Micro exam 3
1/117
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
118 Terms
Amoeba cyst form
Formed stool, rigid, non-motile, infectious form
Amoeba trophozoite form
Liquid stool, active feeding, active disease
Amoeba motility mechanism
Pseudopods
Amoeba life cycle
Ingestion of mature cyst, troph migrate to intestine or other body sites and multiply. cysts and trophs passed in feces
E histolytica epidemeology
Warm climates, STI, transmission via mechanical vector
E histolytica clinical manifestations
Amoebic dysentery (invasive intestinal amebiasis), extraintestinal amebiasis, often spreads to liver
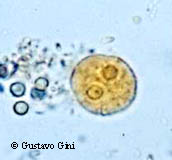
E histolytica cyst
10-20um, Round, cigar shaped chromatoidal bars. 1, 2, or 4 nuclei. Small central karyosome. Even PC
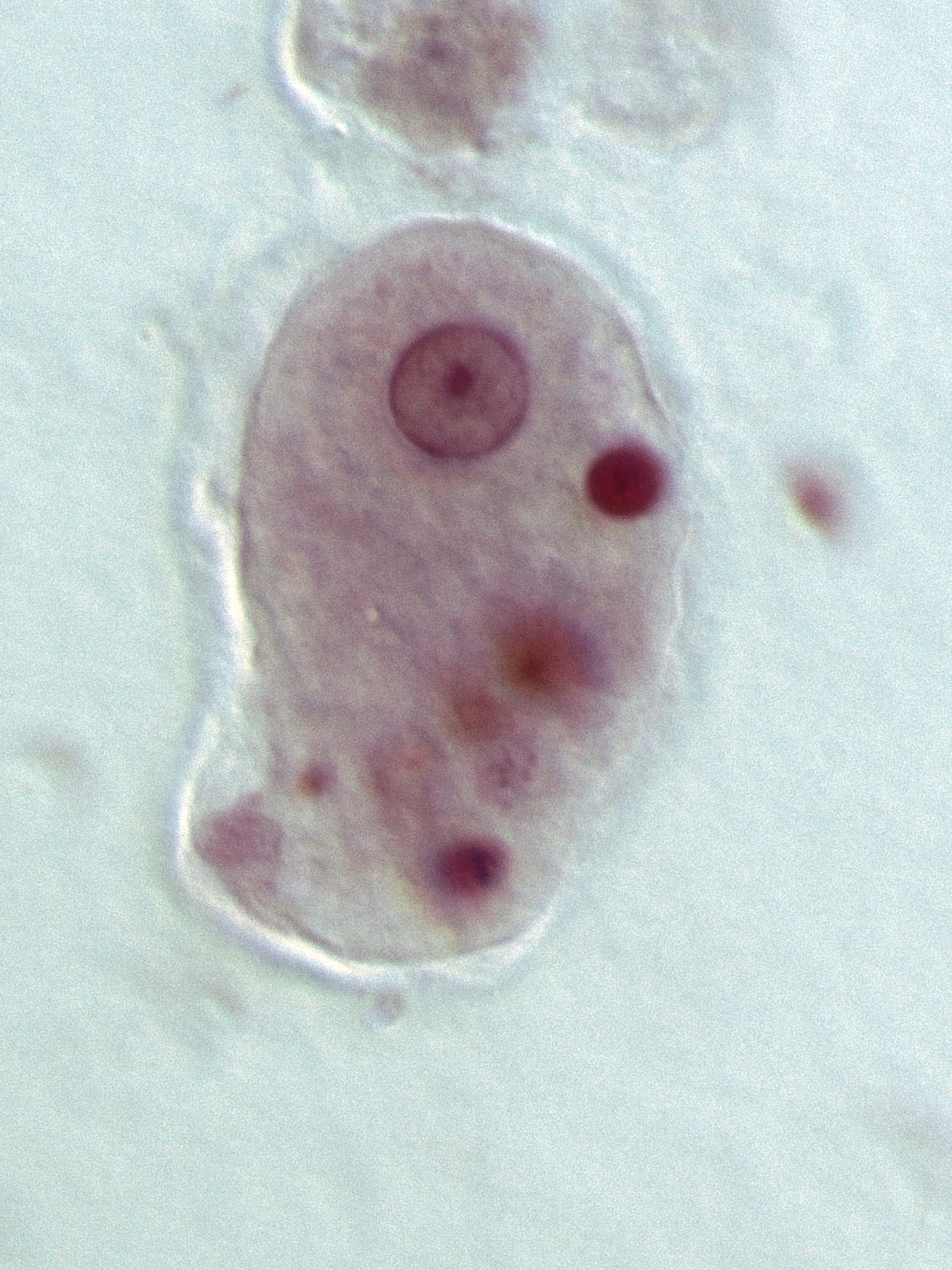
E histolytica troph
15-25 um, One nucleus with even PC and central karyosome, granular cytoplasm, may have ingested RBCs
Charcot leyden crystals
Breakdown product of eosinophils, may appear as chromatoidal bars but they are outside the org
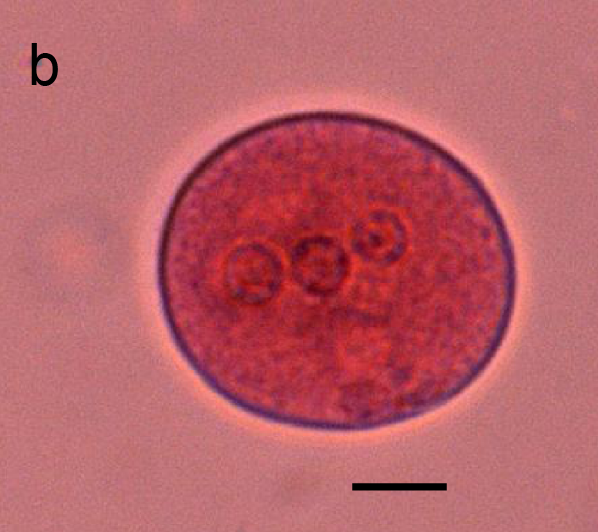
Entamoeba hartmanii cyst
<10 um, 1, 2 or 4 nuclei. “Small race” E. histolytica (even PC, central karyosome, chromatoidal bars)
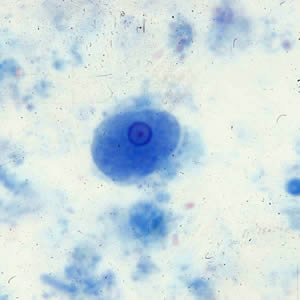
Entamoeba hartmanii troph
3-12 um, 1 nucleus. No ingested RBC
Entamoeba coli cyst
10-35 um, 1-8 nuclei, sharp chromatoidal bars, eccentric karyosome, irregular PC, granular cytoplasm
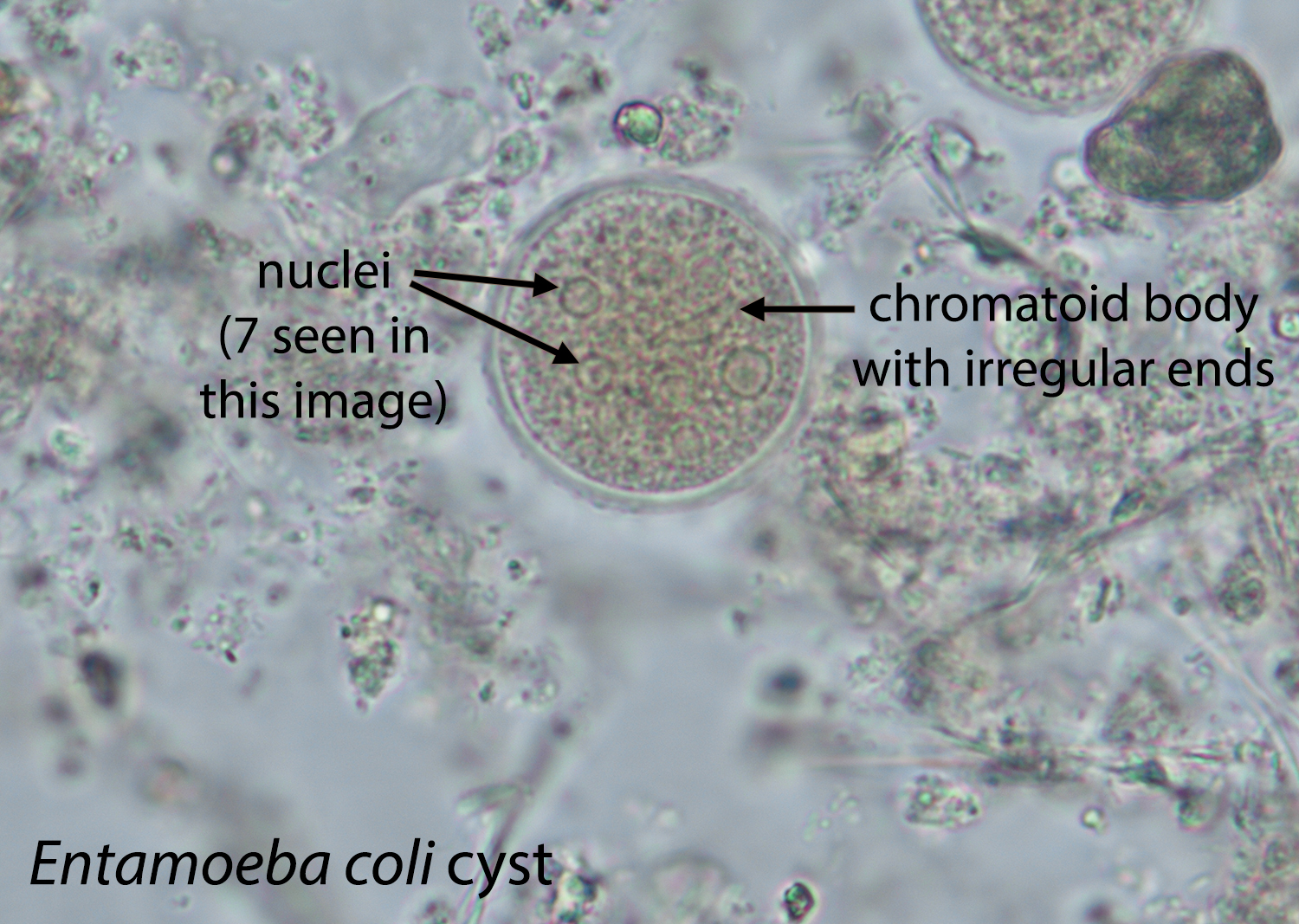
Entamoeba coli troph
12-30um, short blunt pseudopods, dirty cytoplasm/ingested bacteria, 1 nucleus with eccentric karyosome and irregular PC
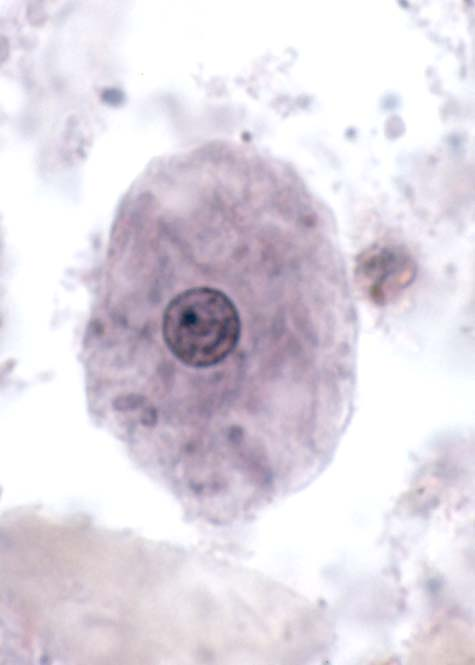
Endolimax nana cyst
15-12 um, 1-4 nuclei, irregular clumped karyosome, no PC, oval
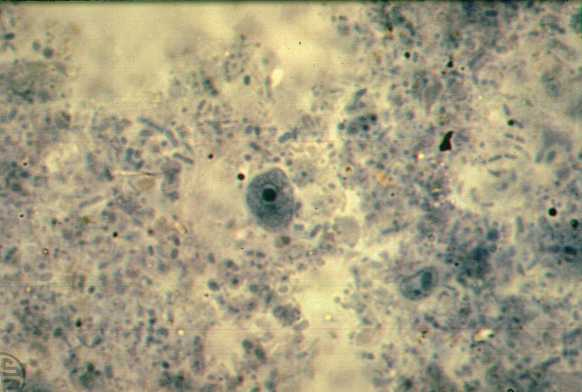
Endolimax nana troph
5-12 um, blunt hyaline pseudopods, large irregular karyosome with no PC
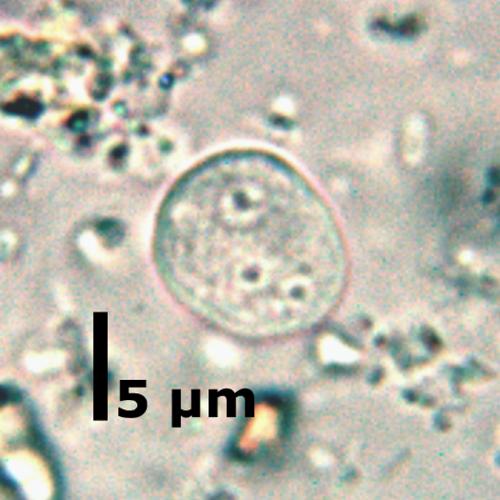
Iodamoeba butschlii cyst
6-15 um, well defined glycogen mass, 1 nucleus with huge karyosome and no PC
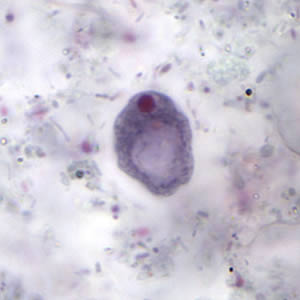
Iodamoeba butschlii troph
Rarely seen, 6-20 um, nucleus similar to E nana
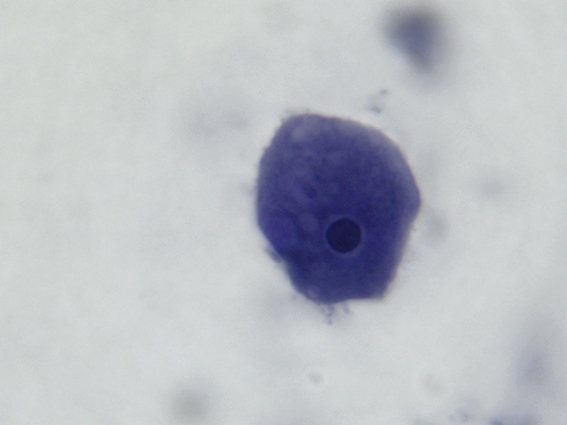
Blastocystis hominis
Lab should report quantitatively. Strict anaerobe, has 4 forms, diagnosis based on finding vacuolar form in stool
Blastocystis hominis vacuolar
5-15 um, up to 4 nuclei, large central vacuole takes up 90% of cell volume, push nuclei to side
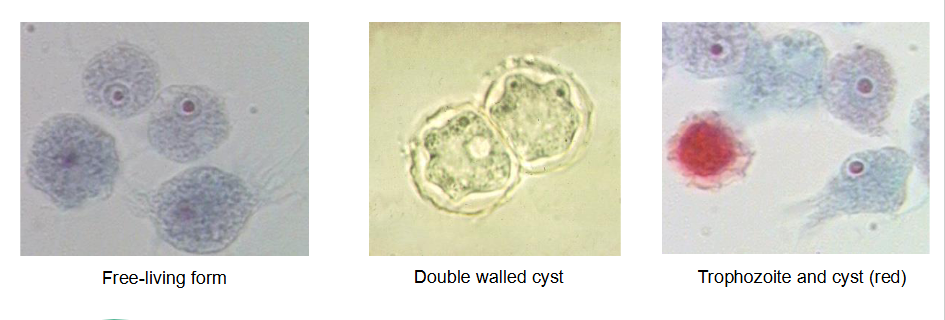
Free living amoeba
Found in many water sources. Acanthamoeba, Balamuthia and Naegleria. We are not a part of their life cycle
GAE
Granulomatous amoebic encephalitis. Acanthamoeba and Balamuthia
PAM
Primary amoebic meningoencephalitis. Naegleria
Naegleria fowleri
Free living amoeba, Causes PAM, only troph found in CSF, lives in warm water. Don’t usually ID
Acanthamoeba
Free living amoeba, causes GAE, contaminated contact lens use. Cyst is doubled walled, troph has central prominent karyosome
Balamuthia mandrillaris
Free living amoeba, causes GAE, found in soil and dust
What flagellates lack a cyst stage?
Dientamoeba fragilis, Trichomonas vaginalis, and Pentatrichomonas hominis
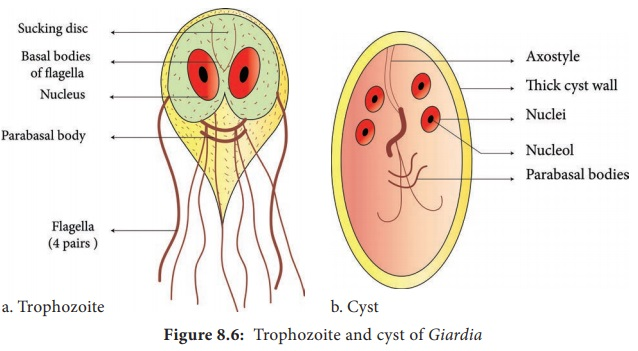
Flagellate structure
Troph flagella arise from basal body, cytostome (mouth), undulating membrane. Cyst has axostyle. Both have parabasal bodies
Giardia epidemiology
Intestinal flagellate, nicknamed beaver fever (bodies of water), highly contagious (infectious dose <10 cysts)
Giardia dx
DFA (direct fluorescent antibody), EIA (enzyme immunoassay). It is a reportable disease
Giardia cyst
9-12 um, oval, 2-4 nuclei, central axostyle, 4 median bodies, internal flagella. Cytoplasm often shrinks from cyst wall

Giardia troph
10-20 um, 2 axostyles and 2 median bodies, ventral sucking disc for attachment, 4 pairs of flagella (8 total), 2 nuclei with large central karyosomes, no PC.
Dientamoeba
Troph form only, looks like amoeba but is a flagellate
Dientamoeba transmission/CM
Ingestion of helminth egg, infection stays in intestine
What characteristic motility feature of giardia?
Falling leaf
Dientamoeba troph
6-15um, Binucleate, no external flagella. Large karyosome, no PC. May have vacuoles with ingested bacteria
Chilomastix cyst
6-10 um, Lemon shaped with nipple like projection, large single nucleus with distinct central karyosome. Prominent cytostomal fibers (shephards crook)
Chilomastix troph
6-20 um, Pear shaped, curved and twisted posteriorly, large single nucleus with distinct central karyosome, cytostomal fibrils
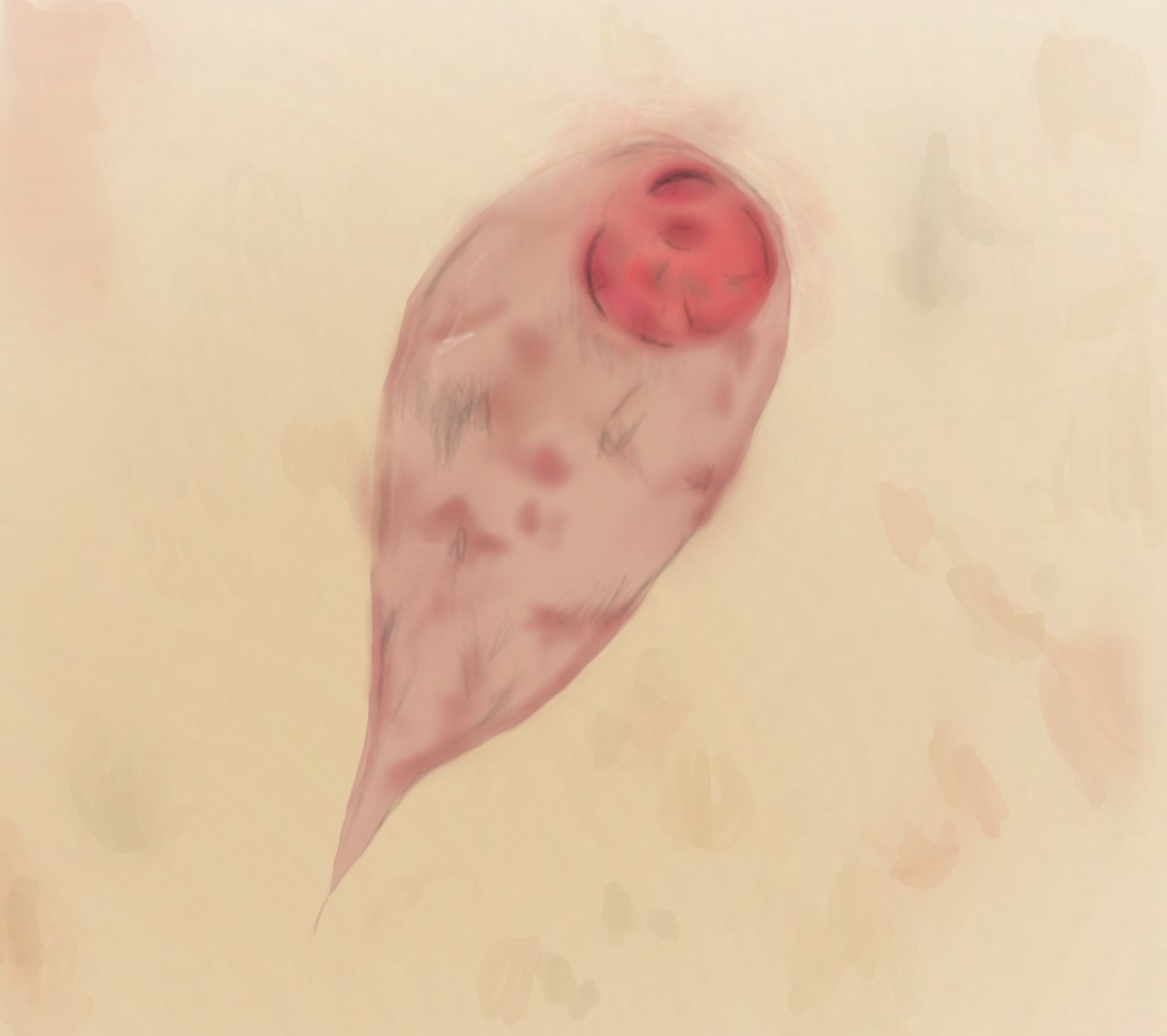
Pentatrichomonas hominis troph
6-14 um, teardrop shaped, quick jerky movement undulating membrane and axostyle through body. No cyst form
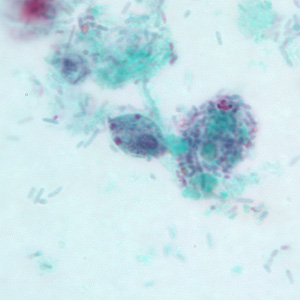
Enteromonas hominis cyst
Ellipsoidal, 4-8um. 1, 2 or 4 nuclei (binucleate most common), large central karyosome, no PC
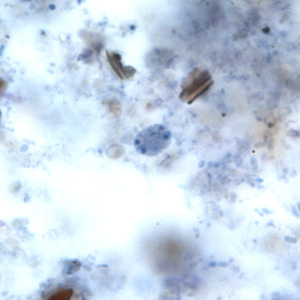
Intestinal flagellates
Giardia and dientamoeba
Atrial (Urethral and vagina) flagellates
Trichomonas vaginalis
Non pathogenic flagellates
Chilomastix, Rerotamonas, pentatrichomonas, enteromonas
Enteromonas hominis troph
Oval, 4-10 um, 3 anterior flagella 1 posterior, one side of body is flattened, jerky motility
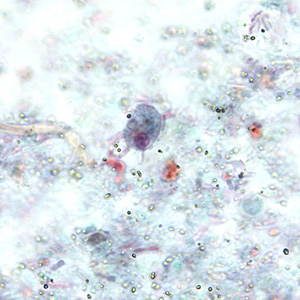
Retortamonas intestinalis cyst
4×6 um, pear shaped (resembles chilomastix), single large nucleus, 2 fibrils (bird beak arrangement)
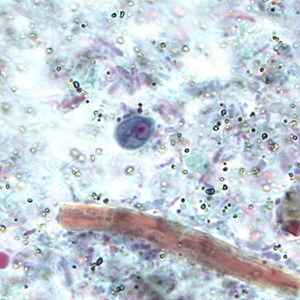
retortamonas intestinalis troph
4-9 um, Pear/oval shaped, jerky movement, promininent cytostomal groove extending half of body, exterior flagella, one nucleus
Trichomonas overview
STI, Worldwide, multiplies in genitourinary tract, male is resevoir
Trichomoniasis
Caused by trichomonas, symptoms are itching and discharge for women, men often don’t experience symptoms
Trichomonas diagnosis
Molecular testing, wet prep, urinalysis, gram stain
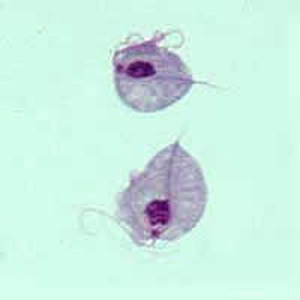
Trichomonas vaginalis troph
7-23 um, jerky non directional movement, 4 anterior flagella, short undulating membrane, long axostyle, large nucleus and small kinetoplast. No cyst
Balantidium coli overview
Intestinal ciliate, only one pathogenic to humans. Largest protozoan parasite. Pig is natural host, human is accidental host. Most common in Philippines
Balantidium coli CM
Severe infections termed Balantidiasis, ulceration in intestines
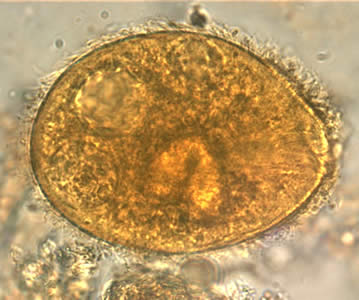
Balantidium coli cyst
45-65 um, round to elliptical shape. Cilia inside double cyst wall. Nuclei, macro (kidney shaped) and micronucleus
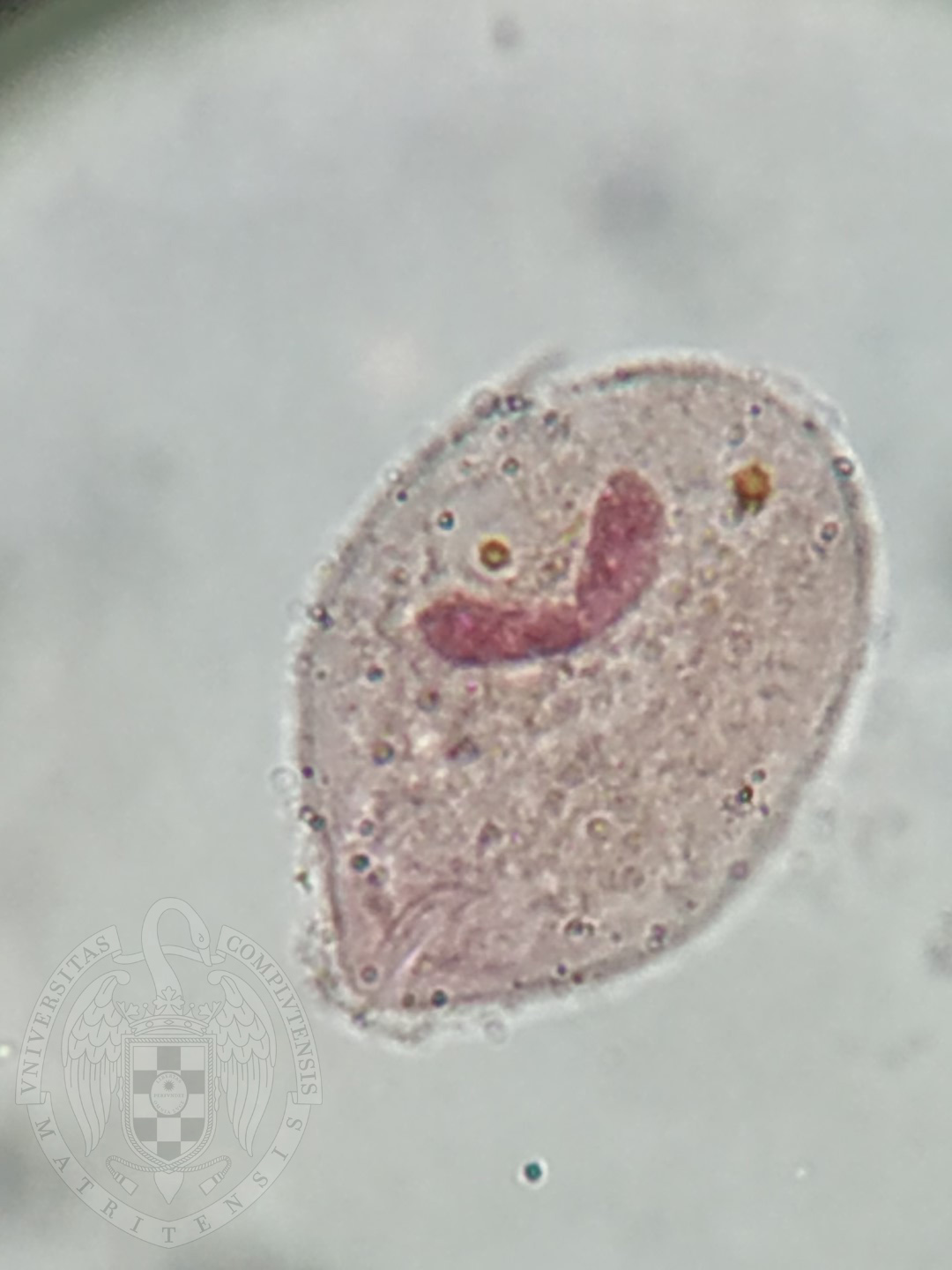
Balantidium coli troph
30-100 um, cilia present along periphery, anterior end is tapered with cytostome. Contractile vacuoles in cytoplasm. 2 nuclei with macro (kidney shaped) and micronucleus
Blood and tissue flagellates
Leishmania and Trypanosoma. Transmitted by insect vector, which is the intermediate host and required for their life cycle
What flagellate forms are found in humans?
Amsatigote and trypomastigote
Amastigote
Found in tissue, seen with Leishmania and Trypanosoma cruzi. Retracted flagellum, kinetoplast and nucleus. 2-3 um, so can only see nucleus on scope
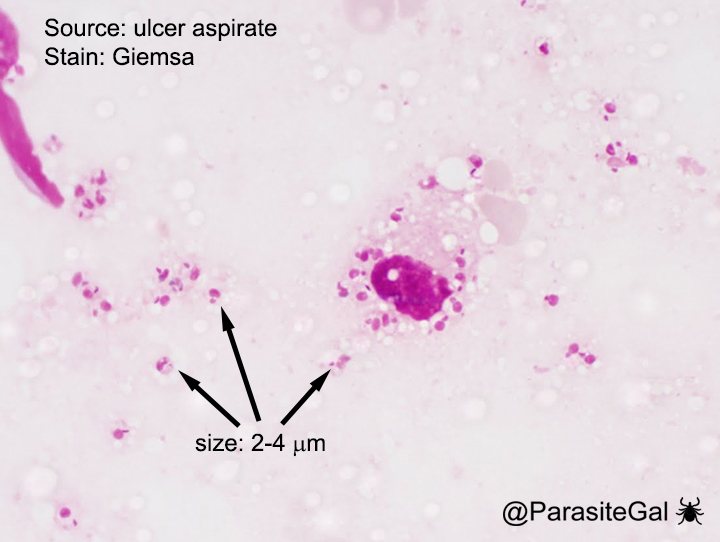
Flagellate development steps
Amastigote, promastigote, epimastigote, trypomastigote
Promastigote
In sand fly, not in humans. anterior kinetoplast where anterior flagella attaches. central nucleus
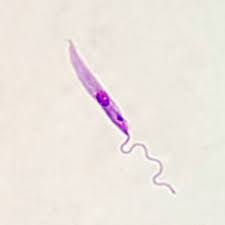
Epimastigote
In tsetse fly and reduviid bug (not in humans). Single anterior flagellum, undulating membrane down half of body that is attached anterior to nucleus. Kinetoplast is anterior to the posterior nucleus
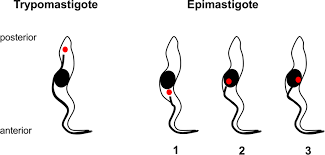
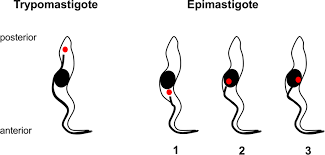
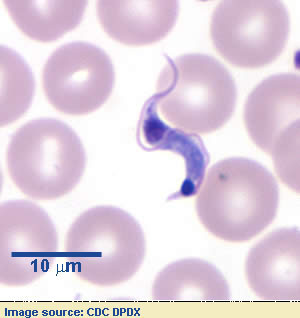
Trypomastigote
In human blood and CSF, Trypanosoma. Free anterior flagellum, undulating membrane on entire bdoy. Kinetoplast is posterior of nucleus
Leishmaniae vector
Sand fly
Cutaneous Leishmaniasis
skin sores, develops within a few weeks or months. Usually painless but can be painful. L. tropica complex, L mexicana complex, and L braziliensis complex
Visceral leishamniasis
Affects internal organs (spleen, liver BM). Can be life threatening. Develops within months to years
Leishmania tropica complex
Old world cutaneous (E hemisphere). Re papule at site, lesions are benign and self healing. secondary bacterial infections common.
Leishmania mexicana complex
New World cutaneous (W hemisphere). 60% occur on ear lobes. Has other mammal resevoirs
Leishmania braziliensis complex
Mucocutaneous. Aggresive chronic cutaneous ulcers with mucous membrane spread. Can be deadly and/or recur years later causing espundia (destruction of nose and ear cartilage)
Leishmania donovani complex
Visceral, AKA Kala-azar. Organisms disseminate and spread to the viscera. Found intracellularly as LD bodies. Death in 2-3 years if untreated
Leishmania life cycle
Promastigote injected into human during sandly meal, promastigote becomes amastigote which replicates and infects other cells
Leishmania diagnosis
Giemsa and H&E stains. Amastigotes of Leishmania and Trypanosoma cruzi are indistinguishable.
African trypanosomiasis
Sleeping sickness. Tsetse fly is insect vector and intermediate host. Includes Trypanosoma brucei rhodesiense (E Africa) and Trypanosoma brucei gambiense (W Africa)
Trypanosoma brucei gambiense
90%, W Africa, mild and more chronic form
Trypanosoma brucei rhodesiense
10%, East Africa. Less prevalent, acute with rapid course and death
Trypanosoma brucei CM
Inflammation at bite site, lymphadenopathy in neck = Winterbottoms sign. CNS involvement causing coma (Sleeping sickness)
African Trypanosomiasis life cycle
Tsetse fly takes blood meal, injects trypomastigote which multiply.
African trypanosomiasis diagnosis
Winterbottoms sign, trypomastigote in blood.
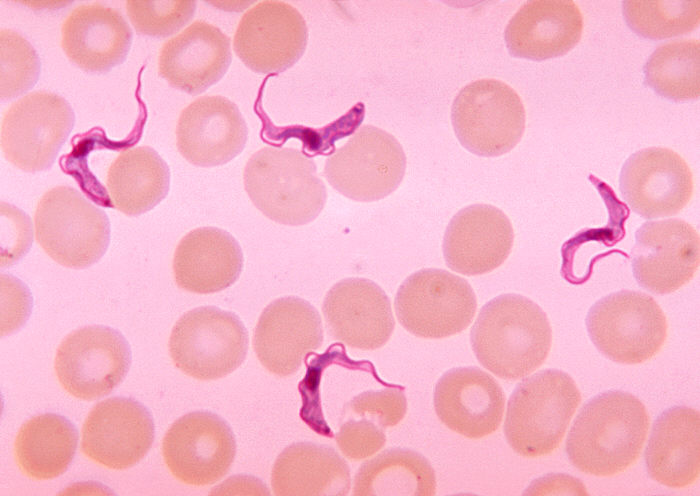
American trypanosomiasis
Chagas disease, caused by Trypanosoma cruzei. Intracellular amastigote develops in tissues, trypomastigote circulates in blood.
American trypanosomiasis transmission
Feces of reuviid bug
American trypanosomiasis CM
Acute: Primary lesion at bite site called chagoma
Chronic: can be decades long with few orgs in the blood.
Symptoms: Edema of eyelids (Romanas sign)
Trypanosoma cruzi life cycle
Reduviid bug takes blood meal, trypomastigote in feces enters wound site. Amastigotes multiply in tissue and some transform into trypomastigotes and circulate in the blood
Trypanosoma cruzi diagnosis
Romanas sign (Unilateral edema of eyelids). Cruzi —> Chagas disease —> Curls (trypomastigote)
Immunoassay of stool sample
Ag-Ab complexes used to detect specific parasites. Rapid detection, specific to an org, but can’t determine number of org
Saline mount of feces
Used to see motility
Iodine mount for feces
Used to see nuclear structures
Trichrome stain
Used to see morphology
Film array (IA)
PCR multiplex w 22 targets. Bacteria, viruses and parasites
ELISA
Ab to capture the Ag (E histolytica). Enzyme linked to secondary Ab
DFA
Ab linked toa fluorocrhome binds cell Ag (cell wall). Giardia, cryptosporidium
Coccidia
Complex life cycles that include sexual and asexual stages. May include intermediate host and definitive host. Fecal oral route, includes oocysts, sporocysts, sporozoites.
Coccidia intermediate host stage
Asexual
Coccidia definitive host stage
Sexual
Cryptosporidium
Most important coccidia, watery diarrhea, increased risk for those with weak immune systems
Cryptosporidium CM
Immunocompetent: incubation (2-10 days), self limiting. General GI symptoms
Immunocompromised: Chronic diarrhea —> dehydration
Cryptosporidium epidemiology
Lives in intestine, infected person sheds parasites in stool. Found all over the world
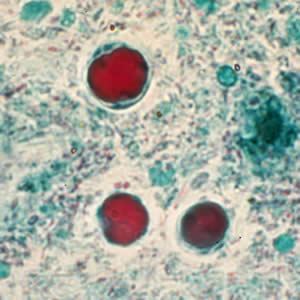
Cryptosporidium oocyst
Infective and diagnostic
Cryptosporidium diagnosis
Acid-fast staining they appear pink and 4-5 um, contains 4 sporozoites. DFA, EIA, PCR
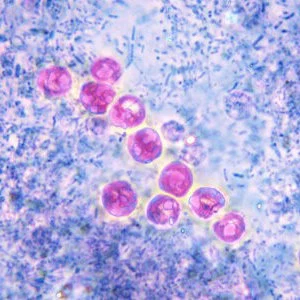
Cyclospora
Coccidia, humans are only known host. Endemic in Nepal, Pakistan, and India
Cyclospora CM
Cyclospora infects small intestine causing self-limiting diarrhea. Sometimes asymptomatic and recurring
Cyclospora life cycle
Oocyst in stool is immature (unsporulated) and not infective. Oocyst sporulates in the environment and gets ingested again
Cyclospora diagnosis
Acid fast they appear light pink to deep red/purple and 8-10um, contain immature sporoblast (bigger than crypto). Cyclospora oocysts are auto fluorescent
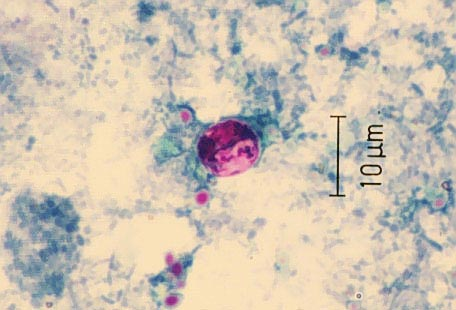
Cystoisospora
Coccidia found worldwide. both sexual and asexual repro occur in GI tract