Amines
1/22
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
23 Terms
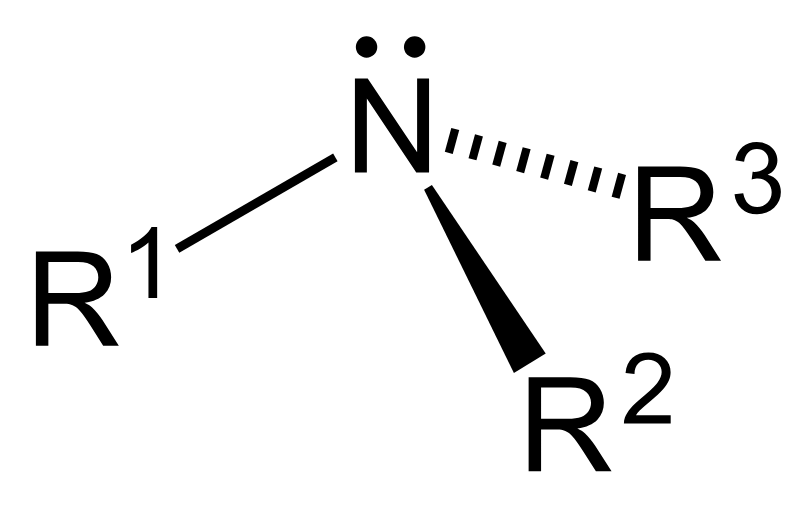
What is a primary amine?
1 carbon group attached to N and 2 hydrogens
E.g R-NH2
What is a secondary amine?
2 carbon groups attached to N and 1 Hydrogen
E.g R-NH-R
What is a tertiary amine?
3 Carbon groups attached to N and no remaining H
How do you prepare aromatic amines?
Reduction of nitro compounds
Using Sn (catalyst) and HCL
Room temp
Equation for the reduction of a nitro compound into an aromatic amine
Nitro compound + 6 [H] → Amine + 2H2O
Reducing agent written as [H] in equation
![<ul><li><p>Nitro compound + 6 [H] → Amine + 2H<sub>2</sub>O </p></li><li><p>Reducing agent written as [H] in equation </p></li></ul><p></p>](https://assets.knowt.com/user-attachments/788769fd-421b-4c6e-9834-6aa93cc0dc34.png)
Aromatic amines are used in the manufacture of..
Dyes (specifically azo dyes)
Method 1 of preparing aliphatic amines
2 step synthesis with a halogenoalkane
Final step is reduction of nitrile
Outline the 2 step synthesis of a halogenoalkane to make aliphatic amines
Preparing the nitrile
-Via nucleophilic substitution
-KCN / aq +ethanolic
- e.g. CH3CH2Br + KCN → CH3CH2CN + KBr
Reduction
-Add H2 and nickel catalyst (or Pt/Pd)
-e.g. CH3CH2CN + 2H2 → CH3CH2CH2NH2
Advantages and disadvantages of method 1 (2 step synthesis of halogenoalkane) to make aliphatic amines
Advantages
Higher % atom economy ( desired product is formed = primary amine)
Disadvantages
But reaction only produces primary amines and lengthens carbon chain
2 step reaction → lower % yield
( % yield tells you how much product has been obtained compared with the maximum possible amount and % atom economy tells you how many of the atoms in the reactants end up in the desired product )
What is method 2 for preparing aliphatic amines ?
Reacting ammonia with halogenoalkanes
Nucleophilic substitution
+When an excess of NH3 is used the major product is primary amine
What is the problem with method 2 (preparation of aliphatic amine)
Amine produced can act as a nucleophile as it has a lone pair of electrons on N atom
Even with an excess of NH3 further substitution can occur
This reaction thus produces primary, secondary and tertiary amines and quaternary ammonium salts.
A mixture of all these chemicals can be formed therefore product is impure
Thus low % atom economy
In method 2 if excess halogenoalkane is used..
The production of secondary and tertiary amines are favoured.
But major product is quaternary ammonium salt
Why are amines bases and why weak bases
Because N atom has a lone pair of electrons that can accept H+
But lone pair is often less available to accept H+→ making it a weak base
What happens when you react amine with strong acid ?
Reaction goes to completion
E.g CH3CH2NH2 + HCL → CH3CH2NH3 +Cl-
What does base strength of amines depend on?
Availability of the lone pair of electrons on the N atom
Why are aliphatic amines strong bases?
Alkyl groups are electron releasing
This is known as the positive inductive effect
Lone pair is more available to accept H+ ( Electron density pushed towards N)
Why are aromatic amines the weakest base?
In benzene there is a a delocalised pi electron system
Lone pair of nitrogen is delocalised into the pi cloud of the benzene
Lone pair is thus less available to accept H+
In aliphatic amines the more alkyl groups attached to the amine..
The greater the positive inductive effect
Thus tertiary amines are stronger bases
What are nucleophilic properties of amines
Amines are nucleophiles
What are the 2 mechanisms for amine reactions
Nucleophilic substitution
Nucleophilic addition-elimination
How can quaternary ammonium salts form?
In nucleophilic substitution reactions where there is a large excess of halogenoalkane
E.g ( CH3)3 N + CH3Cl → N (CH3)4Cl
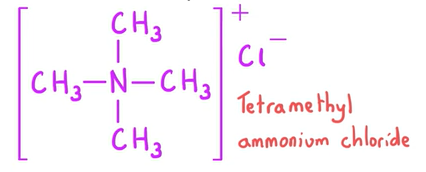
What are quaternary ammonium salts used as?
Cationic surfactants such as
→Fabric softener
→Hair conditioner
→Disinfectants
Why can Quaternary ammonium salts be used as cationic surfactants
Because they contain a positively charged ammonium head and long hydrocarbon tails
The charged ammonium head is attracted to H2O
While the non polar tail dissolves in grease