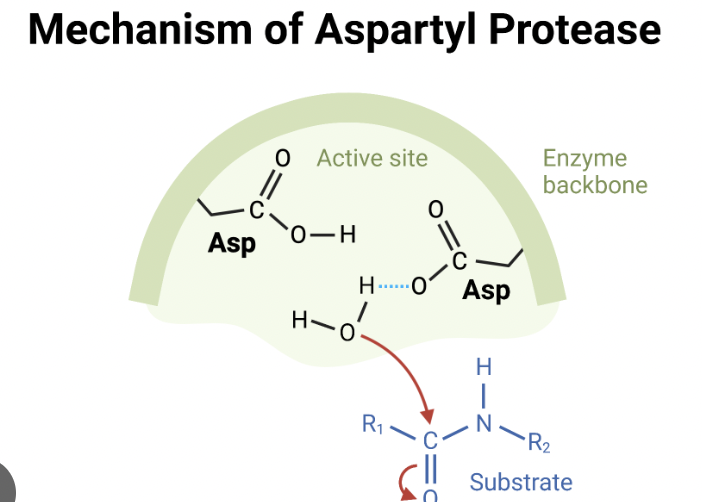
Aspartyl Protease
Cleaves at carbonyl group
specific for aspartic acid
aids in digestion, protein degradation
Metalloprotease
cleaves at carbonyl group
specific for metal atoms
cell proliferation, angiogenesis, apoptosis