Midterm #1- Intro to Climate Science
1/35
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
36 Terms
emissivity
a value between 0 and 1 that represents an object’s efficiency in/ability to emit radiation (1 is emitting everything, perfect blackbody)
heat
motion of atoms/molecules (faster=hotter)
power
rate of energy moving (in Joules per second, J/sec a.k.a. watts)
perfect blackbody has emissivity of ___
1
argon makes up _____
1% of the atmosphere
Average energy: E= _____
1/2kT, where k is Boltzmann’s constant (1.38 x 10^-23 J/K) and T is temperature in K (this gives average energy but some molecules will have more and some will have less)
Boltzmann said that energy is ______ within a piece of matter
randomly distributed among molecules
change in _____ induces changes in _____ (perpendicular to each other!)
electricity, magnetism
infrared light has ____ wavelength, ____ frequency, ____ energy
long, low, low
ultraviolet light has ____ wavelength, ____ frequency, ____ energy
short, high, high
outgoing energy depends on ____
temperature
incoming radiation to the Earth is _____, outgoing is ______
short wave, long wave
ozone is a _____ gas that absorbs ____ radiation but not ______ radiation
nonconservative, incoming, outgoing
What two things are considered blackbodies?
solids and deep liquids
What are the three ways a molecule can move to generate heat/internal energy?
translational, rotational, vibrational
molecules that _______ make more efficient greenhouse gases
stay in the atmosphere for a while
_____ blackbody emits more than a ____ blackbody
hot, cold
celerity=
wavelength x frequency
Blackbody radiation
the emissions from a blackbody, with a unique spectrum dependent on temperature
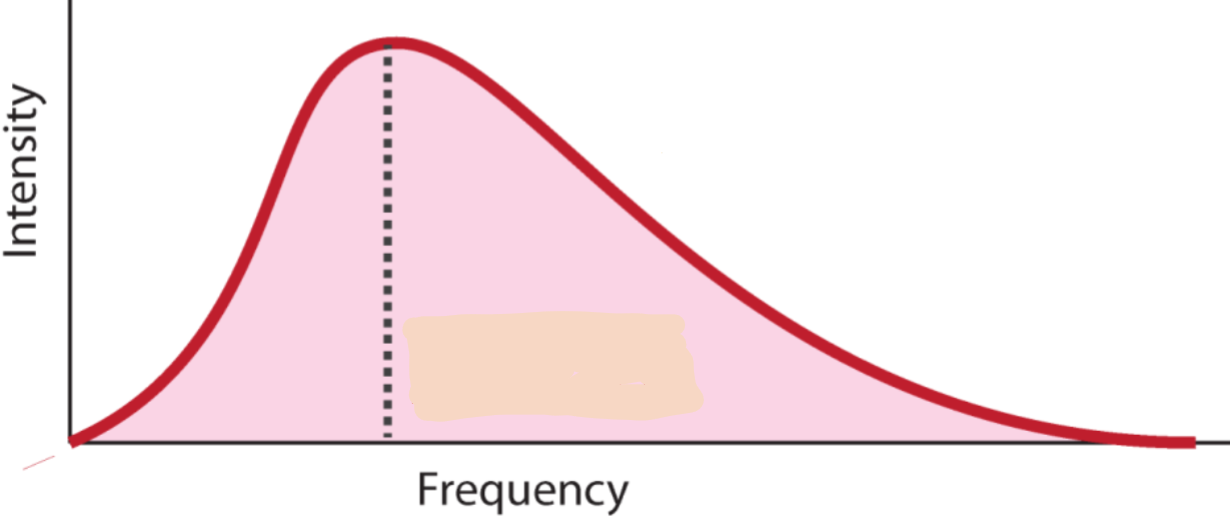
what does this graph represent?
blackbody emissions curve
Flux spectrum of blackbody radiation at
temperature T is determined by quantum
mechanics and the Planck function- formula?
E=hv, where h is Planck’s constant
What 4 things do we need to define a climate?
spatial info (where), temporal info (when/what period of time), variable we’re looking at, that variable’s average with ± variability
climate
average weather and statistical description in terms of the mean and variability over a period of time
there is less seasonal variation over ____ than ____
ocean, land
wavenumber is
one over wavelength
light is made up of _____
many different frequencies traveling together
As a blackbody gets hotter, _____ and ____
Total energy emitted gets larger, peak displacement changes (the composition of energy changes, different peak/most common wavelength)
if _____, then light is absorbed
frequency of light=frequency of matter
What 3 ways can molecules create vibrational energy?
symmetrical stretch, asymmetrical stretch, bend
Bulk Silicate Earth Model
Earth was formed from the same nebula as the Sun and other planets in the solar system
Elemental abundances are estimated from composition of chondritic meteorites and mantle peridotites
albedo
the proportion of light that an object reflects (value from 0, total absorption, to 1, total reflection)
grey gas is identical blocks of _________ and transparent to _____ incoming energy
noncompressible energy